Abstract
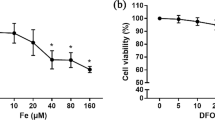
The association between iron overload and osteoporosis has been found in many diseases, such as hemochromatosis, β-thalassemia and sickle cell anemia with multiple blood transfusion. One of the contributing factors is iron toxicity to osteoblasts. Some studies showed the negative effects of iron on osteoblasts; however, the effects of two biological available iron species, i.e., ferric and ferrous, on osteoblasts are elusive. Since most intracellular ionized iron is ferric, osteoblasts was hypothesized to be more responsive to ferric iron. Herein, ferric ammonium citrate (FAC) and ferrous ammonium sulfate (FAS) were used as ferric and ferrous donors. Our results showed that both iron species suppressed cell survival and proliferation. Both also induced osteoblast cell death consistent with the higher levels of cleaved caspase 3 and caspase 7 in osteoblasts, indicating that iron induced osteoblast apoptosis. Iron treatments led to the elevated intracellular iron in osteoblasts as determined by atomic absorption spectrophotometry, thereby leading to a decreased expression of genes for cellular iron import and increased expression of genes for cellular iron export. Effects of FAC and FAS on osteoblast differentiation were determined by the activity of alkaline phosphatase (ALP). The lower ALP activity from osteoblast with iron exposure was found. In addition, ferric and ferrous differentially induced osteoblastic and osteoblast-derived osteoclastogenic gene expression alterations in osteoblast. Even though both iron species had similar effects on osteoblast cell survival and differentiation, the overall effects were markedly stronger in FAC-treated groups, suggesting that osteoblasts were more sensitive to ferric than ferrous.







Similar content being viewed by others
References
Balogh E, Tolnai E, Nagy B Jr, Nagy B, Balla G, Balla J, Jeney V (2016) Iron overload inhibits osteogenic commitment and differentiation of mesenchymal stem cells via the induction of ferritin. Biochim Biophys Acta 1862:1640–1649. https://doi.org/10.1016/j.bbadis.2016.06.003
Boyle WJ, Simonet WS, Lacey DL (2003) Osteoclast differentiation and activation. Nature 423:337–342. https://doi.org/10.1038/nature01658
Bregstein J, Roskind CG, Sonnett FM (2011) CHAPTER 5—emergency medicine A2—Polin, Richard A. In: Ditmar MF (ed) Pediatric secrets, 5th edn. Mosby, Philadelphia, pp 154–196. https://doi.org/10.1016/B978-0-323-06561-0.00005-7
Bressenot A, Marchal S, Bezdetnaya L, Garrier J, Guillemin F, Plenat F (2009) Assessment of apoptosis by immunohistochemistry to active caspase-3, active caspase-7, or cleaved PARP in monolayer cells and spheroid and subcutaneous xenografts of human carcinoma. J Histochem Cytochem 57:289–300. https://doi.org/10.1369/jhc.2008.952044
Cappellen D, Luong-Nguyen NH, Bongiovanni S, Grenet O, Wanke C, Susa M (2002) Transcriptional program of mouse osteoclast differentiation governed by the macrophage colony-stimulating factor and the ligand for the receptor activator of NFkappa B. J Biol Chem 277:21971–21982. https://doi.org/10.1074/jbc.M200434200
Chen B, Li GF, Shen Y, Huang XI, Xu YJ (2015) Reducing iron accumulation: a potential approach for the prevention and treatment of postmenopausal osteoporosis. Exp Ther Med 10:7–11. https://doi.org/10.3892/etm.2015.2484
de Vernejoul MC, Pointillart A, Golenzer CC, Morieux C, Bielakoff J, Modrowski D, Miravet L (1984) Effects of iron overload on bone remodeling in pigs. Am J Pathol 116:377–384
Diamond T, Pojer R, Stiel D, Alfrey A, Posen S (1991) Does iron affect osteoblast function? Studies in vitro and in patients with chronic liver disease. Calcif Tissue Int 48:373–379
Djordjevic VB (2004) Free radicals in cell biology. Int Rev Cytol 237:57–89. https://doi.org/10.1016/S0074-7696(04)37002-6
Ganz T (2007) Molecular control of iron transport. J Am Soc Nephrol: JASN 18:394–400. https://doi.org/10.1681/ASN.2006070802
Guo JP, Pan JX, Xiong L, Xia WF, Cui S, Xiong WC (2015) Iron chelation inhibits osteoclastic differentiation in vitro and in Tg2576 mouse model of Alzheimer’s disease. PLoS ONE 10:e0139395. https://doi.org/10.1371/journal.pone.0139395
He YF et al (2013) Iron overload inhibits osteoblast biological activity through oxidative stress. Biol Trace Elem Res 152:292–296. https://doi.org/10.1007/s12011-013-9605-z
Jeney V (2017) Clinical impact and cellular mechanisms of iron overload-associated bone loss. Front Pharmacol 8:77. https://doi.org/10.3389/fphar.2017.00077
Ke JY, Cen WJ, Zhou XZ, Li YR, Kong WD, Jiang JW (2017) Iron overload induces apoptosis of murine preosteoblast cells via ROS and inhibition of AKT pathway. Oral Dis 23:784–794. https://doi.org/10.1111/odi.12662
Lee FP, Jen CY, Chang CC, Chou Y, Lin H, Chou CM, Juan SH (2010) Mechanisms of adiponectin-mediated COX-2 induction and protection against iron injury in mouse hepatocytes. J Cell Physiol 224:837–847. https://doi.org/10.1002/jcp.22192
Liu F, Zhang WL, Meng HZ, Cai ZY, Yang MW (2017) Regulation of DMT1 on autophagy and apoptosis in osteoblast. Int J Med Sci 14:275–283. https://doi.org/10.7150/ijms.17860
Lukiw WJ, Bazan NG (1998) Strong nuclear factor-kappaB-DNA binding parallels cyclooxygenase-2 gene transcription in aging and in sporadic Alzheimer’s disease superior temporal lobe neocortex. J Neurosci Res 53:583–592. https://doi.org/10.1002/(SICI)1097-4547(19980901)53:5<583::AID-JNR8>3.0.CO;2-5
MacKenzie EL, Iwasaki K, Tsuji Y (2008) Intracellular iron transport and storage: from molecular mechanisms to health implications. Antioxid Redox Signal 10:997–1030. https://doi.org/10.1089/ars.2007.1893
Matsuo K, Irie N (2008) Osteoclast-osteoblast communication. Arch Biochem Biophys 473:201–209. https://doi.org/10.1016/j.abb.2008.03.027
McKie AT et al (2001) An iron-regulated ferric reductase associated with the absorption of dietary iron. Science 291:1755–1759. https://doi.org/10.1126/science.1057206
Messer JG, Kilbarger AK, Erikson KM, Kipp DE (2009) Iron overload alters iron-regulatory genes and proteins, down-regulates osteoblastic phenotype, and is associated with apoptosis in fetal rat calvaria cultures. Bone 45:972–979. https://doi.org/10.1016/j.bone.2009.07.073
Noble JE, Bailey MJ (2009) Quantitation of protein. Methods Enzymol 463:73–95. https://doi.org/10.1016/S0076-6879(09)63008-1
Proff P, Romer P (2009) The molecular mechanism behind bone remodelling: a review. Clin Oral Investig 13:355–362. https://doi.org/10.1007/s00784-009-0268-2
Raggatt LJ, Partridge NC (2010) Cellular and molecular mechanisms of bone remodeling. J Biol Chem 285:25103–25108. https://doi.org/10.1074/jbc.R109.041087
Salgado P, Melin V, Contreras D, Moreno Y, Mansilla HD (2013) Fenton reaction driven by iron ligands. J Chil Chem Soc 58:2096–2101. https://doi.org/10.4067/S0717-97072013000400043
Salvador GA, Uranga RM, Giusto NM (2010) Iron and mechanisms of neurotoxicity. Int J Alzheimers Dis 2011:720658. https://doi.org/10.4061/2011/720658
Tian Q, Wu S, Dai Z, Yang J, Zheng J, Zheng Q, Liu Y (2016) Iron overload induced death of osteoblasts in vitro: involvement of the mitochondrial apoptotic pathway. PeerJ 4:e2611. https://doi.org/10.7717/peerj.2611
Tsay J et al (2010) Bone loss caused by iron overload in a murine model: importance of oxidative stress. Blood 116:2582–2589. https://doi.org/10.1182/blood-2009-12-260083
Winterbourn CC (1995) Toxicity of iron and hydrogen peroxide: the Fenton reaction. Toxicol Lett 82–83:969–974
Xiao W, Beibei F, Guangsi S, Yu J, Wen Z, Xi H, Youjia X (2015) Iron overload increases osteoclastogenesis and aggravates the effects of ovariectomy on bone mass. J Endocrinol 226:121–134. https://doi.org/10.1530/JOE-14-0657
Xiao W et al (2018) Iron-induced oxidative stress stimulates osteoclast differentiation via NF-kappaB signaling pathway in mouse model. Metabolism. https://doi.org/10.1016/j.metabol.2018.01.005
Xie W, Lorenz S, Dolder S, Hofstetter W (2016) Extracellular iron is a modulator of the differentiation of osteoclast lineage cells. Calcif Tissue Int 98:275–283. https://doi.org/10.1007/s00223-015-0087-1
Yamasaki K, Hagiwara H (2009) Excess iron inhibits osteoblast metabolism. Toxicol Lett 191:211–215. https://doi.org/10.1016/j.toxlet.2009.08.023
Zarjou A, Jeney V, Arosio P, Poli M, Zavaczki E, Balla G, Balla J (2010) Ferritin ferroxidase activity: a potent inhibitor of osteogenesis. J Bone Miner Res 25:164–172. https://doi.org/10.1359/jbmr.091002
Zhang D, Wong CS, Wen C, Li Y (2017) Cellular responses of osteoblast-like cells to 17 elemental metals. J Biomed Mater Res A 105:148–158. https://doi.org/10.1002/jbm.a.35895
Zhao GY, Zhao LP, He YF, Li GF, Gao C, Li K, Xu YJ (2012) A comparison of the biological activities of human osteoblast hFOB1.19 between iron excess and iron deficiency. Biol Trace Elem Res 150:487–495. https://doi.org/10.1007/s12011-012-9511-9
Zhao GY, Di DH, Wang B, Zhang P, Xu YJ (2014) Iron regulates the expression of ferroportin 1 in the cultured hFOB 1.19 osteoblast cell line. Exp Ther Med 8:826–830. https://doi.org/10.3892/etm.2014.1823
Acknowledgments
This work was supported by Grants from the Thailand Research Fund (TRF) through the TRF Senior Research Scholar Grant (RTA6080007 to N. Charoenphandhu), TRF International Research Network Program (IRN60W0001 to K. Wongdee and N. Charoenphandhu), Research Grant for New Scholar from TRF, Office of the Higher Education Commission and Mahidol University (MRG6180268 to K. Lertsuwan), RD&E funding (SCH-NR2016-141) from National Science and Technology Development Agency, Thailand, and the CIF grant, Faculty of Science, Mahidol University.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there is no conflict of interest.
Rights and permissions
About this article
Cite this article
Lertsuwan, K., Nammultriputtar, K., Nanthawuttiphan, S. et al. Ferrous and ferric differentially deteriorate proliferation and differentiation of osteoblast-like UMR-106 cells. Biometals 31, 873–889 (2018). https://doi.org/10.1007/s10534-018-0130-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10534-018-0130-6