Abstract
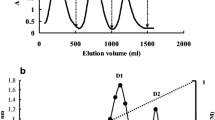
A 28-kDa ribonuclease, with an optimum pH of 4.0 and an optimum temperature at 58 °C, was isolated from fruiting bodies of the edible mushroom Hygrophorus russula. It was purified by ion exchange chromatography on carboxymethyl-cellulose, dithyaminoethyl-cellulose, quaternary amine-sepharose and sulphopropyl-sepharose, followed by fast protein liquid chromatography gel filtration on Superdex 75. The N-terminal amino acid sequence was ASAGG which showed homology to those of other fungal RNases to some degree. It exerted the highest RNase activity on poly C and poly U. The Michaelis constant (K m) value of the RNase on yeast tRNA was 3.6 μM, and the maximal velocity (V max) was 0.6 μM. The RNase activity was suppressed by some ions including Fe2+ and Zn2+ ions. The RNase inhibited the activity of HIV-1 reverse transcriptase with an IC50 of 4.64 μM.





Similar content being viewed by others
References
Norioka, S., Oneyama, C., Takuma, S., Shinkawa, T., Ishimizu, T., Nakanishi, T., et al. (2007). Purification and characterization of a non-S-RNase and S-RNases from styles of Japanese pear (Pyrus pyrifolia). Plant Physiology and Biochemistry, 45(12), 878–886.
Schein, A., Sheffy-Levin, S., Glaser, F., & Schuster, G. (2008). The RNase E/G-type endoribonuclease of higher plants is located in the chloroplast and cleaves RNA similarly to the E. coli enzyme. RNA, 14(6), 1057–1068.
Tanaka, N., Arai, J., Inokuchi, N., Koyama, T., Ohgi, K., Irie, M., et al. (2000). Crystal structure of a plant ribonuclease, RNase LE. Journal of Molecular Biology, 298(5), 859–873.
Fang, E. F., & Ng, T. B. (2011). Ribonucleases of different origins with a wide spectrum of medicinal applications. Biochimica et Biophysica Acta, 1815(1), 65–74.
Newton, D. L., Stockwin, L. H., & Rybak, S. M. (2009). Anti-CD22 onconase: preparation and characterization. Methods Mol Biol, 525, 425–43. xiv.
Mamolen, M., & Andrulis, E. D. (2009). Characterization of the Drosophila melanogaster Dis3 ribonuclease. Biochemical and Biophysical Research Communications, 390(3), 529–534.
Vourekas, A., Vryzaki, E., Toumpeki, C., Stamatopoulou, V., Monastirli, A., Tsambaos, D., et al. (2009). Partial purification and characterization of RNase P from human peripheral lymphocytes. Experimental Dermatology, 18(2), 130–133.
Aphanasenko, G. A., Dudkin, S. M., Kaminir, L. B., Leshchinskaya, I. B., & Severin, E. S. (1979). Primary structure of ribonuclease from Bacillus intermedius 7P. FEBS Letters, 97(1), 77–80.
Sana, B., Ghosh, D., Saha, M., & Mukherjee, J. (2008). Purification and characterization of an extracellular, uracil specific ribonuclease from a Bizionia species isolated from the marine environment of the Sundarbans. Microbiology Research, 163(1), 31–38.
Hlinkova, V., Urbanikova, L., Krajcikova, D., & Sevcik, J. (2001). Purification, crystallization and preliminary X-ray analysis of two crystal forms of ribonuclease Sa3. Acta Crystallographica Section D: Biological Crystallography, 57(Pt 5), 737–739.
Kobayashi, H., Inokuchi, N., Koyama, T., Watanabe, H., Iwama, M., Ohgi, K., et al. (1992). Primary structure of a base non-specific and adenylic acid preferential ribonuclease from the fruit bodies of Lentinus edodes. Bioscience, Biotechnology, and Biochemistry, 56(12), 2003–2010.
Wang, H., & Ng, T. B. (1999). Isolation of a new ribonuclease from fresh fruiting bodies of the straw mushroom. Biochemical and Biophysical Research Communications, 264(3), 714–718.
Ngai, P. H., Wang, H. X., & Ng, T. B. (2003). Purification and characterization of a ubiquitin-like peptide with macrophage stimulating, antiproliferative and ribonuclease activities from the mushroom Agrocybe cylindracea. Peptides, 24(5), 639–645.
Zhang, R. Y., Zhang, G. Q., Hu, D. D., Wang, H. X., & Ng, T. B. (2010). A novel ribonuclease with antiproliferative activity from fresh fruiting bodies of the edible mushroom Lyophyllum shimeiji. Biochemical Genetics, 48(7-8), 658–668.
Ercole, C., Spadaccini, R., Alfano, C., Tancredi, T., & Picone, D. (2007). A new mutant of bovine seminal ribonuclease with a reversed swapping propensity. Biochemistry, 46(8), 2227–2232.
Zhao, S., Zhao, Y., Li, S., Zhang, G., Wang, H., & Ng, T. B. (2010). An antiproliferative ribonuclease from fruiting bodies of the wild mushroom Russula delica. Journal of Microbiology and Biotechnology, 20(4), 693–699.
Guan, G. P., Wang, H. X., & Ng, T. B. (2007). A novel ribonuclease with antiproliferative activity from fresh fruiting bodies of the edible mushroom Hypsizigus marmoreus. Biochimica et Biophysica Acta, 1770(12), 1593–1597.
Wang, H. X., & Ng, T. B. (2001). Purification and characterization of a potent homodimeric guanine-specific ribonuclease from fresh mushroom (Pleurotus tuber-regium) sclerotia. The International Journal of Biochemistry and Cell Biology, 33(5), 483–490.
Wang, H., & Ng, T. B. (2003). A novel ribonuclease from the veiled lady mushroom Dictyophora indusiata. Biochemical Cell Biology, 81(6), 373–377.
Lam, Y. W., & Ng, T. B. (2001). A monomeric mannose-binding lectin from inner shoots of the edible chive (Allium tuberosum). Journal of Protein Chemistry, 20(5), 361–366.
Zhao, J. K., Wang, H. X., & Ng, T. B. (2009). Purification and characterization of a novel lectin from the toxic wild mushroom Inocybe umbrinella. Toxicon, 53(3), 360–366.
Zhao, Y. C., Zhang, G. Q., Ng, T. B., & Wang, H. X. (2011). A novel ribonuclease with potent HIV-1 reverse transcriptase inhibitory activity from cultured mushroom Schizophyllum commune. Journal of Microbiology, 49(5), 803–808.
Acknowledgements
This work was financially supported by National Grants of China (Biomass dissociation and low-molecular fragment green monomerization and transformation, 2010CB732202).
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Zhu, M., Xu, L., Chen, X. et al. A Novel Ribonuclease with HIV-1 Reverse Transcriptase Inhibitory Activity from the Edible Mushroom Hygrophorus russula . Appl Biochem Biotechnol 170, 219–230 (2013). https://doi.org/10.1007/s12010-013-0180-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-013-0180-8