Abstract
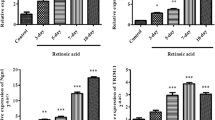
SH-SY5Y cells, a neuroblastoma cell line that is a well-established model system to study the initial phases of neuronal differentiation, have been used in studies to elucidate the mechanisms of neuronal differentiation. In the present study, we investigated alterations of gene expression in SH-SY5Y cells during neuronal differentiation mediated by retinoic acid (RA) treatment. We evaluated important pathways involving nuclear factor kappa B (NF-κB), nuclear E2-related factor 2 (Nrf2), glycolytic, and p53 during neuronal differentiation. We also investigated the involvement of reactive oxygen species (ROS) in modulating the gene expression profile of those pathways by antioxidant co-treatment with Trolox®, a hydrophilic analogue of α-tocopherol. We found that RA treatment increases levels of gene expression of NF-κB, glycolytic, and antioxidant pathway genes during neuronal differentiation of SH-SY5Y cells. We also found that ROS production induced by RA treatment in SH-SY5Y cells is involved in gene expression profile alterations, chiefly in NF-κB, and glycolytic pathways. Antioxidant co-treatment with Trolox® reversed the effects mediated by RA NF-κB, and glycolytic pathways gene expression. Interestingly, co-treatment with Trolox® did not reverse the effects in antioxidant gene expression mediated by RA in SH-SY5Y. To confirm neuronal differentiation, we quantified endogenous levels of tyrosine hydroxylase, a recognized marker of neuronal differentiation. Our data suggest that during neuronal differentiation mediated by RA, changes in profile gene expression of important pathways occur. These alterations are in part mediated by ROS production. Therefore, our results reinforce the importance in understanding the mechanism by which RA induces neuronal differentiation in SH-SY5Y cells, principally due this model being commonly used as a neuronal cell model in studies of neuronal pathologies.




Similar content being viewed by others
References
Fineberg SK, Kosik KS, Davidson BL (2009) MicroRNAs potentiate neural development. Neuron 64(3):303–309. doi:10.1016/j.neuron.2009.10.020
Chen X, Du Z, Shi W, Wang C, Yang Y, Wang F, Yao Y, He K, Hao A (2013) 2-Bromopalmitate modulates neuronal differentiation through the regulation of histone acetylation. Stem Cell Res 12(2):481–491. doi:10.1016/j.scr.2013.12.010
Hadjal Y, Hadadeh O, Yazidi CE, Barruet E, Binetruy B (2013) A p38MAPK-p53 cascade regulates mesodermal differentiation and neurogenesis of embryonic stem cells. Cell Death Dis 4:e737. doi:10.1038/cddis.2013.246
Zhao F, Wu T, Lau A, Jiang T, Huang Z, Wang XJ, Chen W, Wong PK, Zhang DD (2009) Nrf2 promotes neuronal cell differentiation. Free Radic Biol Med 47(6):867–879. doi:10.1016/j.freeradbiomed.2009.06.029
de Bittencourt Pasquali MA, Gelain DP, Zeidan-Chulia F, Pires AS, Gasparotto J, Terra SR, Moreira JC (2013) Vitamin A (retinol) downregulates the receptor for advanced glycation endproducts (RAGE) by oxidant-dependent activation of p38 MAPK and NF-kB in human lung cancer A549 cells. Cell Signal 25(4):939–954. doi:10.1016/j.cellsig.2013.01.013
Salminen A, Kauppinen A, Kaarniranta K (2012) Phytochemicals suppress nuclear factor-kappaB signaling: impact on health span and the aging process. Curr Opin Clin Nutr Metab Care 15(1):23–28. doi:10.1097/MCO.0b013e32834d3ae7
Bhakar AL, Tannis LL, Zeindler C, Russo MP, Jobin C, Park DS, MacPherson S, Barker PA (2002) Constitutive nuclear factor-kappa B activity is required for central neuron survival. J Neurosci 22(19):8466–8475
O'Neill LA, Kaltschmidt C (1997) NF-kappa B: a crucial transcription factor for glial and neuronal cell function. Trends Neurosci 20(6):252–258
Kaltschmidt B, Sparna T, Kaltschmidt C (1999) Activation of NF-kappa B by reactive oxygen intermediates in the nervous system. Antioxid Redox Signal 1(2):129–144
Lane DP (1992) Cancer. p53, guardian of the genome. Nature 358(6381):15–16. doi:10.1038/358015a0
Mukhopadhyay UK, Mak AS (2009) p53: is the guardian of the genome also a suppressor of cell invasion? Cell Cycle 8(16):2481
Gaub P, Tedeschi A, Puttagunta R, Nguyen T, Schmandke A, Di Giovanni S (2010) HDAC inhibition promotes neuronal outgrowth and counteracts growth cone collapse through CBP/p300 and P/CAF-dependent p53 acetylation. Cell Death Differ 17(9):1392–1408. doi:10.1038/cdd.2009.216
Harris JJ, Jolivet R, Attwell D (2012) Synaptic energy use and supply. Neuron 75(5):762–777. doi:10.1016/j.neuron.2012.08.019
Campanucci V, Krishnaswamy A, Cooper E (2010) Diabetes depresses synaptic transmission in sympathetic ganglia by inactivating nAChRs through a conserved intracellular cysteine residue. Neuron 66(6):827–834. doi:10.1016/j.neuron.2010.06.010
Gutteridge JM, Halliwell B (2000) Free radicals and antioxidants in the year 2000. A historical look to the future. Ann N Y Acad Sci 899:136–147
Wang B, Zhu X, Kim Y, Li J, Huang S, Saleem S, Li RC, Xu Y, Dore S, Cao W (2012) Histone deacetylase inhibition activates transcription factor Nrf2 and protects against cerebral ischemic damage. Free Radic Biol Med 52(5):928–936. doi:10.1016/j.freeradbiomed.2011.12.006
Yan W, Wang HD, Hu ZG, Wang QF, Yin HX (2008) Activation of Nrf2-ARE pathway in brain after traumatic brain injury. Neurosci Lett 431(2):150–154. doi:10.1016/j.neulet.2007.11.060
Ramsey CP, Glass CA, Montgomery MB, Lindl KA, Ritson GP, Chia LA, Hamilton RL, Chu CT, Jordan-Sciutto KL (2007) Expression of Nrf2 in neurodegenerative diseases. J Neuropathol Exp Neurol 66(1):75–85. doi:10.1097/nen.0b013e31802d6da9
da Frota Junior ML, Pires AS, Zeidan-Chulia F, Bristot IJ, Lopes FM, de Bittencourt Pasquali MA, Zanotto-Filho A, Behr GA, Klamt F, Gelain DP, Moreira JC (2011) In vitro optimization of retinoic acid-induced neuritogenesis and TH endogenous expression in human SH-SY5Y neuroblastoma cells by the antioxidant Trolox. Mol Cell Biochem 358(1–2):325–334. doi:10.1007/s11010-011-0983-2
Albanus RD, Juliani Siqueira Dalmolin R, Alves Castro MA, Augusto de Bittencourt Pasquali M, de Miranda Ramos V, Pens Gelain D, Fonseca Moreira JC (2013) Reverse engineering the neuroblastoma regulatory network uncovers MAX as One of the master regulators of tumor progression. PLoS One 8(12):e82457
Xun Z, Lee DY, Lim J, Canaria CA, Barnebey A, Yanonne SM, McMurray CT (2012) Retinoic acid-induced differentiation increases the rate of oxygen consumption and enhances the spare respiratory capacity of mitochondria in SH-SY5Y cells. Mech Ageing Dev 133(4):176–185. doi:10.1016/j.mad.2012.01.008
Castro MA, Filho JL, Dalmolin RJ, Sinigaglia M, Moreira JC, Mombach JC, de Almeida RM (2009) ViaComplex: software for landscape analysis of gene expression networks in genomic context. Bioinformatics 25(11):1468–1469. doi:10.1093/bioinformatics/btp246
von Mering C, Jensen LJ, Snel B, Hooper SD, Krupp M, Foglierini M, Jouffre N, Huynen MA, Bork P (2005) STRING: known and predicted protein-protein associations, integrated and transferred across organisms. Nucleic Acids Res 33:D433–D437. doi:10.1093/nar/gki005, Database issue
Birney E, Andrews D, Caccamo M, Chen Y, Clarke L, Coates G, Cox T, Cunningham F, Curwen V, Cutts T, Down T, Durbin R, Fernandez-Suarez XM, Flicek P, Graf S, Hammond M, Herrero J, Howe K, Iyer V, Jekosch K, Kahari A, Kasprzyk A, Keefe D, Kokocinski F, Kulesha E, London D, Longden I, Melsopp C, Meidl P, Overduin B, Parker A, Proctor G, Prlic A, Rae M, Rios D, Redmond S, Schuster M, Sealy I, Searle S, Severin J, Slater G, Smedley D, Smith J, Stabenau A, Stalker J, Trevanion S, Ureta-Vidal A, Vogel J, White S, Woodwark C, Hubbard TJ (2006) Ensembl. Nucleic Acids Res 34:D556–D561. doi:10.1093/nar/gkj133, Database issue
Hooper SD, Bork P (2005) Medusa: a simple tool for interaction graph analysis. Bioinformatics 21(24):4432–4433. doi:10.1093/bioinformatics/bti696
Iyengar BR, Choudhary A, Sarangdhar MA, Venkatesh KV, Gadgil CJ, Pillai B (2014) Non-coding RNA interact to regulate neuronal development and function. Front Cell Neurosci 8:47. doi:10.3389/fncel.2014.00047
Biedler JL, Helson L, Spengler BA (1973) Morphology and growth, tumorigenicity, and cytogenetics of human neuroblastoma cells in continuous culture. Cancer Res 33(11):2643–2652
Mattson MP (2005) NF-kappaB in the survival and plasticity of neurons. Neurochem Res 30(6–7):883–893. doi:10.1007/s11064-005-6961-x
Casaccia-Bonnefil P, Carter BD, Dobrowsky RT, Chao MV (1996) Death of oligodendrocytes mediated by the interaction of nerve growth factor with its receptor p75. Nature 383(6602):716–719. doi:10.1038/383716a0
Carter BD, Kaltschmidt C, Kaltschmidt B, Offenhauser N, Bohm-Matthaei R, Baeuerle PA, Barde YA (1996) Selective activation of NF-kappa B by nerve growth factor through the neurotrophin receptor p75. Science 272(5261):542–545
Burke MA, Bothwell M (2003) p75 neurotrophin receptor mediates neurotrophin activation of NF-kappa B and induction of iNOS expression in P19 neurons. J Neurobiol 55(2):191–203. doi:10.1002/neu.10174
Heissmeyer V, Krappmann D, Wulczyn FG, Scheidereit C (1999) NF-kappaB p105 is a target of IkappaB kinases and controls signal induction of Bcl-3-p50 complexes. EMBO J 18(17):4766–4778. doi:10.1093/emboj/18.17.4766
Dechend R, Hirano F, Lehmann K, Heissmeyer V, Ansieau S, Wulczyn FG, Scheidereit C, Leutz A (1999) The Bcl-3 oncoprotein acts as a bridging factor between NF-kappaB/Rel and nuclear co-regulators. Oncogene 18(22):3316–3323. doi:10.1038/sj.onc.1202717
Gallagher D, Gutierrez H, Gavalda N, O'Keeffe G, Hay R, Davies AM (2007) Nuclear factor-kappaB activation via tyrosine phosphorylation of inhibitor kappaB-alpha is crucial for ciliary neurotrophic factor-promoted neurite growth from developing neurons. J Neurosci 27(36):9664–9669. doi:10.1523/JNEUROSCI.0608-07.2007
Dienel GA (2012) Fueling and imaging brain activation. ASN Neuro 4 (5). doi:10.1042/AN20120021, e00093 [pii], AN20120021 [pii]
Cabrera-Valladares G, German MS, Matschinsky FM, Wang J, Fernandez-Mejia C (1999) Effect of retinoic acid on glucokinase activity and gene expression and on insulin secretion in primary cultures of pancreatic islets. Endocrinology 140(7):3091–3096. doi:10.1210/endo.140.7.6765
Lin YW, Lien LM, Yeh TS, Wu HM, Liu YL, Hsieh RH (2008) 9-cis retinoic acid induces retinoid X receptor localized to the mitochondria for mediation of mitochondrial transcription. Biochem Biophys Res Commun 377(2):351–354. doi:10.1016/j.bbrc.2008.09.122
Truckenmiller ME, Vawter MP, Cheadle C, Coggiano M, Donovan DM, Freed WJ, Becker KG (2001) Gene expression profile in early stage of retinoic acid-induced differentiation of human SH-SY5Y neuroblastoma cells. Restor Neurol Neurosci 18(2–3):67–80
Pasquali MA, Gelain DP, Zanotto-Filho A, de Souza LF, de Oliveira RB, Klamt F, Moreira JC (2008) Retinol and retinoic acid modulate catalase activity in Sertoli cells by distinct and gene expression-independent mechanisms. Toxicol In Vitro 22(5):1177–1183. doi:10.1016/j.tiv.2008.03.007
Gelain DP, de Bittencourt Pasquali MA, Caregnato FF, Zanotto-Filho A, Moreira JC (2008) Retinol up-regulates the receptor for advanced glycation endproducts (RAGE) by increasing intracellular reactive species. Toxicol In Vitro 22(5):1123–1127. doi:10.1016/j.tiv.2008.02.016
Gelain DP, de Bittencourt Pasquali MA, Zanotto-Filho A, de Souza LF, de Oliveira RB, Klamt F, Moreira JC (2008) Retinol increases catalase activity and protein content by a reactive species-dependent mechanism in Sertoli cells. Chem Biol Interact 174(1):38–43. doi:10.1016/j.cbi.2008.04.025
Zanotto-Filho A, Cammarota M, Gelain DP, Oliveira RB, Delgado-Canedo A, Dalmolin RJ, Pasquali MA, Moreira JC (2008) Retinoic acid induces apoptosis by a non-classical mechanism of ERK1/2 activation. Toxicol In Vitro 22(5):1205–1212. doi:10.1016/j.tiv.2008.04.001
Pasquali MA, Gelain DP, Oliveira MR, Behr GA, Motta LL, Rocha RF, Klamt F, Moreira JC (2009) Vitamin A supplementation induces oxidative stress and decreases the immunocontent of catalase and superoxide dismutase in rat lungs. Exp Lung Res 35(5):427–438. doi:10.1080/01902140902747436
Pasquali MA, Gelain DP, de Oliveira MR, Behr GA, da Motta LL, da Rocha RF, Klamt F, Moreira JC (2009) Vitamin A supplementation for different periods alters oxidative parameters in lungs of rats. J Med Food 12(6):1375–1380. doi:10.1089/jmf.2008.0298
Pasquali MA, Schnorr CE, Feistauer LB, Gelain DP, Moreira JC (2010) Vitamin A supplementation to pregnant and breastfeeding female rats induces oxidative stress in the neonatal lung. Reprod Toxicol 30(3):452–456. doi:10.1016/j.reprotox.2010.05.085
de Bittencourt Pasquali MA, Roberto de Oliveira M, De Bastiani MA, da Rocha RF, Schnorr CE, Gasparotto J, Gelain DP, Moreira JC (2012) k-NAME co-treatment prevent oxidative damage in the lung of adult Wistar rats treated with vitamin A supplementation. Cell Biochem Funct 30(3):256–263
Auten RL, O'Reilly MA, Oury TD, Nozik-Grayck E, Whorton MH (2006) Transgenic extracellular superoxide dismutase protects postnatal alveolar epithelial proliferation and development during hyperoxia. Am J Physiol Lung Cell Mol Physiol 290(1):L32–L40. doi:10.1152/ajplung.00133.2005
Dong A, Shen J, Krause M, Akiyama H, Hackett SF, Lai H, Campochiaro PA (2006) Superoxide dismutase 1 protects retinal cells from oxidative damage. J Cell Physiol 208(3):516–526. doi:10.1002/jcp.20683
Holtzclaw WD, Dinkova-Kostova AT, Talalay P (2004) Protection against electrophile and oxidative stress by induction of phase 2 genes: the quest for the elusive sensor that responds to inducers. Adv Enzyme Regul 44:335–367. doi:10.1016/j.advenzreg.2003.11.013
Nguyen T, Nioi P, Pickett CB (2009) The Nrf2-antioxidant response element signaling pathway and its activation by oxidative stress. J Biol Chem 284(20):13291–13295. doi:10.1074/jbc.R900010200
Miao W, Hu L, Scrivens PJ, Batist G (2005) Transcriptional regulation of NF-E2 p45-related factor (NRF2) expression by the aryl hydrocarbon receptor-xenobiotic response element signaling pathway: direct cross-talk between phase I and II drug-metabolizing enzymes. J Biol Chem 280(21):20340–20348. doi:10.1074/jbc.M412081200
Lau A, Villeneuve NF, Sun Z, Wong PK, Zhang DD (2008) Dual roles of Nrf2 in cancer. Pharmacol Res 58(5–6):262–270. doi:10.1016/j.phrs.2008.09.003
Niture SK, Khatri R, Jaiswal AK (2014) Regulation of Nrf2-an update. Free Radic Biol Med 66:36–44. doi:10.1016/j.freeradbiomed.2013.02.008
Chen KF, Chen HL, Tai WT, Feng WC, Hsu CH, Chen PJ, Cheng AL (2011) Activation of phosphatidylinositol 3-kinase/Akt signaling pathway mediates acquired resistance to sorafenib in hepatocellular carcinoma cells. J Pharmacol Exp Ther 337(1):155–161. doi:10.1124/jpet.110.175786
Lopez-Carballo G, Moreno L, Masia S, Perez P, Barettino D (2002) Activation of the phosphatidylinositol 3-kinase/Akt signaling pathway by retinoic acid is required for neural differentiation of SH-SY5Y human neuroblastoma cells. J Biol Chem 277(28):25297–25304. doi:10.1074/jbc.M201869200
Vanlandingham JW, Tassabehji NM, Somers RC, Levenson CW (2005) Expression profiling of p53-target genes in copper-mediated neuronal apoptosis. Neuromolecular Med 7(4):311–324. doi:10.1385/NMM:7:4:311
Culmsee C, Mattson MP (2005) p53 in neuronal apoptosis. Biochem Biophys Res Commun 331(3):761–777. doi:10.1016/j.bbrc.2005.03.149
Nagao M, Campbell K, Burns K, Kuan CY, Trumpp A, Nakafuku M (2008) Coordinated control of self-renewal and differentiation of neural stem cells by Myc and the p19ARF-p53 pathway. J Cell Biol 183(7):1243–1257. doi:10.1083/jcb.200807130, jcb.200807130
Meletis K, Wirta V, Hede SM, Nister M, Lundeberg J, Frisen J (2006) p53 suppresses the self-renewal of adult neural stem cells. Development 133(2):363–369. doi:10.1242/dev.02208
Xavier JM, Morgado AL, Sola S, Rodrigues CM (2014) Mitochondrial translocation of p53 modulates neuronal fate by preventing differentiation-induced mitochondrial stress. Antioxid Redox Signal. doi:10.1089/ars.2013.5417
Acknowledgments
This work was funded by the National Counsel of Technological and Scientific Development (CNPq) (grants: 560221/2010-0, and 470973/2012-9), and FAPERGS (grants: PG 12/1060-6, and IPG 0427-2551/14-0).
Conflict of Interest
The authors declare no conflict of interest
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
de Bittencourt Pasquali, M.A., de Ramos, V.M., Albanus, R.D. et al. Gene Expression Profile of NF-κB, Nrf2, Glycolytic, and p53 Pathways During the SH-SY5Y Neuronal Differentiation Mediated by Retinoic Acid. Mol Neurobiol 53, 423–435 (2016). https://doi.org/10.1007/s12035-014-8998-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-014-8998-9