Abstract
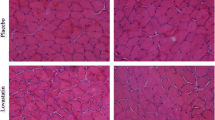
Although statins, the most widely used drugs in the treatment of hyperlipidaemia, are generally accepted as efficient and safe drugs their side-effects on skeletal muscle have been reported with increasing frequency. The lack of an animal model in which these side effects would consistently be observed is one of the important drawbacks in studying statin associated myopathy. To overcome this and enable the studying of the effects of fluvastatin on skeletal muscles an animal model with high blood cholesterol levels was developed. In these animals cholesterol levels rose more than seven fold (from 1.5 ± 0.1 to 10.7 ± 2.0 mmol/l; n = 15 and 16) with a dramatic increase in low density lipoprotein/high density lipoprotein ratio (from 0.29 ± 0.02 to 1.56 ± 0.17). While the latter was reversed by statin treatment, an elevation in blood creatine kinase (CK) level indicated the presence of muscle wasting. Fibers from m. extensor digitorum longus (EDL) showed significant reduction in cross sectional area in the statin treated groups. Statin treatment also decreased the proliferation and fusion of skeletal myotubes in culture. In line with this, resting intracellular calcium concentration ([Ca2+]i) was reduced in statin treated satellite cells and myotubes. On the other hand, in adult skeletal muscle fibers statin treatment increased resting [Ca2+]i (116 ± 4 nM vs. 151 ± 5 nM; n = 33 and 34) and decreased both twitch and tetanic force both in EDL and m. soleus. In addition, in m. soleus the duration of twitch and tetanic force was shortened. These results clearly indicate that statin administration in these animals results in a myopathy characterized by decreased muscle force and elevated plasma CK level.








Similar content being viewed by others
References
Bar SL, Holmes DT, Frohlich J (2007) Asymptomatic hypothroidism and statin-induced myopathy. Can Fam Physician 53(3):428–431
Bruckert E, Hayem G, Dejager S, Yau C, Bégaud B (2005) Mild to moderate muscular symptoms with high-dosage statin therapy in hyperlipidemic patients—the PRIMO study. Cardiovasc Drugs Ther 19(6):403–414
Buettner C, Lecker SH (2008) Molecular basis for statin-induced muscle toxicity: implications and possibilities. Pharmacogenomics 9(8):1133–1142
Cseri J, Szappanos H, Szigeti GP, Csernátony Z, Kovács L, Csernoch L (2002) A purinergic signal transduction pathway in mammalian skeletal muscle cells in culture. Pflügers Archiv 443:731–738
Dirks AJ, Jones KM (2006) Statin-induced apoptosis and skeletal myopathy. Am J Physiol Cell Physiol 291(6):C1208–C1212
Draeger A, Monastyrskaya K, Mohaupt M, Hoppeler H, Savolainen H, Allemann C, Babiychuk EB (2006) Statin therapy induces ultrastructural damage in skeletal muscle in patients without myalgia. J Pathol 210(1):94–102
Draeger A, Sanchez-Freire V, Monastyrskaya K, Hoppeler H, Mueller M, Breil F, Mohaupt MG, Babiychuk EB (2010) Statin therapy and the expression of genes that regulate calcium homeostasis and membrane repair in skeletal muscle. Am J Pathol 177(1):291–299
Dreier JP, Endres M (2004) Statin-associated rhabdomyolysis triggered by grapefruit consumption. Neurology 62(4):670
Fridewald WF, Levy RI, Frederickson DS (1972) Estimation of LDL-cholesterol concentration without use of the preparative ultra-centrifuge. Clin Chem 18:499–502
Ghirlanda G, Oradei A, Manto A, Lippa S, Uccioli L, Caputo S, Greco AV, Littarru GP (1993) Evidence of plasma CoQ10-lowering effect by HMG-CoA reductase inhibitors: a double-blind, placebo-controlled study. J Clin Pharmacol 33:226–229
Haines TH (2001) Do sterols reduce proton and sodium leaks through lipid bilayers? Prog Lipid Res 40:299–324
Harper CR, Jacobson TA (2007) The broad spectrum of statin myopathy: from myalgia to rhabdomyolysis. Curr Opin Lipidol 18(4):401–408
Kaufmann P, Török M, Zahno A, Waldhauser KM, Brecht K, Krähenbühl S (2006) Toxicity of statins on rat skeletal muscle mitochondria. Cell Mol Life Sci 63(19–20):2415–2425
Kohro T, Yamazaki T (2009) Mechanism of statin-induced myopathy investigated using microarray technology. J Atheroscler Thromb 16(1):30–32
Krause BR, Newton RS (1995) Lipid-lowering activity of atorvastatin and lovastatin in rodent species: triglyceride-lowering in rats correlates with efficacy in LDL animal models. Atherosclerosis 117(2):237–244
Legrand C, Giacomello E, Berthier C, Allard B, Sorrentino V, Jacquemond V (2008) Spontaneous and voltage-activated Ca2+ release in adult mouse skeletal muscle fibres expressing the type 3 ryanodine receptor. J Physiol 586(2):441–457
Marcoff L, Thompson PD (2007) The role of coenzyme Q10 in statin-associated myopathy: a systematic review. J Am Coll Cardiol 49:2231–2237. doi:10.1016/j.jacc.2007.02.049
Merz CNB, Alberts MJ, Balady GJ, Ballantyne CM et al (2009) ACCF/AHA/ACP 2009 competence and training statement: a curriculum on prevention of cardiovascular disease: a report of the American College of Cardiology Foundation/American Heart Association/American College of Physicians Task Force on Competence and Training (Writing Committee to Develop a Competence and Training Statement on Prevention of Cardiovascular Disease). Circulation 120:e100–e126
Obata T, Yonemoti H, Aomine M (2009) The protective effect of fluvastatin on hydroxyl radical generation by inhibiting low-density lipoprotein (LDL) oxidation in the rat myocardium. Microvasc Res 77(2):163–165
Oddoux S, Brocard J, Schweitzer A, Szentesi P, Giannesini B, Brocard J, Fauré J, Pernet-Gallay K, Bendahan D, Lunardi J, Csernoch L, Marty I (2009) Triadin deletion induces impaired skeletal muscle function. J Biol Chem 284(50):34918–34929
Pados Gy, Audikovszky M (2007) A statinkezelés biztonságossága. Hyppocrates 9(1):20–25. ISSN 1419-3337
Ranatunga KW (1982) Temperature-dependence of shortening velocity and rate of isometric tension development in rat skeletal muscle. J Physiol 329:465–483
Sacher J, Weigl L, Werner M, Szegedi C, Hohenegger M (2005) Delineation of myotoxicity induced by 3-hydroxy-3-methylglutaryl CoA reductase inhibitors in human skeletal muscle cells. J Pharmacol Exp Ther 314(3):1032–1041
Sirvent P, Mercier J, Vassort G, Lacampagne A (2005) Simvastatin triggers mitochondria-induced Ca2+ signaling alteration in skeletal muscle. Biochem Biophys Res Commun 329(3):1067–1075
Sirvent P, Mercier J, Lacampagne A (2008) New insights into mechanisms of statin-associated myotoxicity. Curr Opin Pharmacol 8(3):333–338
Suckow MA, Weisbroth SH, Franklin CL (2006) The Laboratory Rat. Elsevier Inc., San Diego. ISBN: 978-0-12-074903-4
Szappanos H, Cseri J, Deli T, Kovács L, Csernoch L (2004) Determination of depolarisation- and agonist-evoked calcium fluxes on skeletal muscle cells in primary culture. J Biochem Biophys Methods 59(1):89–101
Thomas L (1992) Labor und diagnose, 4th edn. Die Medizinische Verlagsgesellschaft, Marburg
Thompson PD, Clarkson PM, Rosenson RS, National Lipid Association Statin Safety Task Force Muscle Safety Expert Panel (2006) An assessment of statin safety by muscle experts. Am J Cardiol 97(8A):69C–76C
Wang CY, Liu PY, Liao JK (2008) Pleiotropic effects of statin therapy: molecular mechanisms and clinical results. Trends Mol Med 14(1):37–44
Ward S, Lloyd Jones M, Pandor A, Holmes M, Ara R, Ryan A, Yeo W, Payne M (2007) A systematic review and economic evaluation of statins for the prevention of coronary events. Health Technol Assess 11(14):1–160, iii–iv
Yerroum M, Braconnier F, Chariot P (1999) Influence of handling procedures on rat plasma creatine kinase activity. Muscle Nerve 22(8):1119–1121
Acknowledgments
The authors wish to thank R. Öri for the excellent technical assistance. This work was supported by grants from the Hungarian Scientific Research Found (OTKA K-75604, NK-78398), from the Hungarian Ministry of Health (ETT 186/2009) and from the Hungarian Ministry of Education (K-2009-TÁMOP-4.2.2-08/1/2008-0019).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Füzi, M., Palicz, Z., Vincze, J. et al. Fluvastatin-induced alterations of skeletal muscle function in hypercholesterolaemic rats. J Muscle Res Cell Motil 32, 391–401 (2012). https://doi.org/10.1007/s10974-011-9272-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10974-011-9272-7