Abstract
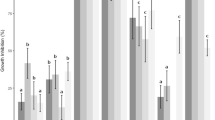
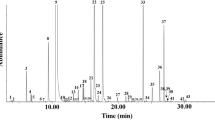
The antifungal activity of essential oil (EO) from the Brazilian epazote (Chenopodium ambrosioides L.) was evaluated by the poison food assay at concentrations of 0.3%, 0.1%, and 0.05% with eight postharvest deteriorating fungi (Aspergillus flavus, Aspergillus glaucus, Aspergillus niger, Aspergillus ochraceous, Colletotrichum gloesporioides, Colletotrichum musae, Fusarium oxysporum, and Fusarium semitectum). EO components were tentatively identified by Kováts retention indices (RIs) using gas chromatography and gas chromatography combined with mass spectrometry (GC-MS). Growth of all fungi was completely inhibited at 0.3% concentration, and by 90% to 100% at 0.1% concentration. The following 13 tentatively identified compounds (relative percent) accounted for 90.4% of the total volatile oil: α-terpinene (0.9), p-cymene (2.0), benzyl alcohol (0.3), p-cresol (0.3), p-mentha-1,3,8-triene (0.2), p-cimen-8-ol (0.6), α-terpineol (0.5), (Z)-ascaridole (61.4), piperitone (0.9), carvacrol (3.9), (E)-ascaridole (18.6), (E)-piperitol acetate (0.5), and (Z)-carvyl acetate (0.3). Autobiographic thin layer chromatography of the EO to separate the principal fungitoxic fraction yielded only one fraction that completely inhibited the growth of all test fungi at a concentration of 0.1%. This fraction was characterized by RIs and GC-MS presenting a composition (%) of p-cymene (25.4), (Z)-ascaridole (44.4), and (E)-ascaridole (30.2). The results suggest ascaridoles were the principal fungitoxic components of the EO.


Similar content being viewed by others
References
Adams, R. P. 1995. Identification of essential oil components by gas chromatography/mass spectroscopy. Allured Publishing Corporation, Carol Stream.
Arispuro, I. V., Bernal, S. A., Tellez, M. A. M., and Nieblas, M. O. 1997. Effect of plant extracts on the growth and aflatoxin production of Aspergillus flavus and Aspergillus parasiticus. Rev. Mex. Fitopatol. 15:91–95.
Atanda, O. O., Akpan, I., and Oluwafemi, F. 2007. The potential of some spice essential oils in the control of A. parasiticus CFR 223 and aflatoxin production. Food Control 18:601–607.
Bauer, L., and Brasil, S. G. A. 1973. Essential oils of Chenopodium ambrosioides and Schinus terebinthifolius from Rio Grande do Sul. Rev. Bras. Farm. 54:240–242.
Boiteau, P. 1986. Médecine Traditionnelle et Pharmacopée: Précis de Matière Médicale Malgache. Agence de Coopération Culturelle et Technique, Paris.
Castaneda, G. R., Tellez, M. A. M., Cohen, S. V., Manilla, G. A., Arispuro, I. C. V., Sanz, P. M., and Yufera, E. P. 2001. In vitro inhibition of mycelial growth of Tilletia indica by extracts of native plants from Sonora, Mexico. Rev. Mex. Fitopatol. 19:214–217.
Cavalli, J. F., Tomi, F., Bernardini, A. F., and Casanova, J. 2004. Combined analysis of the essential oil of Chenopodium ambrosioides by GC, GC-MS and 13C-NMR spectroscopy: quantitative determination of ascaridole, a heat-sensitive compound. Phytochem. Anal. 15:275–279.
Chiasson, H., Bostanian, N. J., and Vincent, C. 2004a. Acaricidal properties of a Chenopodium-based botanical. J. Econ. Entomol. 97:1373–1377.
Chiasson, H., Vincent, C., and Bostanian, N. J. 2004b. Insecticidal properties of a Chenopodium-based botanical. J. Econ. Entomol. 97:1378–1383.
Dhingra, O. D., and Sinclair, J. B. 1995. Basic methods in plant pathology. CRC, Boca Raton.
Dubey, N. K., Kishore, N., Srivastava, O. P., Dikshit, A., and Singh, S. K. 1983. Fungitoxicity of some higher plants against Rhizoctonia solani. Plant Soil 72:91–94.
Duke, S. O., Baerson, S. R., Dayan, F. E., Rimando, A. M., Scheffler, B. E., Tellez, M. R., Wedge, D. E., Schrader, K. K., Akey, D. H., Arthur, F. H., De Lucca, A. J., Gibson, D. M., Harrison Jr, H. F., Peterson, J. K., Gealy, D. R., Tworkoski, T., Wilson, C. L., and Brad Morris, J. 2003. United States Department of Agriculture-Agricultural Research Service research on natural products for pest management. Pest Manag. Sci. 59:708–717.
Ferhout, H., Bohatier, J., Guillot, J., and Chalchat, J. C. 1999. Antifungal activity of selected essential oils, cinnamaldehyde and carvacrol against Malassezia furfur and Candida albicans. J. Essent. Oil Res. 11:119–129.
Guenther, E. 1952. The essential oils, vol VI. Van Nostrand, New York.
Gupta, D., Charles, R., Mehta, V. K., Garg, S. N., and Kumar, S. 2002. Chemical examination of the essential oil of Chenopodium ambrosioides L. from the southern hills of India. J. Essent. Oil Res. 14:93–94.
Huang, X. F., and Kong, L. Y. 2002. Study on chemical constituents of volatile oil from Chenopodium ambrosioides L. J. Chin. Pharm. Univ. 33:256–257.
Jesus Faria, T., Ferreira, R. S., Yassumoto, L., Souza, J. R. P., Ishikawa, N. K., and Melo Barbosa, A. 2006. Antifungal activity of essential oil isolated from Ocimum gratissimum L. (eugenol chemotype) against phytopathogenic fungi. Braz. Arch. Biol. Technol. 49:867–871.
Kasali, A. A., Ekundayo, O., Paul, C., Konig, W. A., Eshilokun, A. O., and Ige, B. 2006. 1,2,3,4-Diepoxy-p-menthane and 1,4-epoxy-p-menth-2-ene: Rare monoterpenoids from the essential oil of Chenopodium ambrosioides L. var ambrosioides leaves. J. Essent. Oil Res. 18:13–15.
Kishore, N., Chansouria, J. P. N., and Dubey, N. K. 1996. Antidermatophytic action of the essential oil of Chenopodium ambrosioides and an ointment prepared from it. Phytother. Res. 10:453–455.
Lall, N., and Meyer, J. J. M. 1999. In vitro inhibition of drug-resistant and drug-sensitive strains of Mycobacterium tuberculosis by ethnobotanically selected South African plants. J. Ethnopharmacol. 66:347–354.
Malik, M. M., and Mujtaba-Naqvi, S. H. 1984. Screening of some indigenous plants as repellants or antifeedants for stored grain insects. J. Stored Prod. Res. 20:41–44.
Mari, M., and Guizzardi, M. 1998. The postharvest phase: Emerging technologies for the control of fungal diseases. Phytoparasitica 26:59–66.
Muhayimana Uhayimana, A., Chalchat, J. C., and Garry, R. P. 1998. Chemical composition of essential oils of Chenopodium ambrosioides L. from Rwanda. J. Essent. Oil Res. 10:690–692.
Noumi, E., and Yomi, A. 2001. Medicinal plants used for intestinal diseases in Mbalmayo Region, Central Province, Cameroon. Fitoterapia 72:246–254.
Onocha, P. A., Ekundayo, O., Eramo, T., and Laakso, I. 1999. Essential oil constituents of Chenopodium ambrosioides L. leaves from Nigeria. J. Essent. Oil Res. 11:220–222.
Pino, J. A., Marbot, R., and Real, I. M. 2003. Essential Oil of Chenopodium ambrosioides L. from Cuba. J. Essent. Oil Res. 15:213–214.
Pollack, Y., Segal, R., and Golenser, J. 1990. The effect of ascaridole on the in vitro development of Plasmodium falciparum. Parasitol. Res. 76:570–572.
Pradeep, A. G., Lokesh, S., and Rai, V. R. 2003. Efficacy of some essential oils on seed mycoflora and seedling quality of some crop species saved by farmers. Adv. Plant Sci. 16:53–58.
Rahalison, L., Hamburger, M., Hostettmann, K., Monod, M., and Frenk, E. A. 1991. Bioautographic agar overlay method for the detection of antifungal compounds from higher plants. Phytochem. Anal. 2:199–203.
Sabulal, B., Dan, M., Pradeep, N. S., Valsamma, R. K., and George, V. 2006. Composition and antimicrobial activity of essential oil from the fruits of Amomum cannicarpum. Acta. Pharm. 56:473–480.
Sridhar, S. R., Rajagopal, R. V., Rajavel, R., Masilamani, S., and Narasimhan, S. 2003. Antifungal activity of some essential oils. J. Agric. Food Chem. 51:7596–7599.
Su, H. C. F. 1991. Toxicity and repellency of Chenopodium oil to four species of stored-product insects. J. Entomol. Sci. 26:178–182.
Szczerbanik, M., Jobling, J., Morris, S., and Holford, P. 2007. Essential oil vapours control some common postharvest fungal pathogens. Aust. J. Exp. Agric. 47:103–109.
Tapondjou, L. A., Adler, C., Bouda, H., and Fontem, D. A. 2002. Efficacy of powder and essential oil from Chenopodium ambrosioides leaves as post-harvest grain protectants against six-stored product beetles. J. Stored Prod. Res. 38:395–402.
Tripathi, P., and Dubey, N. K. 2004. Exploitation of natural products as an alternative strategy to control postharvest fungal rotting of fruit and vegetables. Postharvest Biol. Technol. 32:235–245.
Wink, M. 1993. Production and application of phytochemicals from agricultural perspective, pp. 171–213, in T. A. Van Beek, and H. Breteler (eds.). Phytochemistry and Agriculture, Vol. 34. Clarendon, Oxford.
Yang, J. K., Choi, M. S., Seo, W. T., Rinker, D. L., Han, S. W., and Cheong, G. W. 2007. Chemical composition and antimicrobial activity of Chamaecyparis obtusa leaf essential oil. Fitoterapia 78:149–152.
Acknowledgements
We thank CAPES (Coordenação de Aperfeiçoamento de Pessoal de Nível Superior) and CNPq (Conselho Nacional de Pesquisa) for granting scholarships to the authors and to the technician Mr. Eduardo Pereira Rezende. This work was supported in part from grants from FAPEMIG (Fundação de Ampara de Pesquisa do Estado de Minas Gerais).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jardim, C.M., Jham, G.N., Dhingra, O.D. et al. Composition and Antifungal Activity of the Essential Oil of the Brazilian Chenopodium ambrosioides L.. J Chem Ecol 34, 1213–1218 (2008). https://doi.org/10.1007/s10886-008-9526-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10886-008-9526-z