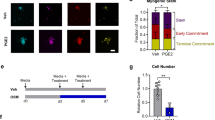
Abstract
Research on skeletal muscles suffers from a lack of appropriate human models to study muscle formation and regeneration on the regulatory level of single cells. This hampers both basic understanding and the development of new therapeutic approaches. The use of imaging multicolour flow cytometry and myogenic stem cells can help fill this void by allowing researchers to visualize and quantify the reaction of individual cultured cells to bioactives or other physiological impulses. As proof of concept, we subjected human CD56+ satellite cells to reference bioactives follistatin and Malva sylvestris extracts and then used imaging multicolor flow cytometry to visualize the stepwise activation of myogenic factors MyoD and myogenin in individual cells. This approach enabled us to evaluate the potency of these bioactives to stimulate muscle commitment. To validate this method, we used multi-photon confocal microscopy to confirm the potential of bioactives to stimulate muscle differentiation and expression of desmin. Imaging multicolor flow cytometry revealed statistically significant differences between treated and untreated groups of myogenic progenitors and we propose the utilization of this concept as an integral part of future muscle research strategies.




Similar content being viewed by others

References
Ali SA et al (2008) Phenotypic transcription factors epigenetically mediate cell growth control. Proc Natl Acad Sci USA 105:6632–6637. doi:10.1073/pnas.0800970105
Anderson JE (2006) The satellite cell as a companion in skeletal muscle plasticity: currency, conveyance, clue, connector and colander. J Exp Biol 209:2276–2292. doi:10.1242/jeb.02088
Barteneva NS, Fasler-Kan E, Vorobjev IA (2012) Imaging flow cytometry: coping with heterogeneity in biological systems. J Histochem Cytochem Off J Histochem Soc 60:723–733. doi:10.1369/0022155412453052
Bian W, Bursac N (2009) Engineered skeletal muscle tissue networks with controllable architecture. Biomaterials 30:1401–1412. doi:10.1016/j.biomaterials.2008.11.015
Carlson B (2008) Muscle Regeneration in Animal Models. In: Skeletal Muscle Repair and Regeneration, vol 3. Advances in Muscle Research. Springer Netherlands, pp 163–180. doi:10.1007/978-1-4020-6768-6_8
Castiglioni A et al (2014) Isolation of Progenitors that Exhibit Myogenic/Osteogenic Bipotency In Vitro by Fluorescence-Activated Cell Sorting from Human Fetal Muscle. Stem cell reports 2:92–106. doi:10.1016/j.stemcr.2013.12.006
Collins CA, Olsen I, Zammit PS, Heslop L, Petrie A, Partridge TA, Morgan JE (2005) Stem cell function, self-renewal, and behavioral heterogeneity of cells from the adult muscle satellite cell niche. Cell 122:289–301. doi:10.1016/j.cell.2005.05.010
Crist Colin G, Montarras D, Buckingham M (2012) Muscle satellite cells are primed for myogenesis but maintain quiescence with sequestration of Myf5 mRNA targeted by microRNA-31 in mRNP granules. Cell stem cell 11:279. doi:10.1016/j.stem.2012.07.010
Danisovic L, Varga I, Polak S, Ulicna M, Bohmer D, Vojtassak J (2008) Morphology of in vitro expanded human muscle-derived stem cells, Biomedical papers of the Medical Faculty of the University Palacky, Olomouc 152:235–238
Das M, Rumsey JW, Bhargava N, Stancescu M, Hickman JJ (2009) Skeletal muscle tissue engineering: a maturation model promoting long-term survival of myotubes structural development of the excitation-contraction coupling apparatus and neonatal myosin heavy chain expression. Biomaterials 30:5392–5402. doi:10.1016/j.biomaterials.2009.05.081
Elliott NT, Yuan F (2011) A review of three-dimensional in vitro tissue models for drug discovery and transport studies. J Pharm Sci 100:59–74. doi:10.1002/jps.22257
Ghaemmaghami AM, Hancock MJ, Harrington H, Kaji H, Khademhosseini A (2012) Biomimetic tissues on a chip for drug discovery. Drug Discov Today 17:173–181. doi:10.1016/j.drudis.2011.10.029
Goetsch KP, Myburgh KH, Niesler CU (2013) In vitro myoblast motility models: investigating migration dynamics for the study of skeletal muscle repair. J Muscle Res Cell Motil 34:333–347. doi:10.1007/s10974-013-9364-7
Kelc R, Trapecar M, Vogrin M, Cencic A (2012) Skeletal muscle-derived cell cultures as potent models in regenerative medicine research. Muscle Nerve. doi:10.1002/mus.23688
Koning M, Werker PM, van Luyn MJ, Krenning G, Harmsen MC (2012) A global downregulation of microRNAs occurs in human quiescent satellite cells during myogenesis. Differentiation 84:314–321. doi:10.1016/j.diff.2012.08.002
Kuang S, Kuroda K, Le Grand F, Rudnicki MA (2007) Asymmetric self-renewal and commitment of satellite stem cells in muscle. Cell 129:999–1010. doi:10.1016/j.cell.2007.03.044
Li Y et al (2007) Decorin gene transfer promotes muscle cell differentiation and muscle regeneration. Mol Ther J Am Soc Gene Ther 15:1616–1622. doi:10.1038/sj.mt.6300250
Lindstrom M, Pedrosa-Domellof F, Thornell LE (2010) Satellite cell heterogeneity with respect to expression of MyoD, myogenin, Dlk1 and c-Met in human skeletal muscle: application to a cohort of power lifters and sedentary men. Histochem Cell Biol 134:371–385. doi:10.1007/s00418-010-0743-5
McKay BR, Toth KG, Tarnopolsky MA, Parise G (2010) Satellite cell number and cell cycle kinetics in response to acute myotrauma in humans: immunohistochemistry versus flow cytometry. J Physiol 588:3307–3320. doi:10.1113/jphysiol.2010.190876
Meng J, Adkin CF, Xu SW, Muntoni F, Morgan JE (2011) Contribution of human muscle-derived cells to skeletal muscle regeneration in dystrophic host mice. PloS One 6:e17454. doi:10.1371/journal.pone.0017454
Pawlikowski B, Lee L, Zuo J, Kramer RH (2009) Analysis of human muscle stem cells reveals a differentiation-resistant progenitor cell population expressing Pax7 capable of self-renewal. Dev Dyn Off Publ Am Assoc Anat 238:138–149. doi:10.1002/dvdy.21833
Peault B et al (2007) Stem and progenitor cells in skeletal muscle development, maintenance, and therapy. Mol Ther J Am Soc Gene Ther 15:867–877. doi:10.1038/mt.sj.6300145
Pirbalouti AG, Azizi S, Koohpayeh A, Hamedi B (2010) Wound healing activity of Malva sylvestris and Punica granatum in alloxan-induced diabetic rats. Acta Pol Pharm 67:511–516
Prudente AS et al (2013) Pre-clinical anti-inflammatory aspects of a cuisine and medicinal millennial herb: Malva sylvestris L. Food Chem Toxicol Int J publ Br Ind Biol Res Assoc 58:324–331. doi:10.1016/j.fct.2013.04.042
Sharifiaghdas F, Taheri M, Moghadasali R (2011) Isolation of human adult stem cells from muscle biopsy for future treatment of urinary incontinence. Urol J 8:54–59
Stewart JD et al (2003) Characterization of proliferating human skeletal muscle-derived cells in vitro: differential modulation of myoblast markers by TGF-beta2. J Cell Physiol 196:70–78. doi:10.1002/jcp.10322
Sun Y, Ge Y, Drnevich J, Zhao Y, Band M, Chen J (2010) Mammalian target of rapamycin regulates miRNA-1 and follistatin in skeletal myogenesis. J Cell Biol 189:1157–1169. doi:10.1083/jcb.200912093
Tedesco FS, Dellavalle A, Diaz-Manera J, Messina G, Cossu G (2010) Repairing skeletal muscle: regenerative potential of skeletal muscle stem cells. J Clin Investig 120:11–19. doi:10.1172/JCI40373
Tripathi AK, Ramani UV, Rank DN, Joshi CG (2011) In vitro expression profiling of myostatin, follistatin, decorin and muscle-specific transcription factors in adult caprine contractile myotubes. J Muscle Res Cell Motil 32:23–30. doi:10.1007/s10974-011-9245-x
Veliça P, Bunce CM (2011) A quick, simple and unbiased method to quantify C2C12 myogenic differentiation. Muscle Nerve 44:366–370. doi:10.1002/mus.22056
Wagers AJ, Conboy IM (2005) Cellular and molecular signatures of muscle regeneration: current concepts and controversies in adult myogenesis. Cell 122:659–667. doi:10.1016/j.cell.2005.08.021
Wei Y, Li Y, Chen C, Stoelzel K, Kaufmann AM, Albers AE (2011) Human skeletal muscle-derived stem cells retain stem cell properties after expansion in myosphere culture. Exp Cell Res 317:1016–1027. doi:10.1016/j.yexcr.2011.01.019
Yaffe D, Saxel ORA (1977) Serial passaging and differentiation of myogenic cells isolated from dystrophic mouse muscle. Nature 270:725–727
Zammit P, Beauchamp J (2001) The skeletal muscle satellite cell: stem cell or son of stem cell? Differentiation 68:193–204 [Research in biological diversity]
Zammit PS, Partridge TA, Yablonka-Reuveni Z (2006) The skeletal muscle satellite cell: the stem cell that came in from the cold. J Histochem Cytochem Off J Histochem Soc 54:1177–1191. doi:10.1369/jhc.6R6995.2006
Zhao P, Hoffman E (2008) Transcriptional Cascades in Muscle Regeneration. In: Skeletal Muscle Repair and Regeneration, vol 3. Advances in Muscle Research. Springer Netherlands, pp 85–106. doi:10.1007/978-1-4020-6768-6_5
Acknowledgments
The authors would like to dedicate this manuscript to Prof. Dr. Avrelija Cencic, without whom this work would not be possible. We also acknowledge Rudi Mlakar for his technical assistance with confocal microscopy. Special thanks go to Nicholson William Price II for his revision and constructive input. This work was supported by the Slovenian Research Agency Program P3-0310-2334 and Grant J3-2290-2334.
Conflict of interest
The authors declare that they have no competing interests.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Trapecar, M., Kelc, R., Gradisnik, L. et al. Myogenic progenitors and imaging single-cell flow analysis: a model to study commitment of adult muscle stem cells. J Muscle Res Cell Motil 35, 249–257 (2014). https://doi.org/10.1007/s10974-014-9398-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10974-014-9398-5