Abstract
Introduction
Colorectal cancer (CRC) is a clinically heterogeneous disease, which necessitates a variety of treatments and leads to different outcomes. Only some CRC patients will benefit from neoadjuvant chemotherapy (NACT).
Objectives
An accurate prediction of response to NACT in CRC patients would greatly facilitate optimal personalized management, which could improve their long-term survival and clinical outcomes.
Methods
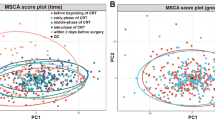
In this study, plasma metabolite profiling was performed to identify potential biomarker candidates that can predict response to NACT for CRC. Metabolic profiles of plasma from non-response (n = 30) and response (n = 27) patients to NACT were studied using UHPLC–quadruple time-of-flight)/mass spectrometry analyses and statistical analysis methods.
Results
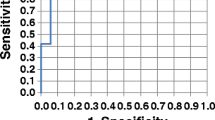
The concentrations of nine metabolites were significantly different when comparing response to NACT. The area under the receiver operating characteristic curve value of the potential biomarkers was up to 0.83 discriminating the non-response and response group to NACT, superior to the clinical parameters (carcinoembryonic antigen and carbohydrate antigen 199).
Conclusion
These results show promise for larger studies that could result in more personalized treatment protocols for CRC patients.




Similar content being viewed by others
Abbreviations
- CRC:
-
Colorectal cancer
- NACT:
-
Neoadjuvant chemotherapy
- MRI:
-
Magnetic resonance imaging
- CEA:
-
Carcinoembryonic antigen
- CA199:
-
Carbohydrate antigen 199
- NMR:
-
Nuclear magnetic resonance
- MS:
-
Mass spectrometry
- TNM:
-
Tumor–node–metastasis
- QC:
-
Quality control
- QTOF:
-
Quadruple time-of-flight
- RT:
-
Retention time
- m/z:
-
Mass-to-charge ratio
- PCA:
-
Principal component analysis
- PLS-DA:
-
Partial least-squares discriminant analysis
- VIP:
-
Variable importance in the projection
- AUC:
-
Area under the receiver operating characteristic curve
- RF:
-
Random forest
References
Balage, M., Dupont, J., Mothe-Satney, I., Tesseraud, S., Mosoni, L., & Dardevet, D. (2011). Leucine supplementation in rats induced a delay in muscle IR/PI3K signaling pathway associated with overall impaired glucose tolerance. The Journal of Nutritional Biochemistry, 22, 219–226.
Benjamini, Y. (2010). Discovering the false discovery rate. Journal of the Royal Statistical Society, 72, 405–416.
Derdak, Z., Mark, N. M., Beldi, G., Robson, S. C., Wands, J. R., & Baffy, G. (2008). The mitochondrial uncoupling protein-2 promotes chemoresistance in cancer cells. Cancer Research, 68, 2813–2819.
Duffy, M. J. (2001). Carcinoembryonic antigen as a marker for colorectal cancer: Is it clinically useful? Clinical Chemistry, 47, 624–630.
Dzik-Jurasz, A., et al. (2002). Diffusion MRI for prediction of response of rectal cancer to chemoradiation. The Lancet, 360, 307–308.
Edge, S. B., & Compton, C. C. (2010). The American Joint Committee on Cancer: The 7th edition of the AJCC cancer staging manual and the future of TNM. Annals of Surgical Oncology, 17, 1471–1474. https://doi.org/10.1245/s10434-010-0985-4.
Ferlay, J., et al. (2015). Cancer incidence and mortality worldwide: Sources, methods and major patterns in GLOBOCAN 2012. International Journal of Cancer, 136, E359–E386.
Glynne-Jones, R., Anyamene, N., Moran, B., & Harrison, M. (2012). Neoadjuvant chemotherapy in MRI-staged high-risk rectal cancer in addition to or as an alternative to preoperative chemoradiation? Annals of Oncology, 23, 2517–2526.
Hinault, C., Van Obberghen, E., & Mothe-Satney, I. (2006). Role of amino acids in insulin signaling in adipocytes and their potential to decrease insulin resistance of adipose tissue. The Journal of Nutritional Biochemistry, 17, 374–378.
Hou, Y., et al. (2014). A metabolomics approach for predicting the response to neoadjuvant chemotherapy in cervical cancer patients. Molecular Biosystems, 10, 2126–2133.
Kuhl, C., Tautenhahn, R., Bottcher, C., Larson, T. R., & Neumann, S. (2012). CAMERA: An integrated strategy for compound spectra extraction and annotation of liquid chromatography/mass spectrometry data sets. Analytical Chemistry, 84, 283–289. https://doi.org/10.1021/ac202450g.
Li, F., et al. (2013). Lipid profiling for early diagnosis and progression of colorectal cancer using direct-infusion electrospray ionization Fourier transform ion cyclotron resonance mass spectrometry. Rapid Communications in Mass Spectrometry, 27, 24–34. https://doi.org/10.1002/rcm.6420.
Liesenfeld, D. B., et al. (2015). Changes in urinary metabolic profiles of colorectal cancer patients enrolled in a prospective cohort study (ColoCare). Metabolomics, 11, 998–1012. https://doi.org/10.1007/s11306-014-0758-3.
Liu, J. J., Huang, B., & Hooi, S. C. (2006). Acetyl-keto-β-boswellic acid inhibits cellular proliferation through a p21-dependent pathway in colon cancer cells. British Journal of Pharmacology, 148, 1099–1107.
Liu, J.-J., Nilsson, Å, Oredsson, S., Badmaev, V., Zhao, W.-Z., & Duan, R.-D. (2002). Boswellic acids trigger apoptosis via a pathway dependent on caspase-8 activation but independent on Fas/Fas ligand interaction in colon cancer HT-29 cells. Carcinogenesis, 23, 2087–2093.
Nicholson, J. K., & Lindon, J. C. (2008). Systems biology: Metabonomics. Nature, 455, 1054.
Pang, X., et al. (2009). Acetyl-11-keto-β-boswellic acid inhibits prostate tumor growth by suppressing vascular endothelial growth factor receptor 2-mediated angiogenesis. Cancer Research, 69, 5893–5900.
Qiu, Y., et al. (2014). A distinct metabolic signature of human colorectal cancer with prognostic potential. Clinical Cancer Research, 20, 2136–2146. https://doi.org/10.1158/1078-0432.ccr-13-1939.
Rosenthal, D. I., et al. (2008). Phase I study of preoperative radiation therapy with concurrent infusional 5-fluorouracil and oxaliplatin followed by surgery and postoperative 5-fluorouracil plus leucovorin for T3/T4 rectal adenocarcinoma: ECOG E1297. International Journal of Radiation Oncology* Biology* Physics, 72, 108–113.
Salvador, J. A., Moreira, V. M., Gonçalves, B. M., Leal, A. S., & Jing, Y. (2012). Ursane-type pentacyclic triterpenoids as useful platforms to discover anticancer drugs. Natural Product Reports, 29, 1463–1479.
Schrag, D., et al. (2014). Neoadjuvant chemotherapy without routine use of radiation therapy for patients with locally advanced rectal cancer: A pilot trial. Journal of Clinical Oncology, 32, 513.
Shah, B. A., Qazi, G. N., & Taneja, S. C. (2009). Boswellic acids: A group of medicinally important compounds. Natural Product Reports, 26, 72–89.
Smith, C. A., Want, E. J., O’Maille, G., Abagyan, R., & Siuzdak, G. (2006). XCMS: Processing mass spectrometry data for metabolite profiling using nonlinear peak alignment, matching, and identification. Analytical Chemistry, 78, 779–787. https://doi.org/10.1021/ac051437y.
Spratlin, J. L., Serkova, N. J., & Eckhardt, S. G. (2009). Clinical applications of metabolomics in oncology: a review. Clinical Cancer Research, 15, 431–440.
Wang, J., et al. (2016). Serum metabolomics for early diagnosis of esophageal squamous cell carcinoma by UHPLC–QTOF/MS. Metabolomics, 12, 116.
Wei, S., et al. (2013). Metabolomics approach for predicting response to neoadjuvant chemotherapy for breast cancer. Molecular Oncology, 7, 297–307.
Zheng, Z.-X., Zheng, R.-S., Zhang, S.-W., & Chen, W.-Q. (2013). Colorectal cancer incidence and mortality in China, 2010. Asian Pacific Journal of Cancer Prevention, 15, 8455–8460.
Zhu, J., et al. (2014). Colorectal cancer detection using targeted serum metabolic profiling. Journal of Proteome Research, 13, 4120–4130. https://doi.org/10.1021/pr500494u.
Funding
This study was funded by National Natural Science Foundation of China (Grant Numbers 81773551, 81473072) and Health and Family Planning Commission of Heilongjiang Province Scientific Program (Grant Number 2014-349).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the Ethical Standards of the Institutional and/or National Research Committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Yang, K., Zhang, F., Han, P. et al. Metabolomics approach for predicting response to neoadjuvant chemotherapy for colorectal cancer. Metabolomics 14, 110 (2018). https://doi.org/10.1007/s11306-018-1406-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11306-018-1406-0