Abstract
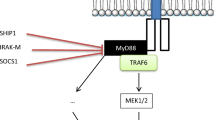
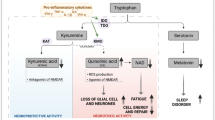
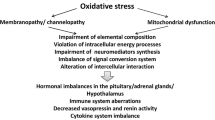
The World Health Organization classifies myalgic encephalomyelitis/chronic fatigue syndrome (ME/cfs) as a nervous system disease. Together with other diseases under the G93 heading, ME/cfs shares a triad of abnormalities involving elevated oxidative and nitrosative stress (O&NS), activation of immuno-inflammatory pathways, and mitochondrial dysfunctions with depleted levels of adenosine triphosphate (ATP) synthesis. There is also abundant evidence that many patients with ME/cfs (up to around 60 %) may suffer from autoimmune responses. A wide range of reported abnormalities in ME/cfs are highly pertinent to the generation of autoimmunity. Here we review the potential sources of autoimmunity which are observed in people with ME/cfs. The increased levels of pro-inflammatory cytokines, e.g., interleukin-1 and tumor necrosis factor-α, and increased levels of nuclear factor-κB predispose to an autoimmune environment. Many cytokine abnormalities conspire to produce a predominance of effector B cells and autoreactive T cells. The common observation of reduced natural killer cell function in ME/cfs is a source of disrupted homeostasis and prolonged effector T cell survival. B cells may be pathogenic by playing a role in autoimmunity independent of their ability to produce antibodies. The chronic or recurrent viral infections seen in many patients with ME/cfs can induce autoimmunity by mechanisms involving molecular mimicry and bystander activation. Increased bacterial translocation, as observed in ME/cfs, is known to induce chronic inflammation and autoimmunity. Low ATP production and mitochondrial dysfunction is a source of autoimmunity by inhibiting apoptosis and stimulating necrotic cell death. Self-epitopes may be damaged by exposure to prolonged O&NS, altering their immunogenic profile and become a target for the host’s immune system. Nitric oxide may induce many faces of autoimmunity stemming from elevated mitochondrial membrane hyperpolarization and blockade of the methionine cycle with subsequent hypomethylation of DNA. Here we also outline options for treatment involving rituximab and endotherapia.
Similar content being viewed by others
References
Morris G, Maes M (2012) A neuro‐immune model of myalgic encephalomyelitis/chronic fatigue syndrome. Metab Brain Dis. 0885‐7490
World Health Organisation (1992) ICD-10 classifications of mental and behavioural disorder: clinical descriptions and diagnostic guidelines. World Health Organisation, Geneva
Carruthers BM, van de Sande MI, De Meirleir KL, Klimas NG, Broderick G, Mitchell T, Staines D, Powles AC, Speight N, Vallings R, Bateman L, Baumgarten-Austrheim B, Bell DS, Carlo-Stella N, Chia J, Darragh A, Jo D, Lewis D, Light AR, Marshall-Gradisbik S, Mena I, Mikovits JA, Miwa K, Murovska M, Pall ML, Stevens S (2011) Myalgic encephalomyelitis: international consensus criteria. J Intern Med 270:327–338
Carruthers BM, Kumar Jain A, De Meirleir KL, Peterson DL, Klimas NG, Lerner AM, Bested AC, Flor-Henry P, Joshi P, Powles ACP, Sherkey JA, van de Sande MI (2003) Myalgic encephalomyelitis/chronic fatigue syndrome: clinical working case definition, diagnostic and treatment protocols. JCFS 11:7–97
Maes M, Kubera M, Uytterhoeven M, Vrydags N, Bosmans E (2011) Increased plasma peroxides as a marker of oxidative stress in myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS). Med Sci Monit 17:SC11–SC15
Morris G, Maes M (2012) Increased nuclear factor-κB and loss of p53 are key mechanisms in myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS). Med Hypotheses 79:607–613
Seishima M, Mizutani Y, Shibuya Y, Arakawa C (2008) Chronic fatigue syndrome after human parvovirus B19 infection without persistent viremia. Dermatology 216:341–346
Chia JK, Chia AY (2008) Chronic fatigue syndrome is associated with chronic enterovirus infection of the stomach. J Clin Pathol 61:43–48
Nicolson GL, Nicolson NL, Haier J (2008) Chronic fatigue syndrome patients subsequently diagnosed with Lyme disease Borrelia burgdorferi: evidence for mycoplasma species co-infections. JCFS 14:5–17
Nicolson GL, Gan R, Haier J (2003) Multiple co-infections (Mycoplasma, Chlamydia, human herpesvirus-6) in blood of chronic fatigue syndrome patients: association with signs and symptoms. APMIS 111:557–566
Sleigh KM, Marra FH, Stiver HG (2002) Influenza vaccination: is it appropriate in chronic fatigue syndrome? Am J Respir Med 1:3–9
Brenu EW, Staines DR, Baskurt OK, Ashton KJ, Ramos SB, Christy RM, Marshall-Gradisnik SM (2010) Immune and hemorheological changes in chronic fatigue syndrome. J Transl Med 8:1
Brenu EW, van Driel ML, Staines DR, Ashton KJ, Ramos SB, Keane J, Klimas NG, Marshall-Gradisnik SM (2011) Immunological abnormalities as potential biomarkers in chronic fatigue syndrome/myalgic encephalomyelitis. J Transl Med 9:81
Brenu EW, van Driel ML, Staines DR, Ashton KJ, Hardcastle SL, Keane J, Tajouri L, Peterson D, Ramos SB, Marshall-Gradisnik SM (2012) Longitudinal investigation of natural killer cells and cytokines in chronic fatigue syndrome/myalgic encephalomyelitis. J Transl Med 10:88
Klimas N, Salvato F, Morgan R, Fletcher MA (1990) Immunologic abnormalities in chronic fatigue syndrome. J Clin Microbiol 28:1403–1410
Hassan IS, Bannister BA, Akbar A, Weir W, Bofill M (1998) A study of the immunology of the chronic fatigue syndrome: correlation of immunologic markers to health dysfunction. Clin Immunol Immunopathol 87:60–67
Maes M, Mihaylova I, Leunis JC (2006) Chronic fatigue syndrome is accompanied by an IgM-related immune response directed against neopitopes formed by oxidative or nitrosative damage to lipids and proteins. Neuro Endocrinol Lett 27:615–621
Maes M, Mihaylova I, Leunis JC (2007) Increased serum IgM antibodies directed against phosphatidyl inositol (Pi) in chronic fatigue syndrome (CFS) and major depression: evidence that an IgM-mediated immune response against Pi is one factor underpinning the comorbidity between both CFS and depression. Neuro Endocrinol Lett 28:861–867
Shungu DC, Weiduschat N, Murrough JW, Mao X, Pillemer S, Dyke JP, Medow MS, Natelson BH, Stewart JM, Mathew SJ (2012) Increased ventricular lactate in chronic fatigue syndrome. III. Relationships to cortical glutathione and clinical symptoms implicate oxidative stress in disorder pathophysiology. NMR Biomed 25(9):1073–1087
Kennedy G, Spence VA, McLaren M, Hill A, Underwood C, Belch JJ (2005) Oxidative stress levels are raised in chronic fatigue syndrome and are associated with clinical symptoms. Free Radic Biol Med 39:584–589
Maes M, Mihaylova I, Kubera M, Uytterhoeven M, Vrydags N, Bosmans E (2009) Coenzyme Q10 deficiency in myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS) is related to fatigue, autonomic and neurocognitive symptoms and is another risk factor explaining the early mortality in ME/CFS due to cardiovascular disorder. Neuro Endocrinol Lett 30:470–476
Maes M, Twisk FN (2009) Why myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS) may kill you: disorders in the inflammatory and oxidative and nitrosative stress (IO&NS) pathways may explain cardiovascular disorders in ME/CFS. Neuro Endocrinol Lett 30:677–693
Maes M, Twisk FNM, Kubera M, Ringel K (2011) Evidence for inflammation and activation of cell-mediated immunity in myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS): increased interleukin-1, tumor necrosis factor-α, PMN-elastase, lysozyme and neopterin. J Affect Disord 136:933–939
Maes M, Mihaylova I, Kubera M, Bosmans E (2007) Not in the mind but in the cell: increased production of cyclo-oxygenase-2 and inducible NO synthase in chronic fatigue syndrome. Neuro Endocrinol Lett 28:463–469
Maes M, Mihaylova I, Bosmans E (2007) Not in the mind of neurasthenic lazybones but in the cell nucleus: patients with chronic fatigue syndrome have increased production of nuclear factor kappa beta. Neuro Endocrinol Lett 28:456–462
Meeus M, Mistiaen W, Lambrecht L, Nijs J (2011) Immunological similarities between cancer and chronic fatigue syndrome: the common link to fatigue? Anticancer Res 29:4717–4726
Maes M, Mihaylova I, Kubera M, Leunis JC, Twisk FN, Geffard M (2012) IgM-mediated autoimmune responses directed against anchorage epitopes are greater in myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS) than in major depression. Metab Brain Dis 27(4):415–423
Maes M, Twisk FN, Johnson C (2012) Myalgic encephalomyelitis (ME), chronic fatigue syndrome (CFS), and chronic fatigue (CF) are distinguished accurately: results of supervised learning techniques applied on clinical and inflammatory data. Psychiatry Res 200:754–760
Lombardi V, Hagen KS, Hunter KW, Diamond JW, Smith-Gagen J, Yang W, Mikovits JA (2011) Xenotropic murine leukemia virus-related virus-associated chronic fatigue syndrome reveals a distinct inflammatory signature. In Vivo 25:307–314
Maes M, Twisk FN, Kubera M, Ringel K, Leunis JC, Geffard M (2012) Increased IgA responses to the LPS of commensal bacteria is associated with inflammation and activation of cell-mediated immunity in chronic fatigue syndrome. J Affect Disord 136:909–917
Maes M, Mihaylova I, Leunis JC (2005) In chronic fatigue syndrome, the decreased levels of omega-3 poly-unsaturated fatty acids are related to lowered serum zinc and defects in T cell activation. Neuro Endocrinol Lett 26:745–751
Vermuelen RCW, Kurt RM, Visser FC, Sluiter W, Scholte HR (2010) Patients with chronic fatigue syndrome performed worse than controls in a controlled repeated exercise study despite a normal oxidative phosphorylation capacity. J Transl Med 8:93
Myhill S, Booth NE, McLaren-Howard J (2009) Chronic fatigue syndrome and mitochondrial dysfunction. Int J Clin Exp Med 2:1–16
Booth NE, Myhill S, McLaren-Howard J (2012) Mitochondrial dysfunction and the pathophysiology of myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS). Int J Clin Exp Med 5:208–220
Wong R, Lopaschuk G, Zhu G, Walker D, Catellier D, Burton D, Teo K, Collins-Nakai R, Montague T (1992) Skeletal muscle metabolism in the chronic fatigue syndrome. In vivo assessment by 31P nuclear magnetic resonance spectroscopy. Chest 102:1716–1722
Plioplys AV, Plioplys S (1995) Electron-microscopic Investigation of muscle mitochondria in chronic fatigue syndrome. Neuropsychobiol 32:175–181
Sharpe MC, Archard LC, Banatvala JE, Borysiewicz LK, Clare AW, David A, Edwards RH, Hawton KE, Lambert HP, Lane RJ, McDonald EM, Mowbray JF, Pearson DJ, Peto TEA, Preedy VR, Smith AP, Smith DG, Taylor DJ, Tyrrell AJ, Wessely S, White PD (1991) A report—chronic fatigue syndrome: guidelines for research. JR Soc Med 84:118–121
Cleare AJ, O’Keane V, Miell J (2001) Plasma leptin in chronic fatigue syndrome and a placebo-controlled study of the effects of low-dose hydrocortisone on leptin secretion. Clin Endocrinol (Oxf) 55:113–119
Lu XY (2007) The leptin hypothesis of depression: a potential link between mood disorders and obesity? Curr Opin Pharmacol 7:648–652
Pasco JA, Jacka FN, Williams LJ, Henry MJ, Nicholson GC, Kotowicz MA, Berk M (2008) Leptin in depressed women: cross-sectional and longitudinal data from an epidemiologic study. J Affect Disord 107(1–3):221–225
Nijs J, McGregor NR, De Becker P, Verhas M, Englebienne P, De Meirleir K (2003) Monitoring a hypothetical channelopathy in chronic fatigue syndrome: preliminary observations. JCFS 11:117–133
Nijs J, Coomans D, Nicolson GL, De Becker P, Christian D, De Meirleir K (2003) Immunophenotyping predictive of Mycoplasma infection in patients with chronic fatigue syndrome. JCFS 11:51–69
Tirelli U, Marotta G, Improta S, Pinto A (1994) Immunological abnormalities in patients with chronic fatigue syndrome. Scand J Immunol 40:601–608
Bradley AS, Ford B, Bansal AS (2013) Altered functional B-cell subset populations in patients with chronic fatigue syndrome compared to healthy controls. 172(1):73–80. doi:10.1111/cei.12043
Tanaka S, Kuratsune H, Hidaka Y, Hakariya Y, Tatsumi KI, Takano T, Kanakura Y, Amino N (2003) Autoantibodies against muscarinic cholinergic receptor in chronic fatigue syndrome. Int J Mol Med 12:225–230
Buchwald MD, Wener MH, Komaroff AL (1991) Antineuronal antibody levels in chronic fatigue syndrome patients with neurologic abnormalities. Arthritis Rheum 34:1485–1486
Plioplys AV (1997) Antimuscle and anti-CNS circulating antibodies in chronic fatigue syndrome. Neurology 48:1717–1719
Bassi N, Amital D, Amital H, Doria A, Shoenfeld Y (2008) Chronic fatigue syndrome: characteristics and possible causes for its pathogenesis. Isr Med Assoc J 10:79–82
Nishikai M (2007) Antinuclear antibodies in patients with chronic fatigue syndrome. Nippon Rinsho 65:1067–1070
Maes M (2010) An intriguing and hitherto unexplained co-occurrence: depression and chronic fatigue syndrome are manifestations of shared inflammatory, oxidative and nitrosative (IO&NS) pathways. Prog Neuropsychopharmacol. Biol Psychiatry 35:784–794
Klein R, Berg PA (1995) High incidence of antibodies to 5-hydroxytryptamine, gangliosides and phospholipids in patients with chronic fatigue and fibromyalgia syndrome and their relatives: evidence for a clinical entity of both disorders. Eur J Med Res 1:21–26
Ljunggren HG, Karre K (1990) In search of the ‘missing self’: MHC molecules and NK cell recognition. Immunol Today 11:237–244
Yokoyama WM, Kim S, French AR (2004) The dynamic life of natural killer cells. Annu Rev Immunol 22:405–429
Lunemann A, Lunemann JD, Munz C (2009) Regulatory NK-cell functions in inflammation and autoimmunity. Mol Med 15:352–358
Strowig T, Brilot F, Munz C (2008) Noncytotoxic functions of NK cells: direct pathogen restriction and assistance to adaptive immunity. J Immunol 180:7785–7791
Zimmer J, Bausinger H, de la Salle H (2001) Autoimmunity mediated by innate immune effector cells. Trends Immunol 22:300–301
Shi F, Ljunggren HG, Sarvetnick N (2001) Innate immunity and autoimmunity: from self-protection to self-destruction. Trends Immunol 22:97–101
Smeltz RB, Wolf NA, Swanborg RH (1999) Inhibition of autoimmune T cell responses in the DA rat by bone marrow-derived NK cells in vitro: implications for autoimmunity. J Immunol 163:1390–1397
Matsumoto Y, Kohyama K, Aikawa Y, Shin T, Kawazoe Y, Suzuki Y, Tanuma N (1998) Role of natural killer cells and TCRγδ T cells in acute autoimmune encephalomyelitis. Eur J Immunol 28:1681–1688
Shi FD, Takeda K, Akira S, Sarvetnick N, Ljunggren HG (2000) IL-18 directs autoreactive T cells and promotes autodestruction in the central nervous system via induction of IFN-γ by NK cells. J Immunol 165:3099–3104
Shi FD, Wang HB, Li H, Hong S, Taniguchi M, Link H, Van Kaer L, Ljunggren HG (2000) Natural killer cells determine the outcome of B cell-mediated autoimmunity. Nat Immunol 1:245–251
Trinchieri G (1989) Biology of natural killer cells. Adv Immunol 47:187–376
Liu R, Van Kaer L, La Cava A, Price M, Campagnolo DI, Collins M, Young DA, Vollmer TL, Shi FD (2006) Autoreactive T cells mediate NK cell degeneration in autoimmune disease. J Immunol 176:5247–5254
Kasaian MT, Whitters MJ, Carter LL, Lowe LD, Jussif JM, Deng B, Johnson KA, Witek JS, Senices M, Konz RF, Wurster AL, Donaldson DD, Collins M, Young DA, Grusby MJ (2002) IL-21 limits NK cell responses and promotes antigen-specific T cell activation: a mediator of the transition from innate to adaptive immunity. Immunity 16:559–569
Vollmer TL, Liu R, Price M, Rhodes S, La Cava A, Shi FD (2005) Differential effects of IL-21 during initiation and progression of autoimmunity against neuroantigen. J Immunol 174:2696–2701
Fehniger TA, Cooper MA, Nuovo GJ, Cella M, Facchetti F, Colonna M, Caligiuri MA (2003) CD56bright natural killer cells are present in human lymph nodes and are activated by T cell-derived IL-2: a potential new link between adaptive and innate immunity. Blood 101:3052–3057
Ferlazzo G, Pack M, Thomas D, Paludan C, Schmid D, Strowig T, Bougras G, Muller WA, Moretta L, Munz C (2004) Distinct roles of IL-12 and IL-15 in human natural killer cell activation by dendritic cells from secondary lymphoid organs. Proc Natl Acad Sci U S A 101:16606–16611
Zingoni A, Sornasse T, Cocks BG, Tanaka Y, Santoni A, Lanier LL (2004) Cross-talk between activated human NK cells and CD4+ T cells via OX40–OX40 ligand interactions. J Immunol 173:3716–3724
Vranes Z, Poljakovic Z, Marusic M (1989) Natural killer cell number and activity in multiple sclerosis. J Neurol Sci 94:115–123
Infante-Duarte C, Weber A, Kratzschmar J, Prozorovski T, Pikol S, Hamann I, Bellmann-Strobl J, Aktas O, Dorr J, Wuerfel J, Sturzebecher CS, Zipp F (2005) Frequency of blood CX3CR1-positive natural killer cells correlates with disease activity in multiple sclerosis patients. FASEB J 19:1902–1904
Schippling DS, Martin R (2008) Spotlight on anti-CD25: daclizumab in MS. Int MS J 15:94–98
Bielekova B, Catalfamo M, Reichert-Scrivner S, Packer A, Cerna M, Waldmann TA, McFarland H, Henkart PA, Martin R (2006) Regulatory CD56z(bright) natural killer cells mediate immunomodulatory effects of IL-2Ralpha-targeted therapy (daclizumab) in multiple sclerosis. Proc Natl Acad Sci U S A 103:5941–5946
Biron CA, Nguyen KB, Pien GC, Cousens LP, Salazar-Mather TP (1999) Natural killer cells in antiviral defense: function and regulation by innate cytokines. Annu Rev Immunol 17:189–220
Su HC, Nguyen KB, Salazar-Mather TP, Ruzek MC, Dalod MY, Biron CA (2001) NK cell functions restrain T cell responses during viral infections. Eur J Immunol 31:3048–3055
Fujinami RS, Oldstone MB, Wroblewska Z, Frankel ME, Koprowski H (1983) Molecular mimicry in virus infection: crossreaction of measles virus phosphoprotein or of herpes simplex virus protein with human intermediate filaments. Proc Natl Acad Sci U S A 80:2346–2350
Li Y, Peng T, Yang Y, Niu C, Archard LC, Zhang H (2000) High prevalence of enteroviral genomic sequences in myocardium from cases of endemic cardiomyopathy (Keshan disease) in China. Heart 83:696–701
Fujinami RS, von Herrath MG, Christen U, Whitton JL (2006) Molecular mimicry, bystander activation, or viral persistence: infections and autoimmune disease. Clin Microbiol Rev 19:80–94
Andersen O, Lygner PE, Bergstrom T, Andersson M, Vahlne A (1993) Viral infections trigger multiple sclerosis relapses: a prospective seroepidemiological study. J Neurol 240:417–422
Edwards S, Zvartau M, Clarke H, Irving W, Blumhardt LD (1998) Clinical relapses and disease activity on magnetic resonance imaging associated with viral upper respiratory tract infections in multiple sclerosis. J Neurol Neurosurg Psychiatry 64:736–741
Johnson RT (1998) Chronic inflammatory and demyelinating diseases. In: Viral infections of the nervous system. 2nd Ed. Lippincott-Raven, Philadelphia, pp. 227–263
Wucherpfennig KW, Ota K, Endo N, Seidman JG, Rosenzweig A, Weiner HL, Hafler DA (1990) Shared human T cell receptor Vb usage to immunodominant regions of myelin basic protein. Science 248:1016–1019
McCoy L, Tsunoda I, Fujinami RS (2006) Multiple sclerosis and virus induced immune responses: autoimmunity can be primed by molecular mimicry and augmented by bystander activation. Autoimmunity 39:9–19
Bangs SC, McMichael AJ, Xu XN (2006) Bystander T cell activation—implications for HIV infection and other diseases. Trends Immunol 27:518–524
Bierer BE, Hahn WC (1993) T cell adhesion, avidity regulation and signaling: a molecular analysis of CD2. Semin Immunol 5:249–261
Unutmaz D, Pilri P, Abrignani S (1994) Antigen-independent activation of naive and memory resting T cells by a cytokine combination. J Exp Med 180:1159–1164
Mathew A, Kurane I, Green S, Vaughn DW, Kalayanarooj S, Suntayakorn S, Ennis FA, Rothman AL (1999) Impaired T cell proliferation in acute dengue infection. J Immunol 162:5609–5615
Zivna I, Green S, Vaughn DW, Kalayanarooj S, Stephens HA, Chandanayingyong D, Nisalak A, Ennis FA, Rothman AL (2002) T cell responses to an HLA-B*07-restricted epitope on the dengue NS3 protein correlate with disease severity. J Immunol 168:5959–5965
Rowland CA, Lertmemongkolchai G, Bancroft A, Haque A, Lever MS, Griffin KF, Jackson MC, Nelson M, O’Garra A, Grencis R, Bancroft GJ, Lukaszewski RA (2006) Critical role of type 1 cytokines in controlling initial infection with Burkholderia mallei. Infect Immun 74:5333–5340
Douek DC, Picker LJ, Koup RA (2003) T cell dynamics in HIV-1 infection. Annu Rev Immunol 21:265–304
Silvestri G, Feinberg MB (2003) Turnover of lymphocytes and conceptual paradigms in HIV infection. J Clin Invest 112:821–824
Fadok VA, Henson PM (1998) Apoptosis: getting rid of the bodies. Curr Biol 8:R693–R695
Chung EY, Liu J, Homma Y, Zhang Y, Brendolan A, Saggese M, Han J, Silverstein R, Selleri L, Ma X (2007) Interleukin-10 expression in macrophages during phagocytosis of apoptotic cells is mediated by homeodomain proteins Pbx1 and Prep-1. Immunity 27:952–964
Cvetanovic M, Ucker DS (2004) Innate immune discrimination of apoptotic cells: repression of proinflammatory macrophage transcription is coupled directly to specific recognition. J Immunol 172:880–889
Hanayama R, Tanaka M, Miyasaka K, Aozasa K, Koike M, Uchiyama Y, Nagata S (2004) Autoimmune disease and impaired uptake of apoptotic cells in MFG-E8-deficient mice. Science 304:1147–1150
Erwig LP, Henson PM (2007) Immunological consequences of apoptotic cell phagocytosis. Am J Pathol 171:2–8
Nagata S, Hanayama R, Kawane K (2010) Autoimmunity and the clearance of dead cells. Cell 140:619–630
Gaipl US, Franz S, Voll RE, Sheriff A, Kalden JR, Herrmann M (2004) Defects in the disposal of dying cells lead to autoimmunity. Curr Rheumatol Rep 6:401–407
Arends MJ, Wyllie AH (1991) Apoptosis: mechanisms and roles in pathology. Int. Rev. Exp Pathol 32:223–254
Scaffidi C, Fulda S, Srinivasan A, Friesen C, Li F, Tomaselli KJ, Debatin KM, Krammer PH, Peter ME (1998) Two CD95 (APO-1/Fas) signaling pathways. EMBO J 17:1675–1687
Medema JP, Scaffidi C, Kischkel FC, Shevchenko A, Mann M, Krammer PH, Peter ME (1997) FLICE is activated by association with the CD95 death-inducing signaling complex (DISC). EMBO J 16:2794–2804
Salvesen GS, Dixit VM (1997) Caspases: intracellular signaling by proteolysis. Cell 91:443–446
Goonetilleke UR, Ward SA, Gordon SB (2009) Could proteomic research deliver the next generation of treatments for pneumococcal meningitis? Interdiscip Perspect Infect Dis 2009:214–216
Thornberry NA, Lazebnik Y (1998) Caspases: enemies within. Science 281:1312–1316
Li P, Nijhawan D, Budihardjo I, Srinivasula SM, Ahmad M, Alnemri ES, Wang X (1997) Cytochrome c and dATP-dependent formation of Apaf-1/caspase-9 complex initiates an apoptotic protease cascade. Cell 91:479–489
Eguchi Y, Shimizu S, Tsujimoto Y (1997) Intracellular ATP levels determine cell death fate by apoptosis or necrosis. Cancer Res 57:1835–1840
Melchior F, Gerace L (1995) Mechanisms of nuclear protein import. Curr Opin Cell Biol 7:310–318
Yasuhara N, Eguchi Y, Tachibana T, Imamoto N, Yoneda Y, Tsujimoto Y (1997) Essential role of active nuclear transport in apoptosis. Genes Cells 2:55–64
Zong WX, Thompson CB (2006) Necrotic death as a cell fate. Genes Dev 20:1–15
Proskuryakov SY, Konoplyannikov AG, Gabai VL (2003) Necrosis: a specific form of programmed cell death? Exp Cell Res 283:1–16
Xu Y, Huang S, Liu ZG, Han J (2006) Poly(ADP-ribose) polymerase-1 signalling to mitochondria in necrotic cell death requires RIP1/TRAF2-mediated JNK1 activation. J Biol Chem 281:8788–8795
Chen Y (2009) Necrosis: an energy-dependent programmed cell death? UTMJ 86:110–112
Blackwell TS, Christman JW (1997) The role of nuclear factor-kappa B in cytokine gene regulation. Am J Respir Cell Mol Biol 17:3–9
Zhang G, Ghosh S (2000) Molecular mechanisms of NF-kappaB activation induced by bacterial lipopolysaccharide through Toll-like receptors. J Endotoxin Res 6:453–457
Siebenlist U, Franzoso G, Brown K (1994) Structure, regulation and function of NF-kappa B. Annu Rev Cell Biol 10:405–455
Gloire G, Legrand-Poels S, Piette J (2006) NF-kappaB activation by reactive oxygen species: fifteen years later. Biochem Pharmacol 72:1493–1505
Jones E, Adcock IM, Ahmed BY, Punchard NA (2007) Modulation of LPS stimulated NF-kappaB mediated nitric oxide production by PKCepsilon and JAK2 in RAW macrophages. J Inflamm (Lond) 4:23
Enzler T, Bonizzi G, Silverman GJ, Otero DC, Widhopf GF, Anzelon-Mills A, Rickert RC, Karin M (2006) Alternative and classical NF-kappa B signaling retain autoreactive B cells in the splenic marginal zone and result in lupus-like disease. Immunity 25:403–415
Wu Y, Bressette D, Carrell JA, Kaufman T, Feng P, Taylor K, Gan Y, Cho YH, Garcia AD, Gollatz E, Dimke D, LaFleur D, Migone TS, Nardelli B, Wei P, Ruben SM, Ullrich SJ, Olsen HS, Kanakaraj P, Moore PA, Baker KP (2000) Tumor necrosis factor (TNF) receptor superfamily member TACI is a high affinity receptor for TNF family members APRIL and BLyS. J Biol Chem 275:35478–35485
Rahman ZS, Rao SP, Kalled SL, Manser T (2003) Normal induction but attenuated progression of germinal center responses in BAFF and BAFF-R signaling-deficient mice. J Exp Med 198:1157–1169
Liu Z, Davidson A (2011) BAFF and selection of autoreactive B cells. Trends Immunol 32:388–394
Senftleben U, Karin M (2002) The IKK/NF-kappa B pathway. Crit Care Med 30:S18–S26
Li ZW, Chu W, Hu Y, Delhase M, Deerinck T, Ellisman M, Johnson R, Karin M (1999) The IKKbeta subunit of IkappaB kinase (IKK) is essential for nuclear factor kappaB activation and prevention of apoptosis. J Exp Med 189:1839–1845
Karin M, Lin A (2002) NF-kappaB at the crossroads of life and death. Nat Immunol 3:221–227
Karin M, Delhase M (2000) The I kappa B kinase (IKK) and NF-kappa B: key elements of proinflammatory signalling. Semin Immunol 12:85–98
Senftleben U, Cao Y, Xiao G, Greten FR, Krahn G, Bonizzi G, Chen Y, Hu Y, Fong A, Sun SC, Karin M (2001) Activation by IKKalpha of a second, evolutionary conserved, NF-kappa B signaling pathway. Science 293:1495–1499
Bonizzi G, Karin M (2004) The two NF-kappaB activation pathways and their role in innate and adaptive immunity. Trends Immunol 25:280–288
Luo JL, Kamata H, Karin M (2005) IKK/NF-kappaB signaling: balancing life and death—a new approach to cancer therapy. J Clin Invest 115:2625–2632
Danial NN, Korsmeyer SJ (2004) Cell death: critical control points. Cell 116:205–219
Hengartner MO (2000) The biochemistry of apoptosis. Nature 407:770–776
Beg AA, Sha WC, Bronson RT, Ghosh S, Baltimore D (1995) Embryonic lethality and liver degeneration in mice lacking the RelA component of NF-kappa B. Nature 376:167–170
Doi TS, Marino MW, Takahashi T, Yoshida T, Sakakura T, Old LJ, Obata Y (1999) Absence of tumor necrosis factor rescues RelA-deficient mice from embryonic lethality. Proc Natl Acad Sci U S A 96:2994–2999
Kucharczak J, Simmons MJ, Fan Y, Gelinas C (2003) To be, or not to be: NF-kappaB is the answer—role of Rel/NF-kappaB in the regulation of apoptosis. Oncogene 22:8961–8982
Zhang Y, Chen F (2004) Reactive oxygen species (ROS), troublemakers between nuclear factor-kappaB (NF-kappaB) and c-Jun NH(2)-terminal kinase (JNK). Cancer Res 64:1902–1905
Wullaert A, Heyninck K, Beyaert R (2006) Mechanisms of crosstalk between TNF-induced NF-kappaB and JNK activation in hepatocytes. Biochem Pharmacol 72:1090–1101
Sakon S, Xue X, Takekawa M, Sasazuki T, Okazaki T, Kojima Y, Piao JH, Yagita H, Okumura K, Doi T, Nakano H (2003) NF-kappaB inhibits TNF-induced accumulation of ROS that mediate prolonged MAPK activation and necrotic cell death. EMBO J 22:3898–3909
Gonzales-Garcia C, Martin-Saavedra FM, Ballestar A, Ballestar S (2009) The Th17 lineage: answers to some immunological questions. Inmunologia 28:32–45
Waite JC, Skokos D (2012) Th17 response and inflammatory autoimmune diseases. Int J Inflamm 2012:819467
Zhou L, Ivanov II, Spolski R, Min R, Shenderov K, Egawa T, Levy DE, Leonard WJ, Littman DR (2007) IL-6 programs T(H)-17 cell differentiation by promoting sequential engagement of the IL-21 and IL-23 pathways. Nat Immunol 8:967–974
Finckh A, Gabay C (2008) At the horizon of innovative therapy in rheumatology: new biologic agents. Curr Opin Rheumatol 20:269–275
Jadidi-Niaragh F, Mirshafiey A (2011) Th17 cell, the new player of neuroinflammatory process in multiple sclerosis. Scand J Immunol 74:1–13
Kryczek I, Wei S, Vatan L, Escara-Wilke J, Szeliga W, Keller ET, Zou W (2007) Cutting edge: opposite effects of IL-1 and IL-2 on the regulation of IL-17+ T cell pool IL-1 subverts IL-2-mediated suppression. J Immunol 179:1423–1426
Joosten LAB (2010) Excessive interleukin-1 signaling determines the development of Th1 and Th17 responses in chronic inflammation. Arthr Rheumatism 62:320–322
Deknuydt F, Bioley G, Valmon D, Ayyoub M (2009) IL-1β and IL-2 convert human Treg into TH17 cells. Clin Immunol 131:298–307
Ziegler SF, Buckner JH (2009) FOXP3 and the regulation of Treg/Th17 differentiation. Microbes Infect 11:594–598
Kebir H, Kreymborg K, Ifergan I, Dodelet-Devillers A, Cayrol R, Bernard M, Giuliani F, Arbour N, Becher B, Prat A (2007) Human TH17 lymphocytes promote blood–brain barrier disruption and central nervous system inflammation. Nat Med 13:1173–1175
McColl BW, Rothwell NJ, Allan SM (2008) Systemic inflammation alters the kinetics of cerebrovascular tight junction disruption after experimental stroke in mice. J Neurosci 28:9451–9462
Sharief MK, Ciardi M, Thompson EJ, Sorice F, Rossi F, Vullo V, Cirelli A (1992) Tumour necrosis factor-alpha mediates blood–brain barrier damage in HIV-1 infection of the central nervous system. Mediators Inflamm 1:191–196
Ma TY, Iwamoto GK, Hoa NT, Akotia V, Pedram A, Boivin MA, Said HM (2004) TNF-alpha-induced increase in intestinal epithelial tight junction permeability requires NF-kappa B activation. Am J Physiol Gastrointest Liver Physiol 286:G367–G376
Al-Sadi RM, Ma TY (2007) IL-1beta causes an increase in intestinal epithelial tight junction permeability. J Immunol 178:4641–4649
Lakhan SE, Kirchgessner A (2010) Gut inflammation in chronic fatigue syndrome. Nutr Metab (Lond) 7:79
Wang H, Sun J, Goldstein H (2008) Human immunodeficiency virus type 1 infection increases the in vivo capacity of peripheral monocytes to cross the blood–brain barrier into the brain and the in vivo sensitivity of the blood–brain barrier to disruption by lipopolysaccharide. J Virol 82:7591–7600
Xing B, Bachstetter AD, Eldik LJ (2011) Microglial p38α MAPK is critical for LPS-induced neuron degeneration, through a mechanism involving TNFα. Mol Neurodegener 6:84
Chauhan VS, Marriott I (2007) Bacterial infections of the central nervous system: a critical role for resident glial cells in the initiation and progression of inflammation. Curr Immunol 3:133–143
Rock RB, Gekker G, Hu S, Sheng WS, Cheeran M, Lokensgard JR, Peterson PK (2004) Role of microglia in central nervous system infections. Clin Microbiol Rev 17:942–964
Yu RK, Usuki S, Ariga T (2006) Ganglioside molecular mimicry and its pathological roles in Guillain–Barré syndrome and related diseases. Infect Immun 74:6517–6527
Ang CW, Noordzij PG, de Klerk MA, Endtz HP, van Doorn PA, Laman JD (2002) Ganglioside mimicry of Campylobacter jejuni lipopolysaccharides determines antiganglioside specificity in rabbits. Infect Immun 70:5081–5085
Wang YS, White TD (1999) The bacterial endotoxin lipopolysaccharide causes rapid inappropriate excitation in rat cortex. J Neurochem 72:652–660
Sherbet G (2009) Bacterial infections and the pathogenesis of autoimmune conditions. BJMP 2:6–13
Tufekci KU, Genc S, Genc K (2011) The endotoxin-induced neuroinflammation model of Parkinson’s disease. Parkinsons Dis 2011:487450
Yacyshyn B, Meddings J, Sadowski D, Bowen-Yacyshyn MB (1996) Multiple sclerosis patients have peripheral blood CD45RO + B cells and increased intestinal permeability. Dig Dis Sci 41:2493–2498
Forsyth CB, Shannon KM, Kordower JH, Voigt RM, Shaikh M, Jaglin JA, Estes JD, Dodiya HB, Keshavarzian A (2011) Increased intestinal permeability correlates with sigmoid mucosa alpha-synuclein staining and endotoxin exposure markers in early Parkinson’s disease. PLoS One 6:e28032
Grune T, Michel P, Sitte N, Eggert W, Albrecht-Nebe H, Esterbauer H, Siems WG (1997) Increased levels of 4-hydroxynonenal modified proteins in plasma of children with autoimmune diseases. Free Radic Biol Med 23:357–360
Kurien BT, Scofield RH (2008) Autoimmunity and oxidatively modified autoantigens. Autoimmun Rev 7:567–573
Khan MF, Wu X, Kaphalia BS, Boor PJ, Ansari GA (1997) Acute hematopoietic toxicity of aniline in rats. Toxicol Lett 92:31–37
Weinberg JB, Granger DL, Pisetsky DS, Seldin MF, Misukonis MA, Mason SN, Pippen AM, Ruiz P, Wood ER, Gilkeson GS (1994) The role of nitric oxide in the pathogenesis of spontaneous murine autoimmune disease: increased nitric oxide production and nitric oxide synthase expression in MRL-lpr/lpr mice, and reduction of spontaneous glomerulonephritis and arthritis by orally administered NG-monomethyl-l-arginine. J Exp Med 179:651–660
Khan F, Siddiqui AA, Ali R (2006) Measurement and significance of 3-nitrotyrosine in systemic lupus erythematosus. Scand J Immunol 64:507–514
Khan F, Ali R (2006) Antibodies against nitric oxide damaged poly l-tyrosine and 3-nitrotyrosine levels in systemic lupus erythematosus. J Biochem Mol Biol 39:189–196
Kurien BT, Hensley K, Bachmann M, Scofield RH (2006) Oxidatively modified autoantigens in autoimmune diseases. Free Radic Biol Med 41:549–556
Christensen B, Refsum H, Garras A, Ueland PM (1992) Homocysteine remethylation during nitrous oxide exposure of cells cultured in media containing various concentrations of folates. J Pharmacol Exp Ther 261:1096–1105
Castro R, Rivera I, Struys EA, Jansen EE, Ravasco P, Camilo ME, Blom HJ, Jakobs C, Tavares de Almeida I (2003) Increased homocysteine and S-adenosylhomocysteine concentrations and DNA hypomethylation in vascular disease. Clin Chem 49:1292–1296
Li Y, Liu Y, Strickland FM, Richardson B (2010) Age-dependent decreases in DNA methyltransferase levels and low transmethylation micronutrient levels synergize to promote overexpression of genes implicated in autoimmunity and acute coronary syndromes. Exp Gerontol 45:312–322
Zhao M, Tang J, Gao F, Wu X, Liang Y, Yin H, Lu Q (2010) Hypomethylation of IL10 and IL13 promoters in CD4+ T cells of patients with systemic lupus erythematosus. J Biomed Biotechnol 2010:931018
Xu X, Yang P, Shu Z, Bai Y, Wang CY (2012) DNA methylation in the pathogenesis of autoimmunity. In: Gu W, Wang Y (eds) Gene discovery for disease models. Wiley, Hoboken
Karouzakis E, Gay RE, Michel BA, Gay S, Neidhart M (2009) DNA hypomethylation in rheumatoid arthritis synovial fibroblasts. Arthritis Rheum 60:3613–3622
Kim YI, Logan JW, Mason JB, Roubenoff R (1996) DNA hypomethylation in inflammatory arthritis: reversal with methotrexate. J Lab Clin Med 128:165–172
Richardson B, Scheinbart L, Strahler J, Gross L, Hanash S, Johnson M (1990) Evidence for impaired T cell DNA methylation in systemic lupus erythematosus and rheumatoid arthritis. Arthritis Rheum 33:1665–1673
Richardson B (1986) Effect of an inhibitor of DNA methylation on T cells. II. 5-Azacytidine induces self-reactivity in antigen-specific T4+ cells. Hum Immunol 17:456–470
Beltran B, Quintero M, Garcia-Zaragoza E, O’Connor E, Esplugues JV, Moncada S (2002) Inhibition of mitochondrial respiration by endogenous nitric oxide: a critical step in Fas signaling. Proc Natl Acad Sci U S A 99:8892–8897
Perl A, Telarico T, Singh R, Fernandez D (2010) Activation of the nitric oxide–mitochondrial hyperpolarization–mTOR–Rab4 signaling pathway precedes disease development in lupus T cells. J Immunol 184:93.36
Nagy G, Koncz A, Telarico T, Fernandez D, Ersek B, Buzas E, Perl A (2010) Central role of nitric oxide in the pathogenesis of rheumatoid arthritis and systemic lupus erythematosus. Arthritis Res Ther 12:210
Hay N, Sonenberg N (2004) Upstream and downstream of mTOR. Genes Dev 18:1926–1945
Pierdominici M, Vacirca D, Delunardo F, Ortona E (2011) mTOR signaling and metabolic regulation of T cells: new potential therapeutic targets in autoimmune diseases. Curr Pharm Des 17:3888–3897
Fernandez-Riejos P, Najib S, Santos-Alvarez J, Martin-Romero C, Perez-Perez A, Gonzalez-Yanes C, Sanchez-Margalet V (2010) Role of leptin in the activation of immune cells. Mediators Inflamm 2010:568343
Delgoffe GM, Powell JD (2009) mTOR: taking cues from the immune microenvironment. Immunology 127:459–465
Yurchenko E, Shio MT, Huang TC, Da Silva Martins M, Szyf M, Levings MK, Oliver M, Piccirillo CA (2012) Inflammation-driven reprogramming of CD4+ Foxp3+ regulatory T cells into pathogenic Th1/Th17 T effectors is abrogated by mTOR inhibition in vivo. PLoS ONE 7:e35572
Haxhinasto S, Mathis D, Benoist C (2008) The AKT-mTOR axis regulates de novo differentiation of CD4 + Foxp3+ cells. J Exp Med 205:565–574
Fernandez D, Perl A (2010) mTOR signaling: a central pathway to pathogenesis in systemic lupus erythematosus? Discov Med 9:173–178
Procaccini C, De Rosa V, Galgani M, Carbone F, Cassano S, Greco D, Qian K, Auvinen P, Calì G, Stallone G, Formisano L, La Cava A, Matarese G (2012) Leptin-induced mTOR activation defines a specific molecular and transcriptional signature controlling CD4+ effector T cell responses. J Immunol 189:2941–2953
Matarese G, Leiter EH, La Cava A (2007) Leptin in autoimmunity: many questions, some answers. Tissue Antigens 70:87–95
Faggioni R, Feingold KR, Grunfeld C (2001) Leptin regulation of the immune response and the immunodeficiency of malnutrition. FASEB J 15:2565–2571
Bettowski J, Wojcicka G, Jamroz A (2003) Stimulatory effect of leptin on nitric oxide production is impaired in dietary-induced obesity. Obes Res 11:1571–1580
Loffreda S, Yang SQ, Lin HZ, Karp CL, Brengman ML, Wang DJ, Klein AS, Bulkley GB, Bao C, Noble PW, Lane MD, Diehl AM (1998) Leptin regulates proinflammatory immune responses. FASEB J 12:57–65
Napoleone E, DI Santo A, Amore C, Baccante G, di Febbo C, Porreca E, de Gaetano G, Donati MB, Lorenzet R (2007) Leptin induces tissue factor expression in human peripheral blood mononuclear cells: a possible link between obesity and cardiovascular risk? J Thromb Haemost 5:1462–1468
Martin-Romero C, Santos-Alvarez J, Goberna R, Sanchez-Margalet V (2000) Human leptin enhances activation and proliferation of human circulating T lymphocytes. Cell Immunol 199:15–24
Fernandez-Riejos P, Goberna R, Sanchez-Margalet V (2008) Leptin promotes cell survival and activates Jurkat T lymphocytes by stimulation of mitogen-activated protein kinase. Clin Exp Immunol 151:505–518
Sanchez-Margalet V, Martin-Romero C, Gonzalez-Yanes C, Goberna R, Rodriguez-Bano J, Muniain MA (2002) Leptin receptor (Ob-R) expression is induced in peripheral blood mononuclear cells by in vitro activation and in vivo in HIV-infected patients. Clin Exp Immunol 129:119–124
Zarkesh-Esfahani H, Pockley G, Metcalfe RA, Bidlingmaier M, Wu Z, Ajami A, Weetman AP, Strasburger CJ, Ross RJ (2001) High-dose leptin activates human leukocytes via receptor expression on monocytes. J Immunol 167:4593–4599
Kiguchi N, Maeda T, Kobayashi Y, Fukazawa Y, Kishioka S (2009) Leptin enhances CC-chemokine ligand expression in cultured murine macrophage. Biochem Biophys Res Commun 384:311–315
Wong CK, Cheung PF, Lam CW (2007) Leptin-mediated cytokine release and migration of eosinophils: implications for immunopathophysiology of allergic inflammation. Eur J Immunol 37:2337–2348
Mattioli B, Giordani L, Quaranta MG, Viora M (2009) Leptin exerts an anti-apoptotic effect on human dendritic cells via the PI3K–Akt signaling pathway. FEBS Lett 583:1102–1106
Lund FE (2008) Cytokine-producing B lymphocytes—key regulators of immunity. Curr Opin Immunol 20:332–338
Pistoia V (1997) Production of cytokines by human B cells in health and disease. Immunol Today 18:343–350
Mizoguchi A, Bhan AK (2006) A case for regulatory B cells. J Immunol 176:705–710
Lund FE, Randall TD (2010) Effector and regulatory B cells: modulators of CD4(+) T cell immunity. Nat Rev Immunol 10:236–247
de Goer de Herve MG, Durali D, Dembele B, Giuliani M, Tran TA, Azzarone B, Eid P, Tardieu M, Delfraissy JF, Taoufik Y (2011) Interferon-alpha triggers B cell effector 1 (Be1) commitment. PLoS One 6:e19366
Sanz I, Anolik JH, Looney RJ (2007) B cell depletion therapy in autoimmune diseases. Front Biosci 12:2546–2567
Stasi R (2010) Rituximab in autoimmune hematologic diseases: not just a matter of B cells. Semin Hematol 47:170–179
Harris DP, Goodrich S, Gerth AJ, Peng SL, Lund FE (2005) Regulation of IFN-gamma production by B effector 1 cells: essential roles for T-bet and the IFN-gamma receptor. J Immunol 174:6781–6790
Harris DP, Goodrich S, Mohrs K, Mohrs M, Lund FE (2005) Cutting edge: the development of IL-4-producing B cells (B effector 2 cells) is controlled by IL-4, IL-4 receptor alpha, and Th2 cells. J Immunol 175:7103–7107
Harris DP, Haynes L, Sayles PC, Duso DK, Eaton SM, Lepak NM, Johnson LL, Swain SL, Lund FE (2000) Reciprocal regulation of polarized cytokine production by effector B and T cells. Nat Immunol 1:475–482
Duddy ME, Alter A, Bar-Or A (2004) Distinct profiles of human B cell effector cytokines: a role in immune regulation? J Immunol 172:3422–3427
Blair PA, Norena LY, Flores-Borja F, Rawlings DJ, Isenberg DA, Ehrenstein MR, Mauri C (2010) CD19+CD24hiCD38hi B cells exhibit regulatory capacity in healthy individuals but are functionally impaired in systemic lupus erythematosus patients. Immunity 32:129–140
Pers JO, Lemoine S, Morva A, Saraux A, Jamin C, Youinou P (2011) Impaired regulatory capacities of B lymphocytes in systemic lupus erytematosus. Ann Rheum Dis 70:A59
Gros MJ, Naquet P, Guinamard RR (2008) Intrinsic TGF-beta 1 regulation of B cells. J Immunol 180:8153–8158
Yanaba K, Bouaziz JD, Haas KM, Poe JC, Fujimoto M, Tedder TF (2008) A regulatory B cell subset with a unique CD1dhiCD5+ phenotype controls T cell-dependent inflammatory responses. Immunity 28:639–650
Mauri C, Ehrenstein MR (2008) The ‘short’ history of regulatory B cells. Trends Immunol 29:34–40
Fluge O, Bruland O, Risa K, Storstein A, Kristoffersen EK, Sapkota D, Næss H, Dahl O, Nyland H, Mella O (2011) Benefit from B-lymphocyte depletion using the anti-CD20 antibody rituximab in chronic fatigue syndrome. A double-blind and placebo-controlled study. PLoS ONE 6:e26358
Eisenberg R (2005) Update on rituximab. Ann Rheum Dis 64:iv55–iv57
Gurcan HM, Keskin DB, Stern JN, Nitzberg MA, Shekhani H, Ahmed AR (2009) A review of the current use of rituximab in autoimmune diseases. Int Immunopharmacol 9:10–25
Edwards JC, Szczepanski L, Szechinski J, Filipowicz-Sosnowska A, Emery P, Close DR, Stevens RM, Shaw T (2004) Efficacy of B-cell-targeted therapy with rituximab in patients with rheumatoid arthritis. N Engl J Med 350:2572–2581
Tokunaga M, Saito K, Kawabata D, Imura Y, Fujii T, Nakayamada S, Tsujimura S, Nawata M, Iwata S, Azuma T, Mimori T, Tanaka Y (2007) Efficacy of rituximab (anti-CD20) for refractory systemic lupus erythematosus involving the central nervous system. Ann Rheum Dis 66:470–475
Devauchelle-Pensec V, Pennec Y, Morvan J, Pers JO, Daridon C, Jousse-Joulin S, Roudaut A, Jamin C, Renaudineau Y, Roue IQ, Cochener B, Youinou P, Saraux A (2007) Improvement of Sjogren’s syndrome after two infusions of rituximab (anti-CD20). Arthritis Rheum 57:310–317
Cross AH, Stark JL, Lauber J, Ramsbottom MJ, Lyons JA (2006) Rituximab reduces B cells and T cells in cerebrospinal fluid of multiple sclerosis patients. J Neuroimmunol 180:63–70
Petereit HF, Moeller-Hartmann W, Reske D, Rubbert A (2008) Rituximab in a patient with multiple sclerosis—effect on B cells, plasma cells and intrathecal IgG synthesis. Acta Neurol Scand 117:399–403
Ireland S, Monson N (2011) Potential impact of B cells on T cell function in multiple sclerosis. Mult Scler Int 2011:423971
Lederer JA, Liou JS, Kim S, Rice N, Lichtman AH (1996) Regulation of NF-kappa B activation in T helper 1 and T helper 2 cells. J Immunol 156:56–63
Jazirehi AR, Huerta-Yepez S, Cheng G, Bonavida B (2005) Rituximab (chimeric anti-CD20 monoclonal antibody) inhibits the constitutive nuclear factor-{kappa}B signaling pathway in non-Hodgkin’s lymphoma B-cell lines: role in sensitization to chemotherapeutic drug-induced apoptosis. Cancer Res 65:264–276
Vigna-Perez M, Hernandez-Castro B, Paredes-Saharopulos O, Portales-Perez D, Baranda L, Abud-Mendoza C, Gonzalez-Amaro R (2006) Clinical and immunological effects of rituximab in patients with lupus nephritis refractory to conventional therapy: a pilot study. Arthritis Res Ther 8:R83
McFarland HF (2008) The B cell—old player, new position on the team. N Engl J Med 358:664–665
Bar-Or A, Fawaz L, Fan B, Darlington PJ, Rieger A, Ghorayeb C, Calabresi PA, Waubant E, Hauser SL, Zhang J, Smith CH (2010) Abnormal B-cell cytokine responses a trigger of T-cell-mediated disease in MS? Ann Neurol 67:452–461
Monson NL, Cravens PD, Frohman EM, Hawker K, Racke MK (2005) Effect of rituximab on the peripheral blood and cerebrospinal fluid B cells in patients with primary progressive multiple sclerosis. Arch Neurol 62:258–264
Geffard M, Duleu S, Bessede A, Vigier V, Bodet D, Mangas A, Covenas R (2012) GEMSP: a new therapeutic approach to multiple sclerosis. Cent Nerv Syst Agents Med Chem 12:173–181
Conflict of Interest
MB has received grant/research support from the NIH, Cooperative Research Centre, Simons Autism Foundation, Cancer Council of Victoria, Stanley Medical Research Foundation, MBF, NHMRC, Beyond Blue, Rotary Health, Geelong Medical Research Foundation, Bristol Myers Squibb, Eli Lilly, Glaxo SmithKline, Meat and Livestock Board, Organon, Novartis, MaynePharma, Servier, and Woolworths; has been a speaker for Astra Zeneca, Bristol Myers Squibb, Eli Lilly, Glaxo SmithKline, Janssen Cilag, Lundbeck, Merck, Pfizer, SanofiSynthelabo, Servier, Solvay, and Wyeth; and served as a consultant to Astra Zeneca, Bristol Myers Squibb, Eli Lilly, Glaxo SmithKline, Janssen Cilag, Lundbeck Merck, and Servier. The other authors do not report any conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Morris, G., Berk, M., Galecki, P. et al. The Emerging Role of Autoimmunity in Myalgic Encephalomyelitis/Chronic Fatigue Syndrome (ME/cfs). Mol Neurobiol 49, 741–756 (2014). https://doi.org/10.1007/s12035-013-8553-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-013-8553-0