Abstract
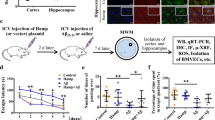
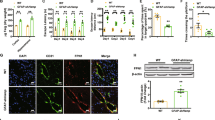
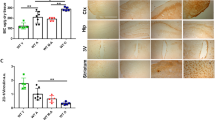
Iron accumulates progressively in the brain with age, and iron-induced oxidative stress has been considered as one of the initial causes for Alzheimer’s disease (AD) and Parkinson’s disease (PD). Based on the role of hepcidin in peripheral organs and its expression in the brain, we hypothesized that this peptide has a role to reduce iron in the brain and hence has the potential to prevent or delay brain iron accumulation in iron-associated neurodegenerative disorders. Here, we investigated the effects of hepcidin expression adenovirus (ad-hepcidin) and hepcidin peptide on brain iron contents, iron transport across the brain–blood barrier, iron uptake and release, and also the expression of transferrin receptor-1 (TfR1), divalent metal transporter 1 (DMT1), and ferroportin 1 (Fpn1) in cultured microvascular endothelial cells and neurons. We demonstrated that hepcidin significantly reduced brain iron in iron-overloaded rats and suppressed transport of transferrin-bound iron (Tf-Fe) from the periphery into the brain. Also, the peptide significantly inhibited expression of TfR1, DMT1, and Fpn1 as well as reduced Tf-Fe and non-transferrin-bound iron uptake and iron release in cultured microvascular endothelial cells and neurons, while downregulation of hepcidin with hepcidin siRNA retrovirus generated opposite results. We concluded that, under iron-overload, hepcidin functions to reduce iron in the brain by downregulating iron transport proteins. Upregulation of brain hepcidin by ad-hepcidin emerges as a new pharmacological treatment and prevention for iron-associated neurodegenerative disorders.








Similar content being viewed by others
Abbreviations
- AD:
-
Alzheimer’s disease
- BMECs:
-
Brain microvascular endothelial cells
- DMT1:
-
Divalent metal transporter 1
- DMT1+IRE:
-
Divalent metal transporter 1 with iron response element
- DMT1−IRE:
-
Divalent metal transporter 1 without iron response element
- Fpn1:
-
Ferroportin 1
- NTBI:
-
Non-transferrin-bound iron
- PKAN:
-
Pantothenate kinase-associated neurodegeneration
- PD:
-
Parkinson’s disease
- Tf-Fe:
-
Transferrin-bound iron
- TfR1:
-
Transferrin receptor
References
Swaiman KF (2001) Hallervorden-Spatz syndrome. Pediatr Neurol 25:102–108
Zhou B, Westaway SK, Levinson B, Johnson MA, Gitschier J, Hayflick SJ (2001) A novel pantothenate kinase gene (PANK2) is defective in Hallervorden-Spatz syndrome. Nat Genet 28:345–349
Angelini L, Nardocci N, Rumi V, Zorzi C, Strada L, Savoiardo M (1992) Hallervorden-Spatz disease: clinical and MRI study of 11 cases diagnosed in life. J Neurol 239:417–425
Hayflick SJ, Penzien JM, Michl W, Sharif U, Rosman N, Wheeler PG (2001) Cranial MRI changes may precede symptoms in Hallervorden-Spatz syndrome. Pediatr Neurol 25:166–169
Rouault TA (2001) Iron on the brain. Nat Genet 28:299–300
Martin WR, Ye FQ, Allen PS (1998) Increasing striatal iron content associated with normal aging. Mov Disord 13:281–286
Ke Y, Qian ZM (2003) Iron misregulation in the brain: a primary cause of neurodegenerative disorders. Lancet Neurol 2:246–253
Zecca L, Youdim MB, Riederer P, Connor JR, Crichton RR (2004) Iron, brain ageing and neurodegenerative disorders. Nat Rev Neurosci 5:863–873
Cass WA, Grondin R, Andersen AH, Zhang Z, Hardy PA, Hussey-Andersen LK, Rayens WS, Gerhardt GA, Gash DM (2007) Iron accumulation in the striatum predicts aging-related decline in motor function in rhesus monkeys. Neurobiol Aging 28:258–271
Hardy PA, Gash D, Yokel R, Andersen A, Ai Y, Zhang Z (2005) Correlation of R2 with total iron concentration in the brains of rhesus monkeys. J Magn Reson Imaging 21:118–127
Aquino D, Bizzi A, Grisoli M, Garavaglia B, Bruzzone MG, Nardocci N, Savoiardo M, Chiapparini L (2009) Age-related iron deposition in the basal ganglia: quantitative analysis in healthy subjects. Radiology 252:165–172
Kastman EK, Willette AA, Coe CL, Bendlin BB, Kosmatka KJ, McLaren DG, Xu G, Canu E, Field AS, Alexander AL, Voytko ML, Beasley TM, Colman RJ, Weindruch RH, Johnson SC (2010) A calorie-restricted diet decreases brain iron accumulation and preserves motor performance in old rhesus monkeys. J Neurosci 30:7940–7947
Bartzokis G, Bartzokis G, Lu PH, Tingus K, Peters DG, Amar CP, Tishler TA, Finn JP, Villablanca P, Altshuler LL, Mintz J, Neely E, Connor JR (2011) Gender and iron genes may modify associations between brain iron and memory in healthy aging. Neuropsychopharmacology 36:1375–1384
Qian ZM, Shen X (2001) Brain iron transport and neurodegeneration. Trends Mol Med 7:103–108
Zecca L, Bellei C, Costi P, Albertini A, Monzani E, Casella L, Gallorini M, Bergamaschi L, Moscatelli A, Turro NJ, Eisner M, Crippa PR, Ito S, Wakamatsu K, Bush WD, Ward WC, Simon JD, Zucca FA (2008) New melanic pigments in the human brain that accumulate in aging and block environmental toxic metals. Proc Natl Acad Sci U S A 105:17567–17572
Dwyer BE, Zacharski LR, Balestra DJ, Lerner AJ, Perry G, Zhu X, Smith MA (2009) Getting the iron out phlebotomy for Alzheimer’s disease? Med Hypotheses 72:504–509
Ke Y, Qian ZM (2007) Brain iron metabolism: neurobiology and neurochemistry. Prog Neurobiol 83(3):149–173
Stankiewicz JM, Brass SD (2009) Role of iron in neurotoxicity: a cause for concern in the elderly? Curr Opin Clin Nutr Metab Care 12:22–29
Nicolas G, Bennoun M, Devaux I, Beaumont C, Grandchamp B, Kahn A, Vaulont S (2001) Lack of hepcidin gene expression and severe tissue iron overload in upstream stimulatory factor 2 (USF2) knockout mice. Proc Natl Acad Sci U S A 98:8780–8785
Park CH, Valore EV, Waring AJ, Ganz T (2001) Hepcidin, a urinary antimicrobial peptide synthesized in the liver. J Biol Chem 276:7806–7810
Pigeon C, Ilyin G, Courselaud B, Leroyer P, Turlin B, Brissot P, Loréal O (2001) A new mouse liver-specific gene, encoding a protein homologous to human antimicrobial peptide hepcidin, is overexpressed during iron overload. J Biol Chem 276:7811–7819
Clardy SL, Wang X, Boyer PJ, Earley CJ, Allen RP, Connor JR (2006) Is ferroportin-hepcidin signaling altered in restless legs syndrome? J Neurol Sci 247:173–179
Zechel S, Huber-Wittmer K, von Bohlen und Halbach O (2006) Distribution of the iron-regulating protein hepcidin in the murine central nervous system. J Neurol Res 84:790–800
Wang Q, Du F, Qian ZM, Ge XH, Zhu L, Yung WH, Yang L, Ke Y (2008) Lipopolysaccharide induces a significant increase in expression of iron regulatory hormone hepcidin in the cortex and substantia nigra in rat brain. Endocrinology 149:3920–3925
Du F, Qian C, Qian ZM, Wu XM, Xie H, Yung WH, Ke Y (2011) Hepcidin directly inhibits transferrin receptor 1 expression in astrocytes via a cyclic AMP-protein kinase A pathway. Glia 59(6):936–945
Chang YZ, Qian ZM, Wang K, Zhu L, Yang XD, Du JR, Jiang L, Ho KP, Wang Q, Ke Y (2005) Effects of development and iron status on ceruloplasmin expression in rat brain. J Cell Physiol 204:623–631
Qian ZM, Chang YZ, Zhu L, Yang L, Du JR, Ho KP, Wang Q, Li LZ, Wang CY, Ge X, Jing NL, Li L, Ke Y (2007) Development and iron-dependent expression of hephaestin in different brain regions of rats. J Cell Biochem 102(5):1225–1233
Ke Y, Ho K, Du J, Zhu L, Xu Y, Wang Q, Wang CY, Li L, Ge X, Chang Y, Qian ZM (2006) Role of soluble ceruloplasmin in iron uptake by midbrain and hippocampus neurons. J Cell Biochem 98(4):912–919
Demeuse P, Kerkhofs A, Struys-Ponsar C, Knoops B, Remacle C, van den Bosch de Aguilar P (2002) Compartmentalized coculture of rat brain endothelial cells and astrocytes: a syngenic model to study the blood-brain barrier. J Neurosci Methods 121(1):21–31
Ge XH, Wang Q, Qian ZM, Zhu L, Du F, Yung WH, Yang L, Ke Y (2009) The iron regulatory hormone hepcidin reduces ferroportin 1 content and iron release in H9C2 cardiomyocytes. J Nutr Biochem 20:860–865
Ke Y, Chen Y, Chang YZ, Duan XL, Ho KP, Jiang DH, Wan K, Qian ZM (2003) Post-transcriptional expression of DMT1 in the heart of rat. J Cell Physiol 196:124–130
Chen Y, Qian ZM, Du J, Duan X, Chang Y, Wang Q, Wang C, Ma YM, Xu Y, Li L, Ke Y (2005) Iron loading inhibits ferroportin1 expression in PC12 cells. Neurochem Int 47(7):507–513
Chang YZ, Ke Y, Du J, Halpern GM, Ho KP, Zhu L, Gu XS, Xu YJ, Wang Q, Li L, Wang CY, Qian ZM (2006) Increased divalent metal transporter 1 expression might be associated with the neurotoxicity of L-DOPA. Mol Pharmacol 69(3):968–974
Qian ZM, Morgan EH (1990) Effect of lead on the transport of transferrin-free and transferrin-bound iron into rabbit reticulocytes. Biochem Pharmacol 40:1049–1054
Qian ZM, Tang PL, Morgan EH (1996) Effect of lipid peroxidation on transferrin-free iron uptake by rabbit reticulocytes. Biochim Biophys Acta 1310:293–302
Qian ZM, Tsoi YK, Ke Y, Wong MS (2001) Ceruloplasmin promotes iron uptake rather than release in BT325 cells. Exp Brain Res 140(3):369–374
Ke Y, Chang YZ, Duan XL, Du JR, Zhu L, Wang K, Yang X, Ho KP, Qian ZM (2005) Iron-independent and age-dependent expression of the two mRNA isoforms of divalent metal transporter 1 in different brain regions of rats. Neurobiol Aging 26:739–748
Whitnall M, Richardson DR (2006) Iron: a new target for pharmacological intervention in neurodegenerative diseases. Semin Pediatr Neurol 13:186–197
Chaston TB, Richardson DR (2003) Iron chelators for the treatment of iron overload disease: relationship between structure, redox activity, and toxicity. Am J Hematol 73:200–210
Kalinowski DS, Richardson DR (2005) The evolution of iron chelators for the treatment of iron overload disease and cancer. Pharmacol Rev 57:547–583
Qian ZM, Li H, Sun H, Ho KP (2002) Targeted drug delivery via the transferrin receptor-mediated endocytosis pathway. Pharmacol Rev 54:561–587
Qian ZM, Wang Q (1998) Expression of iron transport proteins and excessive iron accumulation in the brain in neurodegenerative disorders. Brain Res Rev 27:257–267
Bradbury MW (1997) Transport of iron in the blood-brain-cerebrospinal fluid system. J Neurochem 69:443–454
Li H, Qian ZM (2002) Transferrin/transferrin receptor-mediated drug delivery. Med Res Rev 22:225–250
Malecki EA, Cook BM, Devenyi AG, Beard JL, Connor JR (1999) Transferrin is required for normal distribution of 59Fe and 54Mn in mouse brain. J Neurol Sci 170:112–118
Johanson CE, Duncan JA 3rd, Klinge PM, Brinker T, Stopa EG, Silverberg GD (2008) Multiplicity of cerebrospinal fluid functions: new challenges in health and disease. Cerebrospinal Fluid Res 5:10
Acknowledgments
The studies in our laboratories were supported the General Grant of National Natural Science Foundation of China (NSFC) (31271132, 31371092), by the Competitive Earmarked Grants of The Hong Kong Research Grants Council (GRF 466713), National 973 Programs (2011CB510004), and Key Project Grant of NSFC (31330035).
Conflict of Interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Du, F., Qian, ZM., Luo, Q. et al. Hepcidin Suppresses Brain Iron Accumulation by Downregulating Iron Transport Proteins in Iron-Overloaded Rats. Mol Neurobiol 52, 101–114 (2015). https://doi.org/10.1007/s12035-014-8847-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-014-8847-x