Abstract
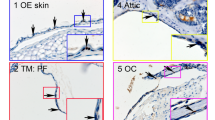
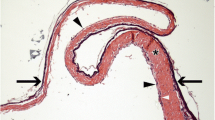
The human tympanic membrane (hTM), known more commonly as the eardrum, is a thin, multi-layered membrane that is unique in the body as it is suspended in air. When perforated, the hTM’s primary function of sound-pressure transmission is compromised. For the purposes of TM reconstruction, we investigated the phenotype and genotype of cultured primary cells derived from hTM tissue explants, compared to epithelial (HaCaT cells) and mesenchymal (human dermal fibroblasts (HDF)) reference cells. Epithelium-specific ets-1 (ESE-1), E-cadherin, keratinocyte growth factor-1 (KGF-1/FGF-7), keratinocyte growth factor-2 (KGF-2/FGF10), fibroblast growth factor receptor 1 (FGFR1), variants of fibroblast growth factor receptor 2 (FGFR2), fibroblast surface protein (FSP), and vimentin proteins were used to assess the phenotypes of all cultured cells. Wholemount and paraffin-embedded hTM tissues were stained with ESE-1 and E-cadherin proteins to establish normal epithelial-specific expression patterns within the epithelial layers. Immunofluorescent (IF) cell staining of hTM epithelial cells (hTMk) demonstrated co-expression of both epithelial- and mesenchymal-specific proteins. Flow cytometry (FCM) analysis further demonstrated co-expression of these epithelial and mesenchymal-specific proteins, indicating the subcultured hTMk cells possessed a transitional phenotype. Gene transcript analysis of hTMk cells by reverse transcriptase polymerase chain reaction (RT-PCR) revealed a down regulation of ESE-1, E-cadherin, FGFR2, variant 1 and variant 2 (FGFR2v1 and FGFR2v2) between low and high passages, and up-regulation of KGF-1, KGF-2, and FGFR1. All results indicate a gradual shift in cell phenotype of hTMk-derived cells from epithelial to mesenchymal.




Similar content being viewed by others
References
Atala A (2006) Tissue-engineered autologous bladders for patients needing cystoplasty. Lancet 367:1241–1246
Boukamp P, Petrussevska R, Breitkreutz D, Hornung J, Markham A, Fusenig N (1988) Normal keratinization in a spontaneously immortalized aneuploid human keratinocyte cell line. J Cell Biol 106:761–771
Broekaert D (1995) The tympanic membrane: a biochemical updating of structural components. Acta Otorhinolaryngol Belg 49:127–137
Finch P, Rubin J, Miki T, Ron D, Aaronson S (1989) Human KGF is FGF-related with properties of a paracrine effector of epithelial cell growth. Science 245:752–755
Ghassemifar R, Redmond SL, Zainuddin, Chirila TV (2010) Advancing towards a tissue-engineered tympanic membrane: silk fibroin as a substratum for growing human eardrum keratinocytes. J Biomater Appl 24:591–606
Hay ED (2005) The mesenchymal cell, its role in the embryo, and the remarkable signaling mechanisms that create it. Dev Dyn 233:706–720
Karasek MA (1975) In vitro growth and maturation of epithelial cells from postembryonic skin. J Invest Dermatol 65:60–66
Lee J, Dedhar S, Kalluri R, Thompson E (2006) The epithelial-mesenchymal transition: new insights in signaling, development, and disease. J Cell Biol 172:973–981
Levin B, Rajkhowa R, Redmond SL, Atlas MD (2009) Grafts in myringoplasty: utilizing a silk fibroin scaffold as a novel device. Exp Rev Med Dev 6:653–664
Levin B, Redmond SL, Rajkhowa R, Eikelboom RH, Marano RJ, Atlas MD (2010) Preliminary results of the application of a silk fibroin scaffold to otology. Otolaryngol Head Neck Surg 142:S33–S35
Lim D (1995) Structure and function of the tympanic membrane: a review. Acta Otorhinolaryngol Belg 49:101–115
Parrott JA, Kim G, Mosher R, Skinner MK (2000) Expression and action of keratinocyte growth factor (KGF) in normal ovarian surface epithelium and ovarian cancer. Mol Cell Endocrinol 167:77–87
Pereira CT, Herndon DN, Rocker R, Jeschke MG (2007) LIposomal gene transfer of keratinocyte growth factor improves wound healing by altering growth factor and collagen expression. J Surg Res 139:222–228
Radisky DC (2005) Epithelial-mesenchymal transition. J Cell Sci 118:4325–4326
Ronnov-Jessen L, Celis J, Van Deurs B, Petersen O (1992) A fibroblast-associated antigen: characterization in fibroblasts and immunoreactivity in smooth muscle differentiated stromal cells. J Histochem Cytochem 40:475–486
Rubin J (1989) Purification and characterization of a newly identified growth factor specific for epithelial cells. Proc Natl Acad Sci USA 86:802
Santa Maria PL, Redmond SL, Atlas MD, Ghassemifar R (2010a) Histology of the healing tympanic membrane following perforation in rats. Laryngoscope 120:2061–2070
Santa Maria PL, Redmond SL, Atlas MD and Ghassemifar R (2010b) The role of epidermal growth factor in the healing tympanic membrane following perforation in rats. J Mol Histol (in press)
Shook D (2003) Mechanisms, mechanics and function of epithelial-mesenchymal transitions in early development. Mech Dev 120:1351–1383
Stenfeldt K, Johansson C, Hellstrom S (2006) The collagen structure of the tympanic membrane: collagen types I, II, and III in the healthy tympanic membrane, during healing of a perforation, and during infection. Arch Otolaryngol Head Neck Surg 132:293–298
Stenfors L (1984) The tympanic membrane. Acta Otolaryngol 98:28
Taylor M, McMinn R (1967) Cytology of repair in experimental perforation of the tympanic membrane and its relationship to chronic perforations in man. Trans Am Acad Ophthalmol Otolaryngol 71:802–812
Thiery J (2003) Epithelial-mesenchymal transitions in development and pathologies. Curr Opin Cell Biol 15:740–746
Thiery J, Sleeman J (2006) Complex networks orchestrate epithelial-mesenchymal transitions. Nat Rev Mol Cell Biol 7:131–142
Werner S (1998) Keratinocyte growth factor: a unique player in epithelial repair processes. Cytokine Growth Factor Rev 9:153–165
Werner S, Peters K, Longaker M, Fuller-Pace F, Banda M, Williams L (1992) Large induction of keratinocyte growth factor expression in the dermis during wound healing. Proc Natl Acad Sci USA 89:6896–6900
Werner S, Krieg T, Smola H (2007) Keratinocyte-fibroblast interactions in wound healing. J Invest Dermatol 127:998–1008
Acknowledgments
This study has full human ethics approval from the St John of God Health Care Ethics Committee (St John of God Hospital, Subiaco, Western Australia). The Garnett Passe and Rodney Williams Memorial Foundation, and the Medical and Health Research Infrastructure Fund, Western Australian Department of Health, funded this study. We gratefully acknowledge Winthrop Professor Fiona Wood (Burn Injury Research Unit, UWA), for supplying the human HDF cells and Winthrop Professor Marcus Atlas for the supply of valuable hTM whole tissue samples for this study. Adjunct Professor Robert Eikelboom and Dr Bing Teh are thanked for valuable comments relating to the manuscript. The authors acknowledge the facilities, scientific and technical assistance of the Australian Microscopy & Microanalysis Research Facility at the Centre for Microscopy, Characterisation & Analysis, The University of Western Australia, a facility funded by The University, State, and Commonwealth Governments.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Redmond, S.L., Levin, B., Heel, K.A. et al. Phenotypic and genotypic profile of human tympanic membrane derived cultured cells. J Mol Hist 42, 15–25 (2011). https://doi.org/10.1007/s10735-010-9303-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10735-010-9303-5