Abstract
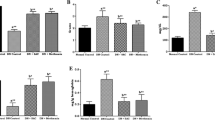
Zinc (Zn) is a component of numerous enzymes that function in a wide range of biological process, including growth, development, immunity and intermediary metabolism. Zn may play a role in chronic states such as cardiovascular disease and diabetes mellitus. Zn acts as cofactor and for many enzymes and proteins and has antioxidant, antiinflammatory and antiapoptotic effects. Taking into consideration that lung is a possible target organ for diabetic complications, the aim of this study was to investigate the protective role of zinc on the glycoprotein content and antioxidant enzyme activities of streptozotocin (STZ) induced diabetic rat tissues. Female Swiss albino rats were divided into four groups. Group I, control; Group II, control + zinc sulfate; Group III, STZ-diabetic; Group IV, diabetic + zinc sulfate. Diabetes was induced by intraperitoneal injection of STZ (65 mg/kg body weight). Zinc sulfate was given daily by gavage at a dose of 100 mg/kg body weight every day for 60 days to groups II and IV. At the last day of the experiment, rats were sacrificed, lung tissues were taken. Also, glycoprotein components, tissue factor (TF) activity, protein carbonyl (PC), advanced oxidative protein products (AOPP), hydroxyproline, and enzyme activities in lung tissues were determined. Glycoprotein components, TF activity, lipid peroxidation, non enzymatic glycation, PC, AOPP, hydroxyl proline, lactate dehydrogenase, catalase, superoxide dismutase, myeloperoxidase, xanthine oxidase, adenosine deaminase and prolidase significantly increased in lung tissues of diabetic rats. Also, glutathione levels, paraoxonase, arylesterase, carbonic anhydrase, and Na+/K+- ATPase activities were decreased. Administration of zinc significantly reversed these effects. Thus, the study indicates that zinc possesses a significantly beneficial effect on the glycoprotein components and oxidant/antioxidant enzyme activities.
Similar content being viewed by others
References
Abel P, Wussow S, Blücher H, Gros G, Retting R, Honig A (1997) Erythrocyte carbonic anhydrase activity in smokers and diabetic patients. Exp Clin Endocrinol Diabetes 105:17–19
Aebi H (1984) Catalase in vitro. Methods Enzymol 105:121–126
American Diabetes Association (2010) Standards of medical care in diabetes-2010. Diabetes Care 33:S11–S61
Araki S, Haneda M, Koya D, Isshiki K, Kume S, Sugimoto T, Kawai H, Nishio Y, Kashiwagi A, Uzu T, Maegawa H (2010) Association between urinary type IV collagen level and deterioration of renal function in type 2 diabetic patients without overt proteinuria. Diabetes Care 33:1805–1810
Aricioğlu A, Aydin S, Turkozkan N, Durmus O (1994) The effect of allopurinol on Na + K + ATPase related lipid peroxidation in ischemic and reperfused rabbit kidney. Gen Pharmacol 25:341–344
Badran M, Morsy R, Soliman H, Elnimr T (2016) Assessment of trace elements levels in patients with Type 2 diabetes using multivariate statistical analysis. J Trace Elem Med Biol 33:114–119
Baskol G, Gumus K, Oner A, Arda H, Karakucuk S (2008) The role of advanced oxidation protein products and total thiols in diabetic retinopathy. Eur J Ophthalmol 18:792–798
Beutler E (1975) Red cell metabolism. A manual of biochemical methods, 2nd edn. Grune and Stratton, New York, pp 112–114
Biswas UK, Kumar A (2012) Study on the changes of carbonic anhydrase activity in insulin resistance and the effect of methylglyoxal. J Pak Med Assoc 62:417–421
Bolkent S, Yanardag R, Bolkent S, Mutlu O (2009) The influence of zinc supplementation on the pancreas of streptozotocin-diabetic rats. Dig Dis Sci 54:2583–2587
Bradley JL, King RH, Muddle JR, Thomas PK (2000) The extracellular matrix of peripheral nerve in diabetic polyneuropathy. Acta Neuropathol 99:539–546
Brzović-Šarić V, Landeka I, Šarić B, Barberić M, Andrijašević L, Cerovski B, Oršolić N, Đikić D (2015) Levels of selected oxidative stress markers in the vitreous and serum of diabetic retinopathy patients. Mol Vis 21:649–664
Camps J, Marsillach J, Joven J (2009) The paroxanases: role in human diseases and methodological difficulties in measurement. Crit Rev Clin Lab Sci 46:83–106
Chinard FP (1952) Photometric estimation of proline and ornithine. J Biol Chem 199:91–95
Corte ED, Stirpe F (1968) Regulation of xanthine oxidase in rat liver: modifications of the enzyme activity of rat liver supernatant on the storage at 20 degrees. Biochem J 108:349–351
Devarajan A, Shih D, Sirinivasa TR (2014) Inflammation, infection, cancer and all that…the role of paraoxonases. Adv Exp Med Biol 824:33–41
Dische Z, Shettles LB (1948) A specific color reaction of methylpentoses and a spectrophotometric micromethod for their determination. J Biol Chem 175:595–603
Emekli-Alturfan E, Kasikci E, Yarat A (2007) Tissue factor activities of streptozotocin induced diabetic rat tissues and the effect of peanut consumption. Diabetes/Metab Res Rev 23:653–658
Furlong CE, Richter RJ, Seidel SL, Motulsky AG (1988) Role of genetic polymorphism of human plasma paraoxonase/arylesterase in hydrolysis of the insecticide metabolites chlorpyrifos oxon and paraoxon. Am J Hum Genet 43:230–238
Gan KN, Smolen A, Eckerson HW, La Du BN (1991) Purification of human serum paraoxonase/arylesterase. Evidence for one esterase catalyzing both activities. Drug Metab Dispos 19:100–106
Gezginci-Oktayoglu S, Basaraner H, Yanardag R, Bolkent S (2009) The effects of combined treatment of antioxidants on the liver injury in STZ diabetic rats. Dig Dis Sci 54:538–546
Gezginci-Oktayoglu S, Sacan O, Yanardag R, Karatug A, Bolkent S (2011) Exendin-4 improves hepatocyte injury by decreasing proliferation through blocking NGF/TrkA in diabetic mice. Peptides 32:223–231
Gezginci-Oktayoglu S, Sacan O, Bolkent S, Ipci Y, Kabasakal L, Sener G, Yanardag R (2014) Chard (Beta vulgaris L. var. cicla) extract ameliorates hyperglycemia by increasing GLUT2 through Akt2 and antioxidant defense in the liver of rats. Acta Histochem 116:32–39
Gopalakrishnan V, Arunakaran J, Aruldhas MM, Srinivasan N (2006) Effects of streptozotocin-induced diabetes mellitus on some bone turnover markers in the vertebrae of ovary-intact and ovariectomized adult rats. Biochem Cell Biol 84:728–736
Hennig B, Meerarani P, Toborek M, McClain CJ (1999) Antioxidant-like properties of zinc in activated endothelial cells. J Am Coll Nutr 18:152–158
Hoshino T, Yamada K, Masuoka K, Tsuboi I, Itoh K, Nonaka K, Oizumi K (1994) Elevated adenosine deaminase activity in the serum of patients with diabetes mellitus. Diabetes Res Clin Pract 25:97–102
Ikeda Y, Suehiro T, Inoue T, Nakauchi Y, Morita T, Arii K, Ito H, Kumon Y, Hashimoto K (1998) Serum paraoxonase activity and its relationship to diabetic complications in patients with non-insulin-dependent diabetes mellitus. Metabolism 47:598–602
Ingram GI, Hills M (1976) Reference method for the one-stage prothrombin-time test on human blood. Thromb Haemost 36:237–238
Jansen J, Karges W, Rink L (2009) Zinc and diabetes-clinical links and molecular mechanisms. J Nutr Biochem 20:399–417
Kanth VR, Reddy PUM, Raju TN (2008) Attenuation of streptozotocin-induced oxidative stress in hepatic and intestinal tissues of Wistar rat by methanolic-garlic extract. Acta Diabetol 45:243–251
Karatug A, Sacan O, Coskun ZM, Bolkent S, Yanardag R, Turk N, Bolkent S (2012) Regulation of gene expression and biochemical changes in small intestine of newborn diabetic rats by exogenous ghrelin. Peptides 33:101–108
Karatug A, Kaptan A, Bolkent S, Mutlu O, Yanardag R (2013) Alterations in kidney tissue following zinc supplementation to STZ-induced diabetic rats. J Trace Elem Med Biol 27:52–57
Karker H (1964) Method for estimation of serum adenosine deaminase. Scand J Clin Lab Invest 16:570–574
Koksel O, Ozdulger A, Ercil M, Tamer L, Ercan B, Atik U, Cinel L, Cinel L, Kanik A (2004) Effects of N-acetylcysteine on oxidant-antioxidant balance in oleic acid-induced lung injury. Exp Lung Res 30:431–436
Konukoglu D, Serin O, Akcay T, Hatemi H (1999) Relationship between diabetic angiopathic complications and serum total and lipid associated sialic acid concentrations. Med Sci Res 27:53–55
Kumar PA, Haseeb A, Suryanarayana P, Ehtesham NZ, Reddy GB (2005) Elevated expression of αA- and αB-crystallins in streptozotocin-induced diabetic rat. Arch Biochem Biophys 444:77–83
Ledwozyw A, Michalak J, Stepień A, Kadziołka A (1986) The relationship between plasma triglycerides, cholesterol, total lipids and lipid peroxidation products during human atherosclerosis. Clin Chim Acta 155:275–283
Levine RL, Garland D, Oliver CN, Amici A, Climent I, Lenz AG, Ahn BW, Shaltiel S, Stantman ER (1990) Determination of carbonyl content in oxidatively modified proteins. In: Packer L, Glazer AN (eds) Methods in enzymology, oxygen radicals in biological systems, vol 186. Academic Press, California, pp 464–478
Lorentz K, Weiss T, Kraas E (1986) Sialic acid in human serum and cerebrospinal fluid. Comparison of methods and reference values. J Clin Chem Clin Biochem 24:189–198
Lowry OH, Rosebrough HJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193:265–275
Mackness B, Durrington PN, Abuashia B, Boulton AJM, Mackness MI (2000) Low paraoxonase activity in type II diabetes mellitus complicated by retinopathy. Clin Sci 98:355–363
Maritim AC, Sanders RA, Watkins JB III (2003) Diabetes oxidative stress, and antioxidants: a review. J Biochem Mol Toxicol 17:24–38
Marjani A, Moradi A, Saeedi M (2005) Plasma lipid peroxidation, zinc and erythrocyte Cu—Zn superoxide dismutase enzyme activity in patients with type 2 diabetes mellitus in Gorgan city (south east of the Caspian Sea). J Med Sci 7:585–590
Marvisi Mi Bartolini L, del Borello P, Brianti M, Marani G, Guariglia A, Cuomo A (2001) Pulmonary function in non-insulin-dependent diabetes mellitus. Respiration 68:268–272
Matsumoto S, Koshiishi I, Inoguchi T, Nawata H, Utsumi H (2003) Confirmation of superoxide generation via xanthine oxidase in streptozotocin-induced diabetic mice. Free Radic Res 37:767–772
Muruganathan U, Srinivasan S, Indumathi D (2013) Antihyperglycemic effect of carvone: effect on the levels of glycoprotein components in streptozotocin-induced diabetic rats. J Acute Disease 2:310–315
Mylorie AA, Colins H, Umbles C, Kyle J (1986) Erythrocyte superoxide dismutase activity and other parameters of copper status in rats ingesting lead acetate. Toxicol Appl Pharmacol 82:512–520
Oberley LW (1988) Free radicals and diabetes. Free Radic Biol Med 5:113–124
Ozsoy N, Can A, Mutlu O, Akev N, Yanardag R (2012) Oral zinc supplementation protects rat kidney tissue from oxidative stress in diabetic rats. Kafkas Univ Vet Fak 18:545–550
Paglia DE, Valentine WN (1967) Studies on the quantitative and qualitative characterization of erythrocyte glutathione peroxidase. J Lab Clin Med 70:158–169
Pari L, Ashokkumar N (2006) Glycoprotein changes in non-insulin dependent diabetic rats: effect of N-benzoyl-d-phenylalanine and metformin. Therapie 61:125–131
Parker KM, England JD, Da Costa J, Hess RL, Goldstein DE (1981) Improved colorimetric assay for glycosylated haemoglobin. Clin Chem 27:669–672
Parui R, Gambhir KK, Cruz I, Hosten AO (1992) Erythrocyte carbonic anhydrase: a major intracellular enzyme to regulate cellular sodium metabolism in chronic renal failure patients with diabetes and hypertension. Biochem Int 26:809–820
Patti ME, Virkamäki A, Landaker EJ, Kahn CR, Yki-Järvinen H (1999) Action of the hexosamine pathway by glucosamine in vivo induces insulin of early postreceptor insulin signaling events in skeletal muscle. Diabetes 48:1562–1571
Pinter E, Peyman JA, Snow K, Jamieson JD, Warshaw JB (1991) Effects of maternal diabetes on fetal rat lung ion transport. Contribution of alveolar and bronchiolar epithelial cells to Na+, K+-ATPase expression. J Clin Invest 87:821–830
Prasad AS (1993) Clinical spectrum of human zinc deficiency. In: Prasad AS (ed) Biochemistry of zinc. Plenum Press, New York, pp 219–258
Prasad AS (2008) Zinc in human health: effect of zinc on immune cells. Mol Med 14:353–357
Prasad AS (2014) Zinc is an antioxidant and anti-inflammatory agent: its role in human health. Front Nutr 1:14
Prasad AS, Bao B, Beck FWJ, Kucuk O, Sarkar FH (2004) Antioxidant effect of zinc in humans. Free Radic Biol Med 37:1182–1190
Punithavathi VR, Prince PSM, Kumar MR, Selvakumari CJ (2011) Protective effects of gallic acid on hepatic lipid peroxide metabolism, glycoprotein components and lipid in streptozotocin-induced type II diabetic wistar rats. J Biochem Mol Toxicol 25:68–76
Rauch U, Nemerson Y (2000) Circulating tissue factor and thrombosis. Curr Opin Hematol 7:273–277
Reedy GK, Enwemeka CS (1996) A simplified method for the analysis of hydroxyproline in biological tissues. Clin Biochem 29:225–229
Ridderstap AS, Bonting SL (1969) Na+-K+-activated ATPase and exocrine pancreatic secretion in vitro. Am J Physiol 217:1721–1727
Rodrigo R, Trujillo S, Bosco C, Orellana M, Thielemann L, Araya J (2002) Changes in (Na + K)-adenosine triphosphatase activity and ultrastructure of lung and kidney associated with oxidative stress induced by acute ethanol intoxication. Chest 121:589–596
Rutkiewicz J, Górski J (1990) On the role of insulin in regulation of adenosine deaminase activity in rat tissues. FEBS Lett 271:79–80
Shah GS, Morofuji Y, Banks WA, Price TO (2013) High glucose-induced mitochondrial respiration and reactive oxygen species in mouse cerebral pericytes is reversed by pharmacological inhibition of mitochondrial carbonic anhydrases: implications for cerebral microvascular disease in diabetes. Biochem Biophys Res Commun 440:354–358
Shulman GI (2000) Cellular mechanisms of insulin resistance. J Clin Invest 106:171–176
Sulaiman GM, Al-Amiery AA, Mohammed AA, Al-Temimi AA (2012) The effect of cherry sticks extract on the levels of glycoproteins in alloxan-induced experimental diabetic mice. Ann Clin Lab Sci 42:34–41
Sundaram R, Naresh R, Shanthi P, Sachdanandam P (2012) Antihyperglycemic effect of iridoid glucoside, isolated from the leaves of Vitex negundo in streptozotocin-induced diabetic rats with special reference to glycoprotein components. Phytomedicine 19:211–216
Tran B, Oliver S, Rosa J, Galassetti P (2012) Aspects of inflammation and oxidative stress in pediatric obesity and type 1 diabetes: An overview of ten years of studies. Exp Diabetes Res. Article ID 683680
Tunali S, Yanardag R (2013) Protective effect of vanadyl sulfate on skin injury in streptozotocin-induced diabetic rats. Hum Exp Toxicol 32:1206–1212
Vallee BL, Falchuk KH (1993) The biochemical basis of zinc physiology. Physiol Rev 73:79–118
Verpoorte JA, Mehta S, Edsall JT (1967) Esterase activities of human carbonic anhydrases B and C. J Biol Chem 242:4221–4229
Wei H, Frenkel K (1991) In vivo formation of oxidized DNA bases in tumor promoter-treated mouse skin. Cancer Res 51:4443–4449
Wendel A (1981) Glutathione peroxidase. Methods Enzymol 77:325–333
Wiese TJ, Dunlap JA, Yorek MA (1997) Effect of l-fucose and d-glucose concentration on l-fucoprotein metabolism in human Hep G2 cells and changes in fucoyltransferase and α-l-fucosidase activity in liver of diabetic rats. Biochim Biophys Acta 1335:61–72
Wijesekara N, Chimienti F, Wheeler MB (2009) Zinc, a regulator of islet function and glucose homeostasis. Diabetes Obes Metab 11:202–214
Winzler RJ (1955) Determination of serum glycoproteins. In: Glick DP (ed) Methods of biochemical analysis. Interscience Publisher Inc, New York, pp 279–311
Witko-Sarsat V, Friedlander M, Capeillére-Blandin C, Nguyen-Khoa T, Nguyen AT, Zingraff J, Jungers P, Descamps-Latscha B (1996) Advanced oxidation protein products as a novel marker of oxidative stress in uremia. Kidney Int 49:1304–1313
Wroblewski F (1957) Clinical significance of serum enzyme alterations associated with myocardial infarction. Am Heart J 54:219–224
Yanardag R, Tunali S (2006) Vanadyl sulfate administration protects the streptozotocin-induced oxidative damage to brain tissue in rats. Mol Cell Biochem 286:153–159
Acknowledgments
This work was supported by Istanbul University Scientific Research Projects. Project Number: UDP-37282.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sacan, O., Turkyilmaz, I.B., Bayrak, B.B. et al. Zinc supplementation ameliorates glycoprotein components and oxidative stress changes in the lung of streptozotocin diabetic rats. Biometals 29, 239–248 (2016). https://doi.org/10.1007/s10534-016-9911-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10534-016-9911-y