Abstract
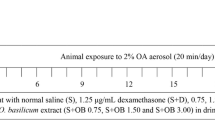
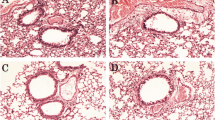
The objective of this study is to detect the effect of beta-glucan derived from Aureobasidium pullulans SM-2001, a UV induced mutant of A. pullulans on the ovalbumin (OVA) induced allergic asthma. The test articles were orally administered to OVA-inducing asthmatic mice 4 days after sensitization for 13 days at 31.25, 62.5 or 125 mg/kg levels. Three days after the OVA sensitization, ten mice were selected per group based on body weight and were sacrificed three days after the OVA aerosol challenge. The changes on the body weight, lung weight, total leukocytes in peripheral blood and total cells in bronchoalveolar lavage fluid (BALF) were observed with changes on the lung histopathology and histomorphometry. The results were compared with dexamethasone (DEXA) 3 mg/kg intraperitoneally treated mice. The results showed increases of body weight after the OVA aerosol challenge, lung weight, total leukocytes and eosinophils in peripheral blood, total cell numbers, neutrophil and eosinophils in BALF were detected in the OVA control compared to sham control (non-OVA). However, these changes from asthmatic responses were significantly or dose-dependently decreased in the beta-glucan-dosing groups compared to those of the OVA control. Therefore, it is concluded that beta-glucan has favorable effects on asthmatic response induced by OVA. It was found that beta-glucan 125 mg/kg showed similar or slightly lower efficacy compared with DEXA 3 mg/kg.
Similar content being viewed by others
References
Adcock, I. M., Caramori, G., and Kirkham, P. A., Strategies for improving the efficacy and therapeutic ratio of glucocorticoids. Curr. Opin. Pharmacol., 12, 1–6 (2012).
Agusti, C., Takeyama, K., Cardell, L. O., Ueki, I., Lausier, J., Lou, Y. P., and Nadel, J. A., Goblet cell degranulation after antigen challenge in sensitized guinea pigs. Role of neutrophils. Am. J. Respir. Crit. Care Med., 158, 1253–1258 (1998).
Allen, S., Britton, J. R., and Leonardi-Bee, J. A., Association between antioxidant vitamins and asthma outcome measures: systematic review and meta-analysis. Thorax, 64, 610–619 (2009).
Babu, N. P., Pandikumar, P., and Ignacimuthu, S., Anti-inflammatory activity of AlbizialebbeckBenth., an ethnomedicinal plant, in acute and chronic animal models of inflammation. J. Ethnopharmacol., 125, 356–360 (2009).
Bani, D., Ballati, L., Masini, E., Bigazzi, M., and Sacchi, T. B., Relaxin counteracts asthma-like reaction induced antigen in sensitized guinea pigs. Endocrinology, 138, 1909–1915 (1997).
Broekema, M., Timens, W., Vonk, J. M., Volbeda, F., Lodewijk, M. E., Hylkema, M. N., Ten Hacken, N. H. T., and Postma, D. S., Persisting remodeling and less airway wall eosinophil activation in complete remission of asthma. Am. J. Respir. Crit. Care Med., 183, 310–316 (2011).
Ceyhan, Y., Sungur, M., Celikel, C. A., and Ceilikel, T., Effects of inhaled cyclosporin on the rat air way: Histologic and bronchoalveolar lavage assessment. Respiration, 65, 71–78 (1998).
Chen, Y., Xie, Q. M., Yang, Q. H., and Chen, J. Q., Effect of inhaled cyclosporin A on antigen-induced airway inflammation in asthmatic rats. Yao Xue Xue Bao, 39, 486–490 (2004).
Choi, H.-Y., Jung, T.-Y., Ku, S.-K., Yang, H.-B., and Lee, H.-S., Toxico-pathological study of p,p-DDE after experimental aerosol exposed to ICR Mouse. J. Toxicol. Pub. Health, 21, 151–160 (2005).
Comhair, S. and Erzurum, S. C., Redox control of asthma molecular mechanisms and therapeutic opportunities. Antioxid. Redox Signal., 12, 93–124 (2010).
Czop, J. K., The role of beta-glucan receptors on blood and tissue leukocytes in phagocytosis and metabolic activation. Pathol. Immunopathol. Res., 5, 286–296 (1986).
Dimeloe, S., Nanzer, A., Ryanna, K., and Hawrylowicz, C., Regulatory T cells, inflammation and the allergic response-The role of glucocorticoids and Vitamin D. J. Steroid Biochem. Mol. Biol., 120, 86–95 (2010).
Douwes, J., Zuidhof, A., Doekes, G., van der Zee, S. C., Wouters, I., Boezen, M. H., and Brunekreef, B., (1→3)-beta-D-Glucan and endotoxin in house dust and peak flow variability in children. Am. J. Respir. Crit. Care Med., 162, 1348–1354 (2000).
Drazen, J. M. and Austen, K. F., Pulmonary response to antigen infusion in the sensitized quinea pig: Modification by atropine. J. Appl. Physiol., 39, 916–919 (1975).
El Khoury, D., Cuda, C., Luhovyy, B. L., and Anderson, G. H., Beta glucan: health benefits in obesity and metabolic syndrome. J. Nutr. Metab., 2012, 851362 (2012), Doi:10.1155 /2012/851362.
Fahlbusch, B., Koch, A., Douwes, J., Bischof, W., Gehring, U., Richter, K., Wichmann, H. E., and Heinrich, J., The effect of storage on allergen and microbial agent levels in frozen house dust. Allergy, 58, 150–153 (2003).
Gong, J.-H., Shin, D., Han, S.-Y., Kim, J.-L., and Kang, Y.-H., Kaempferol suppresses eosionphil infiltration and airway inflammation in airway epithelial cells and in mice with allergic asthma. J. Nutr., 142, 47–56 (2012).
Grainge, C. L., Lau, L. C. K., Ward, J. A., Dulay, V., Lahiff, G., Wilson, S., Holgate, S., Davies, D. E., and Howarth, P. H., Effect of bronchoconstriction on airway remodeling in asthma. N. Engl. J. Med., 364, 2006–2015 (2011).
Hessel, E. M., Van Oosterhout, A. J., Hofsta, C. L., De Bie, J. J., Garssen, J., Van Loveren, H., Verheyen, A. K., Savelkoul, H. F., and Nijkamp, F. P., Bronchoconstriction and airway hyperresponsiveness after ovalbumin inhalation in sensitized mice. Eur. J. Pharmacol., 293, 401–412 (1995).
Holgate, S. T., The Airway epithelium is central to the pathogenesis of asthma. Allergol. Int., 57, 1–10 (2008).
Im, S.-A., Lee, Y.-R., Lee, Y.-H., Lee, M.-K., Park, Y.-I., Lee, S.-W., Kim, K.-J., and Lee, C.-K., In vivo evidence of the immunomodulatory activity of orally administered Aloe vera gel. Arch. Pharm. Res., 33, 451–456 (2010).
Instanes, C., Ormstad, H., Rydjord, B., Wiker, H. G., and Hetland, G., Mould extracts increase the allergic response to ovalbumin in mice. Clin. Exp. Allergy, 34, 1634–1641 (2004).
Johansson, A. K., Sergejeva, S., Sjostrand, M., Lee, J.-J., and Lotvall, J., Allergen-induced traffic of bone marrow eosinophils, neutrophils and lymphocytes to airways. Eur. J. Immunol., 34, 3135–3145 (2004).
Kim, H.-D., Cho, H.-R., Moon, S.-B., Shin, H.-D., Yang, K.-J., Park, B.-R., Jang, H.-J., Kim, L.-S., Lee, H.-S., and Ku, S.-K., Effect of exopolymers from Aureobasidumpullulans on formalin-induced chronic paw inflammation in mice. J. Microbiol. Biotechnol., 16, 1954–1960 (2006).
Kim, H.-D., Cho, H.-R., Moon, S.-B., Shin, H.-D., Yang, K.-J., Park, B.-R., Jang, H.-J., Kim, L.-S., Lee, H.-S., and Ku, S.-K., Effects of beta-glucan from Aureobasidumpullulans on acute inflammation in mice. Arch. Pharm. Res., 30, 323–328 (2007).
Ku, S.-K., Lee, H.-S., Kim, J.-D., Choi, H.-Y., and Lee, J.-H., Investigation of in vivo efficacy of Rufloxacin against Streptococcus pneumoniae respiratory infection, a histological perspective. J. Basic Sci., 6, 45–54 (2002).
Kwon, O.-K., Ahn, K.-S., Lee, M.-Y., Kim, S.-Y., Park, B.-Y., Kim, M.-K., Lee, I.-Y., Oh, S.-R., and Lee, H.-K., Inhibitory effect of kefiran on ovalbumin-induced lung inflammation in a murine model of asthma. Arch. Pharm. Res., 31, 1590–1596 (2008).
Leclerc, N., Noh, T., Khokhar, A., Smith, E., and Frenkel, B., Glucocorticoids inhibit osteocalcin transcription in osteoblasts by suppressing Egr2/Krox20-binding enhancer. Arthritis Rheum., 52, 929–939 (2005).
Leung, S. Y., Eynott, P., Nath, P., and Chung, K. F., Effects of ciclesonide and fluticasone propionate on allergen-induced airway inflammation and remodeling features. J. Allergy Clin. Immunol., 115, 989–996 (2005).
Mackin, W. M., BrunkeReese, D., Gu, Y., Crotty, C., and Fisette, L., Enhanced microbial activities of human peripheral blood monocytes and neutrophils (PMN) after pre-treatment with PGG-glucan (BetafectinTM). FASEB J., 8, A216 (1994).
Maeda, H., Yahata, T., Satouchi, M., Takenaka, K., and Yokoyama, M., Inhibitory effect of low molecular weight heparin on the bronchoconstriction in ovalbumin-sensitized guinea pigs after antigen exposure. Arerugi, 44, 1282–1289 (1995).
Masihi, K. N., Madaj, K., Hintelmann, H., Gast, G., and Kaneko, Y., Down-regulation of tumor necrosis factor-á, moderate reduction of interleukin-1beta, but not interleukin-6 or interleukin-10, by glucanimmunomodulatorscurdlan sulfate and lentinan. Int. J. Immunopharmacol., 19, 463–468 (1997).
Nagao, K., Akabane, H., Masuda, T., Komai, M., Tanaka, H., and Nagai, H., Effect of MX-68 on airway inflammation and hyperresponsiveness in mice and guinea-pigs. J. Pharm. Pharmacol., 56, 187–196 (2004).
Nakata, J., Tamaoki, J., Takeyama, K., Takeda, Y., Yamakawa, I., Kondo, M., and Nagai, A., Effect of heparin on airway goblet cell secretion in sensitized guinea pigs. Nihon KokyukiGakkaiZasshi, 36, 845–850 (1998).
Piper, S. L., Laron, D., Manzano, G., Pattnaik, T., Liu, X., Kim, H.-T., and Feeley, B. T., A comparison of lidocaine, ropivacaine and dexamethasone toxicity on bovine tenocytes in culture. J. Bone Joint Surg. Br., 94, 856–862 (2012).
Poon, A. H., Eidelman, D. H., Martin, J. G., Laprise, C., and Hamid, Q., Pathogenesis of severe asthma. Clin. Exp. Allergy, 42, 625–637 (2012).
Poutsiaka, D. D., Mengozzi, M., Vannier, E., Sinha, B., and Dinarello, C. A., Cross-linking of the beta-glucan receptor on human monocytes results in interleukin-1 receptor antagonist but not interleukin-1 production. Blood, 82, 3695–3700 (1993).
Reber, L. L., Daubeuf, F., Plantinga, M., De Cauwer, L., Gerlo, S., Waelput, W., Van Calenbergh, S., Tavernier, J., Haegeman, G., Lambrecht, B. N., Frossard, N., and De Bosscher, K., A dissociated glucocorticoid receptor modulator reduces airway hyperresponsiveness and inflammation in a mouse model of asthma. J. Immunol., 188, 3478–3487 (2012).
Roh, G.-S., Seo, S.-W., Yeo, S., Lee, J.-M., Choi, J.-W., Kim, E., Shin, Y., Cho, C., Bae, H., Jung, S.-K., and Kwack, K., Efficacy of a traditional Korean medicine, Chung-Sang-Bo-Ha-Tang, in a murine model of chronic asthma. Int. Immunopharmacol., 5, 427–436 (2005).
Roos, A. B. and Nord, M., The emerging role of C/EBPs in glucocorticoid signaling: lessons from the lung. J. Endocrinol., 212, 291–305 (2012).
Rylander, R. and Lin, R. H., (1→3)-beta-D-Glucan-relationship to indoor air-related symptoms, allergy and asthma. Toxicology, 152, 47–52 (2000).
Savoie, C., Plant, M., Zwikker, M., Van Staden, C. J., Boulet, L., Chan, C. C., Rodger, I. W., and Pon, D. J., Effect of dexamethasone on antigen-induced high molecular weight glycoconjugatesecretionin allergic guinea pigs. Am. J. Respir. Cell Mol. Biol., 13, 133–143 (1995).
Senoglu, N., Yuzbasioglu, M. F., Aral, M., Ezberci, M., Kurutas, E. B., Bulbuloglu, E., Ezberci, F., Oksuz, H., and Ciragil, P., Protective effects of N-acetylcysteine and betaglucan pretreatment on oxidative stress in cecal ligation and puncture model of sepsis. J. Invest. Surg., 21, 237–243 (2008).
Seo, H.-P., Kim, J.-M., Shin, H.-D., Kim, T.-K., Chang, H.-J., Park, B.-R., and Lee, J.-W., Production of beta-1,3/1,6-glucan by Aureobasidiumpullulans SM-2001. Korean J. Biotechnol. Bioeng., 17, 376–380 (2002).
Soltys, J. and Quinn, M. T., Modulation of endotoxin- and enterotoxin-induced cytokine release by in vivo treatment with beta-(1,6)-branched beta-(1,3)-glucan. Infect. Immun., 67, 244–252 (1999).
Takeda, K., Thurman, J. M., Tomlinson, S., Okamoto, M., Shiraishi, Y., Ferreira, V. P., Cortes, C., Pangburn, M. K., Holers, V. M., and Gelfand, E. W., The critical role of complement alternative pathway regulator factor H in allergen-induced airway hyperresponsiveness and inflammation. J. Immunol., 188, 661–667 (2012).
Temelkovski, J., Hogan, S. P., Shepherd, D. P., Foster, P. S., and Kumar, R. K., An improved murine model of asthma: selective airway inflammation, epithelial lesions and increased methacholine responsiveness following chronic exposure to aerosolised allergen. Thorax, 53, 849–856 (1998).
Trifilieff, A., Walker, C., Keller, T., Kottirsch, G., and Neumann, U., Pharmacological profile of PKF242-484 and PKF241-466, novel dual inhibitors of TNF-alpha converting enzyme and matrix metalloproteinases, in models of airway inflammation. Br. J. Pharmacol., 135, 1655–1664 (2002).
Waddell, A. N., Patel, S. K., Toma, A. G., and Maw, A. R., Intranasal steroid sprays in the treatment of rhinitis: is one better than another. J. Laryngol. Otol., 117, 843–845 (2003).
Wilson, R. H., Whitehead, G. S., Nakano, H., Free, M. E., Kolls, J. K., and Cook, D. N., Allergic sensitization through the airway primes Th17-dependent neutrophilia and airway hyperresponsiveness. Am. J. Respir. Crit Care Med., 180, 720–730 (2009).
Xu, X. J., Yasuda, M., Tsuruta, S. N., Mizuno, M., and Ashida, H., β-Glucan from Lentinusedodesinhibits nitric oxide and tumor necrosis factor-á production and phosphorylation of mitogen-activated protein kinases in lipopolysaccharide-stimulated murine RAW 264.7 macrophages. J. Biol. Chem., 287, 871–878 (2012).
Yeh, Y. F. and Huang, S. L., Enhancing effect of dietary cholesterol and inhibitory effect of pravastatin on allergic pulmonary inflammation. J. Biomed. Sci., 11, 599–606 (2004).
Zhang, G. S. and Shen, H. H., The preventive effect of bacillus Calmette-Guerin vaccination in early life on airway inflammation and mucus production in a murine model of asthma. Zhonghua Jie He He Hu Xi Za Zhi, 28, 17–21 (2005).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ku, S.K., Kim, J.W., Cho, H.R. et al. Effect of β-glucan originated from Aureobasidium pullulans on asthma induced by ovalbumin in mouse. Arch. Pharm. Res. 35, 1073–1081 (2012). https://doi.org/10.1007/s12272-012-0615-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12272-012-0615-8