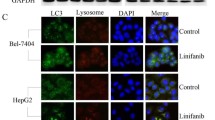
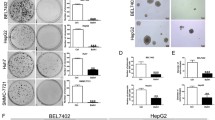
Abstract
Extracellular adenosine triphosphate (eATP) transduces purinergic signal and plays an important regulatory role in many biological processes, including tumor cell growth and cell death. A large amount of eATP exists in the fast-growing tumor center and inflammatory tumor microenvironment. Tumor cells could acquire anoikis resistance and anchorage independence in tumor microenvironment and further cause metastatic lesion. Whether such a high amount of eATP has any effect on the anchored and non-anchored tumor cells in tumor microenvironment has not been elucidated and is investigated in this study. Our data showed that autophagy helped hepatoma cells to maintain survival under the treatment of no more than 1 mM of eATP. Only when eATP concentration reached a relatively high level (2.5 mM), cell organelle could not be further maintained by autophagy, and apoptosis and cell death occurred. In hepatoma cells under treatment of 2.5 mM of eATP, an AMP-activated protein kinase (AMPK) pathway was dramatically activated while mTOR signaling pathway was suppressed in coordination with apoptosis. Further investigation showed that the AMPK/mTOR axis played a key role in tipping the balance between autophagy-mediated cell survival and apoptosis-induced cell death under the treatment of eATP. This work provides evidence to explain how hepatoma cells escape from eATP-induced cytotoxicity as well as offers an important clue to consider effective manipulation of cancer.










Similar content being viewed by others
Abbreviations
- ATP:
-
Adenosine triphosphate
- eATP:
-
Extracellular adenosine triphosphate
- PCR:
-
Polymerase chain reaction
- RT-PCR:
-
Reverse transcriptase polymerase chain reaction
- 3-MA:
-
3-Methyladenine
- LC3:
-
Microtubule-associated protein light chain 3
- CCK-8:
-
Cell Counting Kit-8
- Min:
-
Minutes
- H:
-
Hours
- mTOR:
-
Mammalian target of rapamycin
- S6K1:
-
Ribosomal S6 kinase 1
- PBS:
-
Phosphate-buffered saline
- FBS:
-
Fetal bovine serum
References
Bours MJ, Dagnelie PC, Giuliani AL, Wesselius A, Di Virgilio F (2011) P2 receptors and extracellular ATP: a novel homeostatic pathway in inflammation. Front Biosci (Schol Ed) 3:1443–1456
Apolloni S, Montilli C, Finocchi P, Amadio S (2009) Membrane compartments and purinergic signalling: P2X receptors in neurodegenerative and neuroinflammatory events. FEBS J 276(2):354–364
Deli T, Csernoch L (2008) Extracellular ATP and cancer: an overview with special reference to P2 purinergic receptors. Pathol Oncol Res 14(3):219–231
Pellegatti P, Raffaghello L, Bianchi G, Piccardi F, Pistoia V, Di Virgilio F (2008) Increased level of extracellular ATP at tumor sites: in vivo imaging with plasma membrane luciferase. PLoS One 3(7):e2599
Robson SC, Sevigny J, Zimmermann H (2006) The E-NTPDase family of ectonucleotidases: structure function relationships and pathophysiological significance. Purinergic Signal 2(2):409–430
Feng L, Sun X, Csizmadia E, Han L, Bian S, Murakami T, Wang X, Robson SC, Wu Y (2011) Vascular CD39/ENTPD1 directly promotes tumor cell growth by scavenging extracellular adenosine triphosphate. Neoplasia 13(3):206–216
Taddei ML, Giannoni E, Fiaschi T, Chiarugi P (2011) Anoikis: an emerging hallmark in health and diseases. J Pathol. doi:10.1002/path.3000
Taddei ML, Giannoni E, Fiaschi T, Chiarugi P (2012) Anoikis: an emerging hallmark in health and diseases. J Pathol 226(2):380–393
Zhang Z, Cao L, Li J, Liang X, Liu Y, Liu H, Du J, Qu Z, Cui M, Liu S, Gao L, Ma C, Zhang L, Han L, Sun W (2008) Acquisition of anoikis resistance reveals a synoikis-like survival style in BEL7402 hepatoma cells. Cancer Lett 267(1):106–115
Giansanti V, Torriglia A, Scovassi AI (2011) Conversation between apoptosis and autophagy: "is it your turn or mine?". Apoptosis 16(4):321–333
Thorburn A (2008) Apoptosis and autophagy: regulatory connections between two supposedly different processes. Apoptosis 13(1):1–9
Eisenberg-Lerner A, Bialik S, Simon HU, Kimchi A (2009) Life and death partners: apoptosis, autophagy and the cross-talk between them. Cell Death Differ 16(7):966–975
Corcelle EA, Puustinen P, Jaattela M (2009) Apoptosis and autophagy: targeting autophagy signalling in cancer cells—‘trick or treats’? FEBS J 276(21):6084–6096
Mora R, Regnier-Vigouroux A (2009) Autophagy-driven cell fate decision maker: activated microglia induce specific death of glioma cells by a blockade of basal autophagic flux and secondary apoptosis/necrosis. Autophagy 5(3):419–421
Guillon-Munos A, van Bemmelen MX, Clarke PG (2006) Autophagy can be a killer even in apoptosis-competent cells. Autophagy 2(2):140–142
Cao L, Han L, Zhang Z, Li J, Qu Z, Du J, Liang X, Liu Y, Liu H, Shi Y, Liu S, Gao L, Sun W (2009) Involvement of anoikis-resistance in the metastasis of hepatoma cells. Exp Cell Res 315(7):1148–1156
Almonte-Becerril M, Navarro-Garcia F, Gonzalez-Robles A, Vega-Lopez MA, Lavalle C, Kouri JB (2010) Cell death of chondrocytes is a combination between apoptosis and autophagy during the pathogenesis of osteoarthritis within an experimental model. Apoptosis 15(5):631–638
Kim YM, Hwang JT, Kwak DW, Lee YK, Park OJ (2007) Involvement of AMPK signaling cascade in capsaicin-induced apoptosis of HT-29 colon cancer cells. Ann N Y Acad Sci 1095:496–503
Wang X, Pan X, Song J (2010) AMP-activated protein kinase is required for induction of apoptosis and epithelial-to-mesenchymal transition. Cell Signal 22(11):1790–1797
Ikenoue T, Hong S, Inoki K (2009) Monitoring mammalian target of rapamycin (mTOR) activity. Methods Enzymol 452:165–180
Villalonga P, Fernandez de Mattos S, Ridley AJ (2009) RhoE inhibits 4E-BP1 phosphorylation and eIF4E function impairing cap-dependent translation. J Biol Chem 284(51):35287–35296
Moody CA, Scott RS, Amirghahari N, Nathan CO, Young LS, Dawson CW, Sixbey JW (2005) Modulation of the cell growth regulator mTOR by Epstein-Barr virus-encoded LMP2A. J Virol 79(9):5499–5506
Shabbir M, Burnstock G (2009) Purinergic receptor-mediated effects of adenosine 5'-triphosphate in urological malignant diseases. Int J Urol 16(2):143–150
Stagg J, Smyth MJ (2010) Extracellular adenosine triphosphate and adenosine in cancer. Oncogene 29(39):5346–5358
Zhang Y, Gong LH, Zhang HQ, Du Q, You JF, Tian XX, Fang WG (2010) Extracellular ATP enhances in vitro invasion of prostate cancer cells by activating Rho GTPase and upregulating MMPs expression. Cancer Lett 293(2):189–197
Yaguchi T, Saito M, Yasuda Y, Kanno T, Nakano T, Nishizaki T (2010) Higher concentrations of extracellular ATP suppress proliferation of Caco-2 human colonic cancer cells via an unknown receptor involving PKC inhibition. Cell Physiol Biochem 26(2):125–134
White N, Burnstock G (2006) P2 receptors and cancer. Trends Pharmacol Sci 27(4):211–217
Pantovic A, Krstic A, Janjetovic K, Kocic J, Harhaji-Trajkovic L, Bugarski D, Trajkovic V (2013) Coordinated time-dependent modulation of AMPK/Akt/mTOR signaling and autophagy controls osteogenic differentiation of human mesenchymal stem cells. Bone 52(1):524–531
Liu X, Yuan H, Niu Y, Niu W, Fu L (2012) The role of AMPK/mTOR/S6K1 signaling axis in mediating the physiological process of exercise-induced insulin sensitization in skeletal muscle of C57BL/6 mice. Biochim Biophys Acta 1822(11):1716–1726
Green AS, Chapuis N, Lacombe C, Mayeux P, Bouscary D, Tamburini J (2011) LKB1/AMPK/mTOR signaling pathway in hematological malignancies: from metabolism to cancer cell biology. Cell Cycle 10(13):2115–2120
Bian S, Sun X, Bai A, Zhang C, Li L, Enjyoji K, Junger WG, Robson SC, Wu Y (2013) P2X7 integrates PI3K/AKT and AMPK-PRAS40-mTOR signaling pathways to mediate tumor cell death. PLoS One 8(4):e60184
Sun X, Han L, Seth P, Bian S, Li L, Csizmadia E, Junger WG, Schmelzle M, Usheva A, Tapper EB, Baffy G, Sukhatme VP, Wu Y, Robson SC (2013) Disordered purinergic signaling and abnormal cellular metabolism are associated with development of liver cancer in Cd39/ENTPD1 null mice. Hepatology 57(1):205–216
Grander D, Panaretakis T (2010) Autophagy: cancer therapy's friend or foe? Future Med Chem 2(2):285–297
Moretti L, Yang ES, Kim KW, Lu B (2007) Autophagy signaling in cancer and its potential as novel target to improve anticancer therapy. Drug Resist Updat 10(4–5):135–143
Clayton A, Al-Taei S, Webber J, Mason MD, Tabi Z (2011) Cancer exosomes express CD39 and CD73, which suppress T cells through adenosine production. J Immunol 187(2):676–683
Wood E, Broekman MJ, Kirley TL, Diani-Moore S, Tickner M, Drosopoulos JH, Islam N, Park JI, Marcus AJ, Rifkind AB (2002) Cell-type specificity of ectonucleotidase expression and upregulation by 2,3,7,8-tetrachlorodibenzo-p-dioxin. Arch Biochem Biophys 407(1):49–62
Fausther M, Lecka J, Kukulski F, Levesque SA, Pelletier J, Zimmermann H, Dranoff JA, Sevigny J (2007) Cloning, purification, and identification of the liver canalicular ecto-ATPase as NTPDase8. Am J Physiol Gastrointest Liver Physiol 292(3):G785–G795
Acknowledgments
This study was supported by the National Nature Science Foundation of China (No. 81172352, No. 30700357), Science and Technology Development Project of Shandong Province (2011GGE27020), and Project for Outstanding Young Scientist in Jinan City (No.20080211 ).
Conflict of interest
The authors declare no conflict of interests.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wei, Q., Zhang, Y., Sun, L. et al. High dose of extracellular ATP switched autophagy to apoptosis in anchorage-dependent and anchorage-independent hepatoma cells. Purinergic Signalling 9, 585–598 (2013). https://doi.org/10.1007/s11302-013-9369-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11302-013-9369-0