Abstract
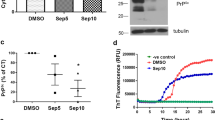
Prion diseases are transmissible and invariably fatal neurodegenerative disorders, which still lack of efficacious prophylactic and therapeutic tools. Our previous study has proposed that the natural phytoalexin, resveratrol, can reduce the amounts of PrPSc in a scrapie-infected cell line SMB-S15. To address its anti-prion efficacy, the inhibitive activity of resveratrol on prion accumulation in vitro and prion infectivity in vivo was analyzed in the present study. Exposure of SMB-S15 cells to various concentrations of resveratrol (0.25 to 200 μM) reduced and even removed cellular PrPSc in a dose-dependent manner, with EC50 0.61 μM. Meanwhile, PrPSc signals in SMB-S15 cells treated with 5 and 10 μM resveratrol maintained undetectable after drug withdrawal, indicating that the removal of PrPSc in SMB-S15 cells by resveratrol is irreversible. Furthermore, the lysates of SMB-S15 cells exposed to 10 μM resveratrol for 2 and 7 days were intracerebrally inoculated into CD1 mice. All mice (n = 9) infected with SMB-S15 cells without treatment of resveratrol appeared typical experimental scrapie symptoms from 155 to 228 day post inoculation (dpi), while all mice (n = 9) inoculated with SMB-S15 cells treated with resveratrol for 7 days maintained healthy by the end of observations (284 dpi). PrP-specific Western blots and neuropathological tests did not identify PrPSc or prion disease-associated pathological abnormality in the brains of mice inoculated with 7-day resveratrol-treated SMB-S15 cells. It indicates that the prion infectivity of SMB-S15 onto CD1 mice is eradicated by 1-week resveratrol treatment. Sensitivity of PrPSc to resveratrol highlights its potential role in prion therapeutics.








Similar content being viewed by others
References
Prusiner SB (1998) Prions. Proc Natl Acad Sci U S A 95(23):13363–13383
Collinge J (2001) Prion diseases of humans and animals: their causes and molecular basis. Annu Rev Neurosci 24:519–550. doi:10.1146/annurev.neuro.24.1.519
Aguzzi A (2006) Prion diseases of humans and farm animals: epidemiology, genetics, and pathogenesis. J Neurochem 97(6):1726–1739. doi:10.1111/j.1471-4159.2006.03909.x
Aguzzi A, Heikenwalder M, Miele G (2004) Progress and problems in the biology, diagnostics, and therapeutics of prion diseases. J Clin Invest 114(2):153–160. doi:10.1172/JCI22438
Marandi Y, Farahi N, Sadeghi A, Sadeghi-Hashjin G (2012) Prion diseases—current theories and potential therapies: a brief review. Folia Neuropathol Assoc Polish Neuropathol Med Res Cent Polish Acad Sci 50(1):46–49
Soleas GJ, Diamandis EP, Goldberg DM (1997) Resveratrol: a molecule whose time has come? And gone? Clin Biochem 30(2):91–113
Bhat KPL, Kosmeder JW 2nd, Pezzuto JM (2001) Biological effects of resveratrol. Antioxid Redox Signal 3(6):1041–1064. doi:10.1089/152308601317203567
Baur JA, Sinclair DA (2006) Therapeutic potential of resveratrol: the in vivo evidence. Nat Rev Drug Discov 5(6):493–506. doi:10.1038/nrd2060
Pervaiz S, Holme AL (2009) Resveratrol: its biologic targets and functional activity. Antioxid Redox Signal 11(11):2851–2897. doi:10.1089/ars.2008.2412
Albani D, Polito L, Signorini A, Forloni G (2010) Neuroprotective properties of resveratrol in different neurodegenerative disorders. Biofactors 36(5):370–376. doi:10.1002/biof.118
Rege SD, Geetha T, Griffin GD, Broderick TL, Babu JR (2014) Neuroprotective effects of resveratrol in Alzheimer disease pathology. Front Aging Neurosci 6:218. doi:10.3389/fnagi.2014.00218
de Oliveira RM, Pais TF, Outeiro TF (2010) Sirtuins: common targets in aging and in neurodegeneration. Curr Drug Targets 11(10):1270–1280
Donmez G (2013) Sirtuins as possible targets in neurodegenerative diseases. Curr Drug Targets 14(6):644–647
Jeong JK, Moon MH, Bae BC, Lee YJ, Seol JW, Kang HS, Kim JS, Kang SJ et al (2012) Autophagy induced by resveratrol prevents human prion protein-mediated neurotoxicity. Neurosci Res 73(2):99–105. doi:10.1016/j.neures.2012.03.005
Jeong JK, Moon MH, Lee YJ, Seol JW, Park SY (2013) Autophagy induced by the class III histone deacetylase Sirt1 prevents prion peptide neurotoxicity. Neurobiol Aging 34(1):146–156. doi:10.1016/j.neurobiolaging.2012.04.002
Bizat N, Peyrin JM, Haik S, Cochois V, Beaudry P, Laplanche JL, Neri C (2010) Neuron dysfunction is induced by prion protein with an insertional mutation via a Fyn kinase and reversed by sirtuin activation in Caenorhabditis elegans. J Neurosci 30(15):5394–5403. doi:10.1523/JNEUROSCI.5831-09.2010
Seo JS, Moon MH, Jeong JK, Seol JW, Lee YJ, Park BH, Park SY (2012) SIRT1, a histone deacetylase, regulates prion protein-induced neuronal cell death. Neurobiol Aging 33(6):1110–1120. doi:10.1016/j.neurobiolaging.2010.09.019
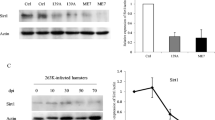
Wang J, Zhang J, Shi Q, Zhang BY, Chen C, Chen LN, Sun J, Wang H et al (2014) Scrapie infection in experimental rodents and SMB-S15 cells decreased the brain endogenous levels and activities of Sirt1. J Molecul Neurosci. doi:10.1007/s12031-014-0459-4
Haig DA, Clarke MC (1971) Multiplication of the scrapie agent. Nature 234(5324):106–107
Gao JM, Gao C, Han J, Zhou XB, Xiao XL, Zhang J, Chen L, Zhang BY et al (2004) Dynamic analyses of PrP and PrP(Sc) in brain tissues of golden hamsters infected with scrapie strain 263K revealed various PrP forms. Biomed Environ Sci 17(1):8–20
Shi Q, Zhang BY, Gao C, Zhang J, Jiang HY, Chen C, Han J, Dong XP (2012) Mouse-adapted scrapie strains 139A and ME7 overcome species barrier to induce experimental scrapie in hamsters and changed their pathogenic features. Virol J 9:63. doi:10.1186/1743-422X-9-63
Sakaguchi S, Katamine S, Shigematsu K, Nakatani A, Moriuchi R, Nishida N, Kurokawa K, Nakaoke R et al (1995) Accumulation of proteinase K-resistant prion protein (PrP) is restricted by the expression level of normal PrP in mice inoculated with a mouse-adapted strain of the Creutzfeldt-Jakob disease agent. J Virol 69(12):7586–7592
Daude N, Marella M, Chabry J (2003) Specific inhibition of pathological prion protein accumulation by small interfering RNAs. J Cell Sci 116(Pt 13):2775–2779. doi:10.1242/jcs.00494
Prusiner SB (1982) Novel proteinaceous infectious particles cause scrapie. Science 216(4542):136–144
Sim VL, Caughey B (2009) Recent advances in prion chemotherapeutics. Infect Disord Drug Targets 9(1):81–91
Wang Q, Xu J, Rottinghaus GE, Simonyi A, Lubahn D, Sun GY, Sun AY (2002) Resveratrol protects against global cerebral ischemic injury in gerbils. Brain Res 958(2):439–447
Mokni M, Elkahoui S, Limam F, Amri M, Aouani E (2007) Effect of resveratrol on antioxidant enzyme activities in the brain of healthy rat. Neurochem Res 32(6):981–987. doi:10.1007/s11064-006-9255-z
Kim JI, Choi SI, Kim NH, Jin JK, Choi EK, Carp RI, Kim YS (2001) Oxidative stress and neurodegeneration in prion diseases. Ann N Y Acad Sci 928:182–186
Pamplona R, Naudí A, Gavín R, Pastrana MA, Sajnani G, Ilieva EV, del Río JA, Portero-Otín M et al (2008) Increased oxidation, glycoxidation, and lipoxidation of brain proteins in prion disease. Free Rad Biol Med 45(8):1159–1166. doi:10.1016/j.freeradbiomed.2008.07.009
Chen D, Steele AD, Hutter G, Bruno J, Govindarajan A, Easlon E, Lin SJ, Aguzzi A et al (2008) The role of calorie restriction and SIRT1 in prion-mediated neurodegeneration. Exp Gerontol 43(12):1086–1093. doi:10.1016/j.exger.2008.08.050
Acknowledgments
This work was supported by Chinese National Natural Science Foundation Grants (81100980, 81301429), SKLID Development Grant (2013SKLID402, 2012SKLID102), and China Mega-Project for Infectious Disease (2011ZX10004-101, 2012ZX10004215).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
None declared.
Rights and permissions
About this article
Cite this article
Wang, J., Zhang, BY., Zhang, J. et al. Treatment of SMB-S15 Cells with Resveratrol Efficiently Removes the PrPSc Accumulation In Vitro and Prion Infectivity In Vivo. Mol Neurobiol 53, 5367–5376 (2016). https://doi.org/10.1007/s12035-015-9464-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-015-9464-z