Abstract
Purpose
The function of ABCG2 (BCRP), a member of the ATP-binding cassette (ABC) superfamily of membrane-associated drug transporters, at the blood-brain barrier remains highly controversial. This project investigates the functional expression of endogenous ABCG2 in cultures of human and rodent brain cellular compartments.
Materials and Methods
RT-PCR, western blot and fluorescent immunocytochemical analyses were performed on ABCG2-overexpressing human breast cancer (MCF-MX100) cells, human and rat brain microvessel endothelial (HBEC and RBE4, respectively), and rat glial cells.
Results
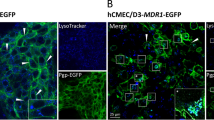
RT-PCR analysis detected ABCG2 mRNA in all the cell culture systems. Western blot analysis with anti-ABCG2 monoclonal BXP-21 antibody detected a robust band at ∼72 kDa in the ABCG2-overexpressing MCF-MX100 cell line, whereas low expression was found in human and rat brain cell systems. Immunofluorescence microscopy detected predominant plasma membrane localization of ABCG2 in MCF-MX100 cells but weak signal in all brain cellular compartments. In the presence of ABCG2 inhibitors, the accumulation of 3H-mitoxantrone and pheophorbide A, two established ABCG2 substrates, was significantly increased in MCF-MX100 cells but not in the human and rodent brain cell culture systems.
Conclusions
Our data show low endogenous ABCG2 protein expression, localization and activity in cultures of human and rat brain microvessel endothelial and glial cells.


















Similar content being viewed by others
Abbreviations
- ABC:
-
ATP-binding cassette
- BCRP:
-
breast cancer resistance protein
- HBEC:
-
human brain endothelial cells
- MRP:
-
multidrug resistance-associated protein
- P-gp:
-
P-glycoprotein
References
L. A. Doyle, W. Yang, L. V. Abruzzo, T. Krogmann, Y. Gao, A. K. Rishi, and D. D. Ross. A multidrug resistance transporter from human MCF-7 breast cancer cells. Proc. Natl. Acad. Sci. U. S. A. 95:15665–15670 (1998).
J. D. Allen, R. F. Brinkhuis, J. Wijnholds, and A. H. Schinkel. The mouse Bcrp1/Mxr/Abcp gene: amplification and overexpression in cell lines selected for resistance to topotecan, mitoxantrone, or doxorubicin. Cancer Res. 59:4237–4241 (1999).
T. Eisenblatter, S. Huwel, and H. J. Galla. Characterisation of the brain multidrug resistance protein (BMDP/ABCG2/BCRP) expressed at the blood-brain barrier. Brain Res. 971:221–231 (2003).
S. Hori, S. Ohtsuki, M. Tachikawa, N. Kimura, T. Kondo, M. Watanabe, E. Nakashima, and T. Terasaki. Functional expression of rat ABCG2 on the luminal side of brain capillaries and its enhancement by astrocyte-derived soluble factor(s). J. Neurochem. 90:526–536 (2004).
K. E. Berge, H. Tian, G. A. Graf, L. Yu, N. V. Grishin, J. Schultz, P. Kwiterovich, B. Shan, R. Barnes, and H. H. Hobbs. Accumulation of dietary cholesterol in sitosterolemia caused by mutations in adjacent ABC transporters. Science 290:1771–1775 (2000).
J. Xu, Y. Liu, Y. Yang, S. Bates, and J. T. Zhang. Characterization of oligomeric human half-ABC transporter ATP binding cassette G2. J. Biol. Chem. 279:19781–19789 (2004).
F. Staud and P. Pavek. Breast cancer resistance protein (BCRP/ABCG2). Int. J. Biochem. Cell Biol. 37:720–725 (2005).
M. Maliepaard, G. L. Scheffer, I. F. Faneyte, M. A. van Gastelen, A. C. L. M. Pijnenborg, A. H. Schinkel, M. J. Van de Vijver, R. J. Scheper, and J. H. M. Schellens. Subcellular localization and distribution of the breast cancer resistance protein transporter in normal human tissues. Cancer Res. 61:3458–3464 (2001).
H. C. Cooray, C. G. Blackmore, L. Maskell, and M. A. Barrand. Localisation of breast cancer resistance protein in microvessel endothelium of human brain. NeuroReport 13:2059–2063 (2002).
W. Zhang, J. Mojsilovic-Petrovic, M. F. Andrade, H. Zhang, M. Ball, and D. B. Stanimirovic. The expression and functional characterization of ABCG2 in brain endothelial cells and vessels. FASEB J. 17:2085–2087 (2003).
Y. J. Lee, H. Kusuhara, J. W. Jonker, A. H. Schinkel, and Y. Sugiyama. Investigation of efflux transport of dehydroepiandrosterone sulfate and mitoxantrone at the mouse blood-brain barrier: A minor role of breast cancer resistance protein. J. Pharmacol. Exp. Ther. 312:44–52 (2005).
A. E. van Herwaarden, J. W. Jonker, E. Wagenaar, R. F. Brinkhuis, J. H. Schellens, J. H. Beijnen, and A. H. Schinkel. The breast cancer resistance protein (Bcrp1/Abcg2) restricts exposure to the dietary carcinogen 2-amino-1-methyl-6-phenylimidazo(4,5-b)pyridine. Cancer Res. 63:6447–6452 (2003).
S. Cisternino, C. Mercier, F. Bourasset, F. Roux, and J. M. Scherrmann. Expression, up-regulation, and transport activity of the multidrug-resistance protein Abcg2 at the mouse blood-brain barrier. Cancer Res. 64:3296–3301 (2004).
P. Breedveld, D. Pluim, G. Cipriani, P. Wielinga, O. van Tellingen, A. H. Schinkel, and J. H. M. Schellens. The effect of Bcrp1 (Abcg2) on the in vivo pharmacokinetics and brain penetration of imatinib mesylate (gleevec): Implications for the use of breast cancer resistance protein and P-glycoprotein inhibitors to enable the brain penetration of imatinib in patients. Cancer Res. 65:2577–2582 (2005).
I. Becker, K. F. Becker, R. Meyermann, and V. Hollt. The multidrug-resistance gene MDR1 is expressed in human glial tumors. Acta Neuropathol. (Berl). 82:516–519 (1991).
H. A. Volk and W. Loscher. Multidrug resistance in epilepsy: Rats with drug-resistant seizures exhibit enhanced brain expression of P-glycoprotein compared with rats with drug-responsive seizures. Brain 128:1358–1368 (2005).
A. Prat, K. Biernacki, B. Becher, and J. P. Antel. B7 expression and antigen presentation by human brain endothelial cells: Requirement for proinflammatory cytokines. J. Neuropathol. Exp. Neurol. 59:129–136 (2000).
F. Roux, O. Durieu-Trautmann, N. Chaverot, M. Claire, P. Mailly, J. M. Bourre, A. D. Strosberg, and P.-O. Couraud. Regulation of gamma-glutamyl transpeptidase and alkaline phosphatase activities in immortalized rat brain microvessel endothelial cells. J. Cell. Physiol. 159:101–113 (1994).
R. Bendayan, G. Lee, and M. Bendayan. Functional expression and localization of P-glycoprotein at the blood-brain barrier. Microsc. Res. Tech. 57:365–380 (2002).
P. T. Ronaldson, M. Bendayan, D. Gingras, M. Piquette-Miller, and R. Bendayan. Cellular localization and functional expression of P-glycoprotein in rat astrocyte cultures. J. Neurochem. 89:788–800 (2004).
G. Lee, L. Schlichter, M. Bendayan, and R. Bendayan. Functional expression of P-glycoprotein in rat brain microglia. J. Pharmacol. Exp. Ther. 299:204–212 (2001).
R. W. Robey, Y. Honjo, A. van de Laar, K. Miyake, J. T. Regis, T. Litman, and S. E. Bates. A functional assay for detection of the mitoxantrone resistance protein, MXR (ABCG2). Biochim. Biophys. Acta 1512:171–182 (2001).
R. W. Robey, K. Steadman, O. Polgar, and S. E. Bates. ABCG2-mediated transport of photosensitizers: Potential impact on photodynamic therapy. Cancer Biol. Ther. 4:187–194 (2005).
H. Minderman, A. Suvannasankha, K. L. O’Loughlin, G. L. Scheffer, R. J. Scheper, R. W. Robey, and M. R. Maer. Flow cytometric analysis of breast cancer resistance protein expression and function. Cytometry 48:59–65 (2002).
T. Litman, M. Brangi, E. Hudson, P. Fetsch, A. Abati, D. D. Ross, K. Miyake, J. H. Resau, and S. E. Bates. The multidrug-resistant phenotype associated with overexpression of the new ABC half-transporter, MXR (ABCG2). J. Cell Sci. 113:2011–2021 (2000).
A. Gupta, Y. Zhang, J. D. Unadkat, and Q. Mao. HIV protease inhibitors are inhibitors but not substrates of the human breast cancer resistance protein (BCRP/ABCG2). J. Pharmacol. Exp. Ther. 310:334–341 (2004).
Y. Honjo, C. A. Hrycyna, Q. W. Yan, W. Y. Medina-Perez, R. W. Robey, A. van de Laar, T. Litman, M. Dean, and S. E. Bates. Acquired mutations in the MXR/BCRP/ABCP gene alter substrate specificity in MXR/BCRP/ABCP-overexpressing cells. Cancer Res. 61:6635–6639 (2001).
A. Regina, A. Koman, M. Piciotti, B. El Hafny, M. S. Center, R. Bergmann, P.-O. Couraud, and F. Roux. Mrp1 multidrug resistance-associated protein and P-glycoprotein expression in rat brain microvessel endothelial cells. J. Neurochem. 71:705–715 (1998).
S. Dallas, L. Schlichter, and R. Bendayan. Multidrug resistance protein (MRP) 4- and MRP 5-mediated efflux of 9-(2-phosphonylmethoxyethyl)adenine by microglia. J. Pharmacol. Exp. Ther. 309:1221–1229 (2004).
A. van Loevezijn, J. D. Allen, A. H. Schinkel, and G. J. Koomen. Inhibition of BCRP-mediated drug efflux by fumitremorgin-type indolyl diketopiperazines. Bioorg. Med. Chem. Lett. 11:29–32 (2001).
R. W. Robey, K. Steadman, O. Polgar, K. Morisaki, M. Blayney, P. Mistry, and S. E. Bates. Pheophorbide a is a specific probe for ABCG2 function and inhibition. Cancer Res. 64:1242–1246 (2004).
J. W. Jonker, M. Buitelaar, E. Wagenaar, M. A. Van Der Valk, G. L. Scheffer, R. J. Scheper, T. Plosch, F. Kuiper, R. P. J Oude Elferink, H. Rosing, J. H. Beijnen, and A. H. Schinkel. The breast cancer resistance protein protects against a major chlorophyll-derived dietary phototoxin and protoporphyria. Proc. Natl. Acad. Sci. USA 99:15649–15654 (2002).
E. Aronica, J. A. Gorter, S. Redeker, E. A. van Vliet, M. Ramkema, G. L. Scheffer, R. J. Scheper, P. Van Der Valk, S. Leenstra, J. C. Baayen, W. G. M. Spliet, and D. Troost. Localization of breast cancer resistance protein (BCRP) in microvessel endothelium of human control and epileptic brain. Epilepsia 46:849–857 (2005).
E. Rocchi, A. Khodjakov, E. L. Volk, C. H. Yang, T. Litman, S. E. Bates, and E. Schneider. The product of the ABC half-transporter gene ABCG2 (BCRP/MXR/ABCP) is expressed in the plasma membrane. Biochem. Biophys. Res. Commun. 271:42–46 (2000).
X. Decleves, A. Regina, J. L. Laplanche, F. Roux, B. Boval, J. M. Launay, and J. M. Scherrmann. Functional expression of P-glycoprotein and multidrug resistance-associated protein (Mrp1) in primary cultures of rat astrocytes. J. Neurosci. Res. 60:594–601 (2000).
S. Dallas, X. Zhu, S. Baruchel, L. Schlichter, and R. Bendayan. Functional expression of the multidrug resistance protein 1 in microglia. J. Pharmacol. Exp. Ther. 307:282–290 (2003).
C. Ozvegy, A. Varadi, and B. Sarkadi. Characterization of drug transport, ATP hydrolysis, and nucleotide trapping by the human ABCG2 multidrug transporter. modulation of substrate specificity by a point mutation. J. Biol. Chem. 277:47980–47990 (2002).
B. Bauer, A. M. Hartz, G. Fricker, and D. S. Miller. Pregnane X receptor up-regulation of P-glycoprotein expression and transport function at the blood–brain barrier. Mol. Pharmacol. 66:413–419 (2004).
R. Bendayan, P. T. Ronaldson, D. Gingras, and M. Bendayan. In Situ localization of P-glycoprotein (ABCB1) in human and rat brain. J. Histochem. and Cytochem. 54:1159–1167 (2006).
Acknowledgments
This work is supported by grants to Reina Bendayan from the Canadian Institutes of Health Research (MOP-56976) and the Ontario HIV Treatment Network (OHTN). Gloria Lee is a recipient of an OHTN Studentship Award. Alexander Prat holds the Donald Paty Career Award from the Multiple Sclerosis Society of Canada as well as “Chercheur Boursier Junior” from the Fonds de la Recherche en Sante du Quebec. Karolina Wosik is a recipient of a fellowship from the Multiple Sclerosis Society of Canada. We would like to thank Dr. J. Henderson for the help and advice with the undertaking of fluorescence microscopy studies and Drs S. Bates and F. Roux for providing the MCF-7 and RBE4 cell culture systems, respectively.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lee, G., Babakhanian, K., Ramaswamy, M. et al. Expression of the ATP-binding Cassette Membrane Transporter, ABCG2, in Human and Rodent Brain Microvessel Endothelial and Glial Cell Culture Systems. Pharm Res 24, 1262–1274 (2007). https://doi.org/10.1007/s11095-007-9244-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11095-007-9244-1