Abstract
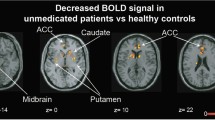
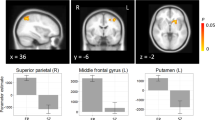
Prior functional magnetic resonance imaging (fMRI) studies have investigated the neural mechanisms underlying cognitive control in patients with psychosis with findings of both hypo- and hyperfrontality. One factor that may contribute to inconsistent findings is the use of complex and polyfactorial tasks to investigate frontal lobe functioning. In the current study we employed a simple response conflict task during fMRI to examine differences in brain activation between patients experiencing their first-episode of psychosis (n = 33) and age- and sex-matched healthy volunteers (n = 33). We further investigated whether baseline brain activation among patients predicted changes in symptom severity and treatment response following 12 weeks of controlled antipsychotic treatment. During the task subjects were instructed to press a response button on the same side or opposite side of a circle that appeared on either side of a central fixation point. Imaging data revealed that for the contrast of opposite-side vs. same-side, patients showed significantly greater activation compared with healthy volunteers in the anterior cingulate cortex and intraparietal sulcus. Among patients, greater baseline anterior cingulate cortex, temporal-parietal junction, and superior temporal cortex activation predicted greater symptom reduction and therapeutic response following treatment. All findings remained significant after covarying for task performance. Intact performance on this relatively parsimonious task was associated with frontal hyperactivity suggesting the need for patients to utilize greater neural resources to achieve task performance comparable to healthy individuals. Moreover, frontal hyperactivity observed using a simple fMRI task may provide a biomarker for predicting treatment response in first-episode psychosis.


Similar content being viewed by others
References
Baran, B., Karahanoglu, F. I., Agam, Y., Mantonakis, L., & Manoach, D. S. (2016). Failure to mobilize cognitive control for challenging tasks correlates with symptom severity in schizophrenia. Neuroimage Clinical, 12, 887–893. https://doi.org/10.1016/j.nicl.2016.10.020.
Botvinick, M. M., Cohen, J. D., & Carter, C. S. (2004). Conflict monitoring and anterior cingulate cortex: An update. Trends in Cognitive Sciences, 8(12), 539–546. https://doi.org/10.1016/j.tics.2004.10.003.
Callicott, J. H., Mattay, V. S., Verchinski, B. A., Marenco, S., Egan, M. F., & Weinberger, D. R. (2003). Complexity of prefrontal cortical dysfunction in schizophrenia: More than up or down. The American Journal of Psychiatry, 160(12), 2209–2215. https://doi.org/10.1176/appi.ajp.160.12.2209.
Carter, C. S., MacDonald 3rd, A. W., Ross, L. L., & Stenger, V. A. (2001). Anterior cingulate cortex activity and impaired self-monitoring of performance in patients with schizophrenia: An event-related fMRI study. The American Journal of Psychiatry, 158(9), 1423–1428.
Carter, J. D., Bizzell, J., Kim, C., Bellion, C., Carpenter, K. L., Dichter, G., et al. (2010). Attention deficits in schizophrenia--preliminary evidence of dissociable transient and sustained deficits. Schizophrenia Research, 122(1–3), 104–112. https://doi.org/10.1016/j.schres.2010.03.019.
Cox, R. W., Chen, G., Glen, D. R., Reynolds, R. C., & Taylor, P. A. (2017). FMRI clustering in AFNI: False-positive rates redux. Brain Connectivity, 7(3), 152–171. https://doi.org/10.1089/brain.2016.0475.
Dazzan, P., Arango, C., Fleischacker, W., Galderisi, S., Glenthoj, B., Leucht, S., et al. (2015). Magnetic resonance imaging and the prediction of outcome in first-episode schizophrenia: A review of current evidence and directions for future research. Schizophrenia Bulletin, 41(3), 574–583. https://doi.org/10.1093/schbul/sbv024.
Dove, A., Pollmann, S., Schubert, T., Wiggins, C. J., & von Cramon, D. Y. (2000). Prefrontal cortex activation in task switching: An event-related fMRI study. Brain Research. Cognitive Brain Research, 9(1), 103–109.
Ettinger, U., Williams, S. C., Fannon, D., Premkumar, P., Kuipers, E., Moller, H. J., et al. (2011). Functional magnetic resonance imaging of a parametric working memory task in schizophrenia: Relationship with performance and effects of antipsychotic treatment. Psychopharmacology, 216(1), 17–27. https://doi.org/10.1007/s00213-011-2214-7.
First, M. B., Spitzer, R. L., Gibbon, M., & Williams, J. B. W. (2002). Structured clinical interview for DSM-IV-TR axis I disorders, research version, non-patient edition (SCID-I/NP). New York: Biometrics Research, New York State Psychiatric Institute.
Fryer, S. L., Roach, B. J., Ford, J. M., Donaldson, K. R., Calhoun, V. D., Pearlson, G. D., et al. (2018). Should I stay or should I go? FMRI study of response inhibition in early illness schizophrenia and risk for psychosis. Schizophrenia Bulletin. https://doi.org/10.1093/schbul/sbx198.
Greicius, M. D., & Menon, V. (2004). Default-mode activity during a passive sensory task: Uncoupled from deactivation but impacting activation. Journal of Cognitive Neuroscience, 16(9), 1484–1492. https://doi.org/10.1162/0898929042568532.
Grutzmann, R., Endrass, T., Kaufmann, C., Allen, E., Eichele, T., & Kathmann, N. (2016). Presupplementary motor area contributes to altered error monitoring in obsessive-compulsive disorder. Biological Psychiatry, 80(7), 562–571. https://doi.org/10.1016/j.biopsych.2014.12.010.
Gur, R. E., Turetsky, B. I., Loughead, J., Snyder, W., Kohler, C., Elliott, M., et al. (2007). Visual attention circuitry in schizophrenia investigated with oddball event-related functional magnetic resonance imaging. The American Journal of Psychiatry, 164(3), 442–449. https://doi.org/10.1176/ajp.2007.164.3.442.
Hasenkamp, W., James, G. A., Boshoven, W., & Duncan, E. (2011). Altered engagement of attention and default networks during target detection in schizophrenia. Schizophrenia Research, 125(2–3), 169–173. https://doi.org/10.1016/j.schres.2010.08.041.
Hazlett, E. A., Goldstein, K. E., & Kolaitis, J. C. (2012). A review of structural MRI and diffusion tensor imaging in schizotypal personality disorder. Current Psychiatry Reports, 14(1), 70–78. https://doi.org/10.1007/s11920-011-0241-z.
Hommel, B. (2011). The Simon effect as tool and heuristic. Acta Psychologica, 136(2), 189–202. https://doi.org/10.1016/j.actpsy.2010.04.011.
Hummer, T. A., Hulvershorn, L. A., Karne, H. S., Gunn, A. D., Wang, Y., & Anand, A. (2013). Emotional response inhibition in bipolar disorder: A functional magnetic resonance imaging study of trait- and state-related abnormalities. Biological Psychiatry, 73(2), 136–143. https://doi.org/10.1016/j.biopsych.2012.06.036.
Ikuta, T., Szeszko, P. R., Gruner, P., DeRosse, P., Gallego, J., & Malhotra, A. K. (2012). Abnormal anterior cingulate cortex activity predicts functional disability in schizophrenia. Schizophrenia Research, 137(1–3), 267–268. https://doi.org/10.1016/j.schres.2011.12.021.
Jamadar, S., Michie, P., & Karayanidis, F. (2010). Compensatory mechanisms underlie intact task-switching performance in schizophrenia. Neuropsychologia, 48(5), 1305–1323. https://doi.org/10.1016/j.neuropsychologia.2009.12.034.
Jeong, B., & Kubicki, M. (2010). Reduced task-related suppression during semantic repetition priming in schizophrenia. Psychiatry Research, 181(2), 114–120. https://doi.org/10.1016/j.pscychresns.2009.09.005.
Jukuri, T., Kiviniemi, V., Nikkinen, J., Miettunen, J., Maki, P., Mukkala, S., et al. (2015). Central executive network in young people with familial risk for psychosis--the Oulu brain and mind study. Schizophrenia Research, 161(2–3), 177–183. https://doi.org/10.1016/j.schres.2014.11.003.
Kerns, J. G., Cohen, J. D., MacDonald 3rd, A. W., Cho, R. Y., Stenger, V. A., & Carter, C. S. (2004). Anterior cingulate conflict monitoring and adjustments in control. Science, 303(5660), 1023–1026. https://doi.org/10.1126/science.1089910.
Kerns, J. G., Cohen, J. D., MacDonald 3rd, A. W., Johnson, M. K., Stenger, V. A., Aizenstein, H., et al. (2005). Decreased conflict- and error-related activity in the anterior cingulate cortex in subjects with schizophrenia. The American Journal of Psychiatry, 162(10), 1833–1839. https://doi.org/10.1176/appi.ajp.162.10.1833.
Keshavan, M. S., Eack, S. M., Wojtalik, J. A., Prasad, K. M., Francis, A. N., Bhojraj, T. S., et al. (2011). A broad cortical reserve accelerates response to cognitive enhancement therapy in early course schizophrenia. Schizophrenia Research, 130(1–3), 123–129. https://doi.org/10.1016/j.schres.2011.05.001.
Kim, D. I., Manoach, D. S., Mathalon, D. H., Turner, J. A., Mannell, M., Brown, G. G., et al. (2009). Dysregulation of working memory and default-mode networks in schizophrenia using independent component analysis, an fBIRN and MCIC study. Human Brain Mapping, 30(11), 3795–3811. https://doi.org/10.1002/hbm.20807.
Kim, M. A., Tura, E., Potkin, S. G., Fallon, J. H., Manoach, D. S., Calhoun, V. D., et al. (2010). Working memory circuitry in schizophrenia shows widespread cortical inefficiency and compensation. Schizophrenia Research, 117(1), 42–51. https://doi.org/10.1016/j.schres.2009.12.014.
Kraguljac, N. V., White, D. M., Hadley, N., Hadley, J. A., Ver Hoef, L., Davis, E., et al. (2016). Aberrant hippocampal connectivity in Unmedicated patients with schizophrenia and effects of antipsychotic medication: A longitudinal resting state functional MRI study. Schizophrenia Bulletin, 42(4), 1046–1055. https://doi.org/10.1093/schbul/sbv228.
Kristo, G., Rutten, G. J., Raemaekers, M., de Gelder, B., Rombouts, S. A., & Ramsey, N. F. (2014). Task and task-free FMRI reproducibility comparison for motor network identification. Human Brain Mapping, 35(1), 340–352. https://doi.org/10.1002/hbm.22180.
Laurens, K. R., Kiehl, K. A., Ngan, E. T., & Liddle, P. F. (2005). Attention orienting dysfunction during salient novel stimulus processing in schizophrenia. Schizophrenia Research, 75(2–3), 159–171. https://doi.org/10.1016/j.schres.2004.12.010.
Lieberman, M. D., & Cunningham, W. A. (2009). Type I and type II error concerns in fMRI research: Re-balancing the scale. Social Cognitive and Affective Neuroscience, 4(4), 423–428. https://doi.org/10.1093/scan/nsp052.
MacDonald 3rd, A. W., & Carter, C. S. (2003). Event-related FMRI study of context processing in dorsolateral prefrontal cortex of patients with schizophrenia. Journal of Abnormal Psychology, 112(4), 689–697.
MacDonald 3rd, A. W., Cohen, J. D., Stenger, V. A., & Carter, C. S. (2000). Dissociating the role of the dorsolateral prefrontal and anterior cingulate cortex in cognitive control. Science, 288(5472), 1835–1838.
MacDonald 3rd, A. W., Carter, C. S., Kerns, J. G., Ursu, S., Barch, D. M., Holmes, A. J., et al. (2005). Specificity of prefrontal dysfunction and context processing deficits to schizophrenia in never-medicated patients with first-episode psychosis. The American Journal of Psychiatry, 162(3), 475–484. https://doi.org/10.1176/appi.ajp.162.3.475.
Manoach, D. S. (2003). Prefrontal cortex dysfunction during working memory performance in schizophrenia: Reconciling discrepant findings. Schizophrenia Research, 60(2–3), 285–298.
Mason, M. F., Norton, M. I., Van Horn, J. D., Wegner, D. M., Grafton, S. T., & Macrae, C. N. (2007). Wandering minds: The default network and stimulus-independent thought. Science, 315(5810), 393–395. https://doi.org/10.1126/science.1131295.
McAvoy, M. P., Ollinger, J. M., & Buckner, R. L. (2001). Cluster size thresholds for assessment of significant activation in fMRI. NeuroImage, 13(6), S198.
McKiernan, K. A., Kaufman, J. N., Kucera-Thompson, J., & Binder, J. R. (2003). A parametric manipulation of factors affecting task-induced deactivation in functional neuroimaging. Journal of Cognitive Neuroscience, 15(3), 394–408. https://doi.org/10.1162/089892903321593117.
McTeague, L. M., Huemer, J., Carreon, D. M., Jiang, Y., Eickhoff, S. B., & Etkin, A. (2017). Identification of common neural circuit disruptions in cognitive control across psychiatric disorders. The American Journal of Psychiatry, 174(7), 676–685. https://doi.org/10.1176/appi.ajp.2017.16040400.
Metzak, P. D., Riley, J. D., Wang, L., Whitman, J. C., Ngan, E. T., & Woodward, T. S. (2012). Decreased efficiency of task-positive and task-negative networks during working memory in schizophrenia. Schizophrenia Bulletin, 38(4), 803–813. https://doi.org/10.1093/schbul/sbq154.
Minzenberg, M. J., Laird, A. R., Thelen, S., Carter, C. S., & Glahn, D. C. (2009). Meta-analysis of 41 functional neuroimaging studies of executive function in schizophrenia. Archives of General Psychiatry, 66(8), 811–822. https://doi.org/10.1001/archgenpsychiatry.2009.91.
Morey, R. A., Inan, S., Mitchell, T. V., Perkins, D. O., Lieberman, J. A., & Belger, A. (2005). Imaging frontostriatal function in ultra-high-risk, early, and chronic schizophrenia during executive processing. Archives of General Psychiatry, 62(3), 254–262. https://doi.org/10.1001/archpsyc.62.3.254.
Murray, G. K., Corlett, P. R., & Fletcher, P. C. (2010). The neural underpinnings of associative learning in health and psychosis: How can performance be preserved when brain responses are abnormal? Schizophrenia Bulletin, 36(3), 465–471. https://doi.org/10.1093/schbul/sbq005.
Nejad, A. B., Madsen, K. H., Ebdrup, B. H., Siebner, H. R., Rasmussen, H., Aggernaes, B., et al. (2013). Neural markers of negative symptom outcomes in distributed working memory brain activity of antipsychotic-naive schizophrenia patients. The International Journal of Neuropsychopharmacology, 16(6), 1195–1204. https://doi.org/10.1017/S1461145712001253.
Pedersen, A., Wilmsmeier, A., Wiedl, K. H., Bauer, J., Kueppers, K., Koelkebeck, K., et al. (2012). Anterior cingulate cortex activation is related to learning potential on the WCST in schizophrenia patients. Brain and Cognition, 79(3), 245–251. https://doi.org/10.1016/j.bandc.2012.03.007.
Polli, F. E., Barton, J. J., Thakkar, K. N., Greve, D. N., Goff, D. C., Rauch, S. L., et al. (2008). Reduced error-related activation in two anterior cingulate circuits is related to impaired performance in schizophrenia. Brain, 131(Pt 4), 971–986. https://doi.org/10.1093/brain/awm307.
Potkin, S. G., Turner, J. A., Brown, G. G., McCarthy, G., Greve, D. N., Glover, G. H., et al. (2009). Working memory and DLPFC inefficiency in schizophrenia: The FBIRN study. Schizophrenia Bulletin, 35(1), 19–31. https://doi.org/10.1093/schbul/sbn162.
Proctor, R. W. (2011). Playing the Simon game: Use of the Simon task for investigating human information processing. Acta Psychologica, 136(2), 182–188. https://doi.org/10.1016/j.actpsy.2010.06.010.
Raichle, M. E., MacLeod, A. M., Snyder, A. Z., Powers, W. J., Gusnard, D. A., & Shulman, G. L. (2001). A default mode of brain function. Proceedings of the National Academy of Sciences of the United States of America, 98(2), 676–682. https://doi.org/10.1073/pnas.98.2.676.
Robinson, D. G., Gallego, J. A., John, M., Petrides, G., Hassoun, Y., Zhang, J. P., et al. (2015). A randomized comparison of aripiprazole and risperidone for the acute treatment of first-episode schizophrenia and related disorders: 3-month outcomes. Schizophrenia Bulletin, 41(6), 1227–1236. https://doi.org/10.1093/schbul/sbv125.
Rubia, K., Russell, T., Bullmore, E. T., Soni, W., Brammer, M. J., Simmons, A., et al. (2001). An fMRI study of reduced left prefrontal activation in schizophrenia during normal inhibitory function. Schizophrenia Research, 52(1–2), 47–55.
Salgado-Pineda, P., Fakra, E., Delaveau, P., McKenna, P. J., Pomarol-Clotet, E., & Blin, O. (2011). Correlated structural and functional brain abnormalities in the default mode network in schizophrenia patients. Schizophrenia Research, 125(2–3), 101–109. https://doi.org/10.1016/j.schres.2010.10.027.
Sarpal, D. K., Argyelan, M., Robinson, D. G., Szeszko, P. R., Karlsgodt, K. H., John, M., et al. (2016). Baseline striatal functional connectivity as a predictor of response to antipsychotic drug treatment. The American Journal of Psychiatry, 173(1), 69–77. https://doi.org/10.1176/appi.ajp.2015.14121571.
Schneider, F., Habel, U., Reske, M., Kellermann, T., Stocker, T., Shah, N. J., et al. (2007). Neural correlates of working memory dysfunction in first-episode schizophrenia patients: An fMRI multi-center study. Schizophrenia Research, 89(1–3), 198–210. https://doi.org/10.1016/j.schres.2006.07.021.
Shafritz, K. M., Kartheiser, P., & Belger, A. (2005). Dissociation of neural systems mediating shifts in behavioral response and cognitive set. NeuroImage, 25(2), 600–606.
Shulman, G. L., Fiez, J. A., Corbetta, M., Buckner, R. L., Miezin, F. M., Raichle, M. E., et al. (1997). Common blood flow changes across visual tasks: II. Decreases in cerebral cortex. Journal of Cognitive Neuroscience, 9(5), 648–663. https://doi.org/10.1162/jocn.1997.9.5.648.
Smucny, J., Lesh, T. A., Newton, K., Niendam, T. A., Ragland, J. D., & Carter, C. S. (2018). Levels of cognitive control: A functional magnetic resonance imaging-based test of an RDoC domain across bipolar disorder and schizophrenia. Neuropsychopharmacology, 43(3), 598–606. https://doi.org/10.1038/npp.2017.233.
Snitz, B. E., MacDonald 3rd, A., Cohen, J. D., Cho, R. Y., Becker, T., & Carter, C. S. (2005). Lateral and medial hypofrontality in first-episode schizophrenia: Functional activity in a medication-naive state and effects of short-term atypical antipsychotic treatment. The American Journal of Psychiatry, 162(12), 2322–2329. https://doi.org/10.1176/appi.ajp.162.12.2322.
Tan, H. Y., Sust, S., Buckholtz, J. W., Mattay, V. S., Meyer-Lindenberg, A., Egan, M. F., et al. (2006). Dysfunctional prefrontal regional specialization and compensation in schizophrenia. The American Journal of Psychiatry, 163(11), 1969–1977. https://doi.org/10.1176/ajp.2006.163.11.1969.
van Veelen, N. M., Vink, M., Ramsey, N. F., van Buuren, M., Hoogendam, J. M., & Kahn, R. S. (2011). Prefrontal lobe dysfunction predicts treatment response in medication-naive first-episode schizophrenia. Schizophrenia Research, 129(2–3), 156–162. https://doi.org/10.1016/j.schres.2011.03.026.
Vanes, L. D., Mouchlianitis, E., Collier, T., Averbeck, B. B., & Shergill, S. S. (2018). Differential neural reward mechanisms in treatment-responsive and treatment-resistant schizophrenia. Psychological Medicine, 1–10. https://doi.org/10.1017/S0033291718000041.
Vercammen, A., Morris, R., Green, M. J., Lenroot, R., Kulkarni, J., Carr, V. J., et al. (2012). Reduced neural activity of the prefrontal cognitive control circuitry during response inhibition to negative words in people with schizophrenia. Journal of Psychiatry & Neuroscience, 37(6), 379–388. https://doi.org/10.1503/jpn.110088.
Voegler, R., Becker, M. P., Nitsch, A., Miltner, W. H., & Straube, T. (2016). Aberrant network connectivity during error processing in patients with schizophrenia. Journal of Psychiatry & Neuroscience, 41(2), E3–E12.
Weinberger, D. R., & Berman, K. F. (1996). Prefrontal function in schizophrenia: Confounds and controversies. Philosophical Transactions of the Royal Society of London. Series B, Biological Sciences, 351(1346), 1495–1503. https://doi.org/10.1098/rstb.1996.0135.
Weiss, E. M., Golaszewski, S., Mottaghy, F. M., Hofer, A., Hausmann, A., Kemmler, G., et al. (2003). Brain activation patterns during a selective attention test-a functional MRI study in healthy volunteers and patients with schizophrenia. Psychiatry Research, 123(1), 1–15.
Weiss, E. M., Siedentopf, C., Golaszewski, S., Mottaghy, F. M., Hofer, A., Kremser, C., et al. (2007). Brain activation patterns during a selective attention test--a functional MRI study in healthy volunteers and unmedicated patients during an acute episode of schizophrenia. Psychiatry Research, 154(1), 31–40. https://doi.org/10.1016/j.pscychresns.2006.04.009.
Wessa, M., Houenou, J., Paillere-Martinot, M. L., Berthoz, S., Artiges, E., Leboyer, M., et al. (2007). Fronto-striatal overactivation in euthymic bipolar patients during an emotional go/nogo task. The American Journal of Psychiatry, 164(4), 638–646. https://doi.org/10.1176/appi.ajp.164.4.638.
Wiggins, J. L., Brotman, M. A., Adleman, N. E., Kim, P., Oakes, A. H., Reynolds, R. C., et al. (2016). Neural correlates of irritability in disruptive mood dysregulation and bipolar disorders. The American Journal of Psychiatry, 173(7), 722–730. https://doi.org/10.1176/appi.ajp.2015.15060833.
Wilmsmeier, A., Ohrmann, P., Suslow, T., Siegmund, A., Koelkebeck, K., Rothermundt, M., et al. (2010). Neural correlates of set-shifting: Decomposing executive functions in schizophrenia. Journal of Psychiatry & Neuroscience, 35(5), 321–329. https://doi.org/10.1503/jpn.090181.
Acknowledgements
We thank Daniel Bentley for assistance with data analysis.
Funding
This work was funded by grants from the National Institute of Mental Health to Dr. Szeszko (R01 MH076995), the NSLIJ Research Institute General Clinical Research Center (M01 RR018535), an Advanced Center for Intervention and Services Research (P30 MH090590) and a Center for Intervention Development and Applied Research (P50 MH080173), and by grants to Dr. Shafritz from Hofstra University.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors declare that they have no conflicts of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Rights and permissions
About this article
Cite this article
Shafritz, K.M., Ikuta, T., Greene, A. et al. Frontal lobe functioning during a simple response conflict task in first-episode psychosis and its relationship to treatment response. Brain Imaging and Behavior 13, 541–553 (2019). https://doi.org/10.1007/s11682-018-9876-2
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11682-018-9876-2