Abstract
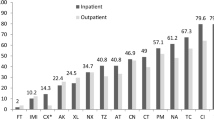
Uropathogenic Escherichia coli (UPEC) isolates contain large genomic segments, termed pathogenicity islands (PAIs), that contribute to their virulence. A total of 150 UPEC and 50 commensal E. coli isolates from outpatients were investigated for antimicrobial susceptibility and the presence of eight PAI markers. One hundred ninety (95 %) isolates were resistant to one or more antimicrobial agents. The most frequent resistance found against amoxicillin (68 %), amoxicillin/clavulanic acid (55 %), aztreonam (50 %), trimethoprim/sulfamethoxazole (46 %) and tetracycline (43.5 %). Antimicrobial resistance among UPEC isolates was higher than that of commensals. PAI markers were detected in substantial percentage of commensal (88 %) and UPEC isolates (98.6 %) (P > 0.05). The most prevalent PAI marker among UPEC and commensal isolates was PAI IV536 (98.7 % UPEC vs. 84 % commensal). We found a high number of PAI markers such as PAI ICFT073, PAI IICFT073, PAI I536, PAI II536, PAI III536 and PAI IIJ96 significantly associated with UPEC. PAI III536 (21.3 %) and PAI IIJ96 (8 %) were detected only in the uropathogenic isolates. Several different combinations of PAIs were found among UPEC isolates. Comparison of PAIs among UPEC and commensal isolates showed that many UPEC isolates (79.3 %) carried two or more PAI markers, while 6 % of commensals had two PAI markers (P < 0.05). The most frequent combinations of PAI markers in UPEC isolates were PAI IV536 + PAI IICFT073 (18 %) and PAI IV536 + PAI ICFT073 + PAI IICFT073 (18 %). These results indicate that PAI markers are widespread among commensal and UPEC isolates and these commensal isolates may be reservoirs for transmission of these markers.

Similar content being viewed by others
References
Ananias M, Yano T (2008) Serogroups and virulence genotypes of Escherichia coli isolated from patients with sepsis. Braz J Med Biol L Res 41(10):877–883
Che D, Hasan MS, Chen B (2014) Identifying pathogenicity islands in bacterial pathogenomics using computational approaches. Pathogens 3:36–56
Clinical and Laboratory Standards Institute (2013) Performance standards for antimicrobial susceptibility testing; 23th informational supplement. CLSI document M100-S23. Vol. 31 No.1
Copur-Cicek A, Ozgumus OB, Saral A, Sandalli C (2014) Antimicrobial resistance patterns and integron carriage of Escherichia coli isolates causing community-acquired infections in Turkey. Ann Lab Med 34:139–144
Dobrindt U, Blum-Oehle G, Nagy G, Schneider G, Johann A, Gottschalk G (2002) Genetic structure and distribution of four pathogenicity islands (PAI I536 to PAI IV536) of uropathogenic Escherichia coli strain 536. Infect Immun 70(11):6365–6372
Farshad S, Ranjbar R, Japoni A, Hosseini M, Anvarinejad M, Mohammadzadegan R (2012) Microbial susceptibility, virulence factors, and plasmid profiles of uropathogenic Escherichia coli strains isolated from children in Jahrom, Iran. Arch Iran Med 15(5):312–316
Gal-Mor O, Finlay BB (2006) Pathogenicity islands: a molecular toolbox for bacterial virulence. Cell Microbiol 8(11):1707–1719
Hacker J, Bender L, Ott M, Wingender J, Lund B, Marre R, Goebel W (1990) Deletions of chromosomal regions coding for fimbriae and hemolysins occur in vitro and in vivo in various extraintestinal Escherichia coli isolates. Microbial pathogenesis 8(3):213–225
Koga VL, Tomazetto G, Cyoia P S, Neves M S, Vidotto M C, Nakazato G, Kobayashi R K T (2014) Molecular screening of virulence genes in extraintestinal pathogenic Escherichia coli isolated from human blood culture in Brazil. Biomed Res Int Article, ID 465054; http://dx.doi.org/10.1155/2014/465054.
Li B, Sun JY, Han LZ, Huang XH, Fu Q, Ni YX (2010) Phylogenetic groups and pathogenicity island markers in fecal Escherichia coli isolates from asymptomatic humans in China. Appl Environ Microb 76(19):6698–6700
Middendorf B, Hochhut B, Leipold K, Dobrindt U, Blum-Oehler G, Hacker J (2004) Instability of pathogenicity islands in uropathogenic Escherichia coli 536. J Bacteriol 186:3086–3096
Murugan K, Savitha T, Vasanthi S (2012) Retrospective study of antibiotic resistance among uropathogens from rural teaching hospital, Tamilnadu, India. Asian Pacific J Tropical Dis 2(5):375–380
Navidinia M, Najar Peerayeh S, Fallah F, Bakhshi B (2013a) Phylogenetic groups and pathogenicity island markers in Escherichia coli isolated from children. Jundishapur J Microbiol 6(10):e8362
Navidinia M, Najar Peerayeh S, Fallah F, Bakhshi B, Adabian S, Alimehr S, Gholinejad Z (2013b) Distribution of the pathogenicity islands markers (PAIs) in uropathogenic E.coli isolated from children in Mofid children hospital, Pediatric Infections Research Center. Arch Pediatr Infect Dis 1(2):75–79
Neamati F, Firoozeh F, Saffari M, Zibaei M (2015) Virulence genes and antimicrobials resistance pattern in uropathogenic Escherichia coli isolated from hospitalized patients in Kashan, Iran. Jundishapur J Microbiol 8(2):e17514
Oliveira FA, Paludo KS, Arend LN, Farah SM, Pedrosa FO, Souza EM, Surek M, Picheth G, Fadel-Picheth CM (2011) Virulence characteristics and antimicrobial susceptibility of uropathogenic Escherichia coli strains. Genet Mol Res 10(4):4114–4125
Östblom A, Adlerberth I, Wold AE, Nowrouzian FL (2011) Pathogenicity island markers, virulence determinants malX and usp, and the capacity of Escherichia coli to persist in infants’ commensal microbiotas. Appl Environ Microb 77(7):2303–2308
Phongpaichit S, Tunyapanit W, Pruekprasert P (2011) Antimicrobial resistance, class I integrons and extended spectrum betalactamases in E. coli clinical isolates from patients in south Thailand. J Health Sci 57:281–288
Pobiega M, Wojkowska-Mach J, Chmielarczyk A, Romaniszyn D, Adamski P, Heczko PB et al (2013) Molecular characterization and drug resistance of Escherichia coli strains isolated from urine from long-term care facility residents in Cracow, Poland. Med Sci Monit 19:317–326
Ravi A, Avershina E, Ludvigsen J, L’Abée-Lund TM, Rudi K (2014) Integrons in the intestinal microbiota as reservoirs for transmission of antibiotic resistance genes. Pathogens 3:238–248
Rezaee MA, Sheikhalizadeh V, Hasani A (2011) Detection of integrons among multi-drug resistant (MDR) Escherichia coli strains isolated from clinical specimens in northern west of Iran. Braz J Microbiol 42(4):1308–1313
Rice LB (2009) The clinical consequences of antimicrobial resistance. Curr Opin Microbiol 12:476–481
Sabaté M, Moreno E, Pérez T, Andreu A, Prats G (2006) Pathogenicity island markers in commensal and uropathogenic Escherichia coli isolates. Clin Microbiol Infect 12:880–886
Schmidt H, Hensel M (2004) Pathogenicity islands in bacterial pathogenesis. Clin Microbiol Rev 17(1):14–56
Sepp E, Stsepetova J, Lõivukene K, Truusalu K, Kõljalg S, Naaber P, Mikelsaar M (2009) The occurrence of antimicrobial resistance and class 1 integrons among commensal Escherichia coli isolates from infants and elderly persons. Annals of Clinical Microbiology and Antimicrobials 8:34
Su J, Shi L, Yang L, Xiao Z, Li X, Yamasaki S (2006) Analysis of integrons in clinical isolates of Escherichia coli in China during the last six years. FEMS Microbiol Lett 254:75–80
Wenting J, Jinling S, Magaly T, Shaohua Z, Jianghong M (2013) Distribution of pathogenicity islands OI-122, OI-43/48, and OI-57 and a high-pathogenicity island in shiga toxin-producing Escherichia coli. Appl Environ Microb 79(11):3406–3412
Wozniak RAF, Waldor MK (2010) Integrative and conjugative elements: mosaic mobile genetic elements enabling dynamic lateral gene flow. Nat Rev Microbiol 8(8):552–563
Yoon SH, Hur CG, Kang HY, Kim YH, Oh TK, Kim JF (2005) A computational approach for identifying pathogenicity islands in prokaryotic genomes. BMC Bioinformatics 6:184
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Samei, A., Haghi, F. & Zeighami, H. Distribution of pathogenicity island markers in commensal and uropathogenic Escherichia coli isolates. Folia Microbiol 61, 261–268 (2016). https://doi.org/10.1007/s12223-015-0433-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12223-015-0433-8