Abstract
To clarify the influence of MTHFR C677T and A1298C polymorphisms on gastric cancer (GC), a meta-analysis of eight case-control studies (1,584/2,785 cases/controls) was carried out. Overall, there was moderate heterogeneity among studies, and the C677T allele T was associated with a 27% increased risk of GC compared with C allele: the random effects (RE) OR (95% confidence interval in parenthesis) was significant [OR=1.27 (1.13–1.44)]. In East Asians, the association was significant: RE OR=1.28 (1.14–1.44), whereas, in Caucasians it was not significant. Regarding gastric cancer adenocarcinoma (GCA), an association for the allele contrast in East Asians was detected: fixed effects (FE) OR=1.36 (1.18–1.56). The recessive model for allele T produced significant results overall and in East Asians for GC [FE OR=1.47 (1.26–1.72) and FE OR=1.61 (1.32–1.96), respectively] and for GCA [RE OR=1.53 (1.13–2.05) and FE OR=1.70 (1.36–2.12)]. The A1298C polymorphism was associated with GCA in East Asians: the FE OR for the allele contrast (C vs. A) was 1.38 (1.18–1.62), and under a recessive model for allele C, OR=1.62 (1.28–2.06). There were no sources of bias in the selected studies; the differential magnitude of effect in large versus small studies was not significant. In conclusion, there is evidence of association between MTHFR polymorphisms and GC, mainly in East Asians.
Similar content being viewed by others
Introduction
Stomach cancer is the second most frequent cancer worldwide, accounting for about 12% of cancer deaths each year (Parkin et al. 1999). Epidemiological studies have indicated that low folate, a constituent of vegetables and fruits, is associated with an increased risk of cancer, including gastric cancer (GC) (Kobayashi et al. 2002; La Vecchia et al. 2002).
Methylenetetrahydrofolate reductase (MTHFR) is a central regulatory enzyme in folate metabolism. MTHFR catalyses the reduction of 5,10-methylenetetrahydrofolate to 5-methyltetrahydrofolate, the predominant circulating form of folate and the carbon donor for the remethylation of homocysteine to methionine (Kim et al. 1999; Sarbia et al. 2005). The MTHFR (Frosst 1995; Weisberg 1998) gene is localized on chromosome 1p36.3. Two common polymorphisms identified in the MTHFR gene are implicated in the development of GC: (1) the MTHFR C677T (exon 4 at codon 222), in which a C>T substitution at position 677 cause an alanine to valine substitution, and (2) the A1298C (exon 7 at codon 429), causing a glutamate to alanine (A>C) substitution. A1298C influences specific activity of the enzyme, homocysteine levels, and plasma folate concentration, but to a lesser extent than the C677T polymorphism does (Blount et al. 1997; Shen et al. 2005). However, other functional polymorphisms in genes associated with impaired folate metabolism may also contribute to the risk of GC (Ueland et al. 2001).
The studies investigating the role of C677T and A1298C polymorphisms in the development of GC have produced contradicting or inconclusive results. In order to provide overall information about the MTHFR C677T and A1298C polymorphisms and the risk of GC, a meta-analysis of all case-control observational studies with available data was performed (Lau et al. 1997). The meta-analysis estimated the effect of allele contrast, the contrast of homozygotes, and the contrasts for the dominant and recessive models (Zintzaras et al. 2005). The heterogeneity between studies and the existence of potential bias were also examined.
Methods
Study identification and selection
Eligible studies were identified by searching the PubMed database for relevant reports in English published before January 2006 using the search criterion: (“MTHFR” or “C677T” or “A1298C”) and (“gastric cancer” or “stomach cancer” or “gastric cancer adenocarcinoma”). The reference lists of retrieved articles were also reviewed to identify additional articles missed by the PubMed search.
Studies that determined the distribution of the C677T and A1298C genotypes in cases with GC and/or GCA diagnosed by histopathological biopsy, and in controls free of cancer were eligible for inclusion in the meta-analysis. Studies based on pedigree data were excluded since they investigate linkage and not association. In studies with overlapping cases or controls, the most recent and/or the largest study with extractable data was included in the meta-analysis. Only studies that have used validated genotyping methods were considered. Meeting abstracts, case reports, editorials and review articles were excluded.
Data extraction and synthesis
The following information was extracted from each study: first author, journal, year of publication, ethnicity of study population, demographics, matching, genotyping method, blindness, and the number of cases and controls for each C677T or A1298C genotype.
The meta-analysis examined the overall association for the allele contrast, the contrast of homozygotes, and the recessive and dominant models. The effect of association was indicated as odds ratio (OR) with the corresponding 95% confidence interval (CI). The pooled OR was estimated using fixed effects (FE) (Mantel-Haenszel) and random effects (RE) (DerSimonian and Laird) models (Lau 1997). RE modelling assumes heterogeneity between the studies, and it incorporates the between-study variability. The heterogeneity between studies was tested using the Q statistic (Zintzaras and Ioannidis 2005). If P<0.10 then the heterogeneity was considered statistically significant. Heterogeneity was quantified using the I2 metric, which is independent of the number of studies in the meta-analysis (I2<25% no heterogeneity; I2=25–50% moderate heterogeneity; I2>50% large or extreme heterogeneity) (Higgins and Thompson 2002). A cumulative meta-analysis (Lau et al. 1992; Whitehead 1997) and recursive meta-analysis were carried out in order to evaluate the trend of pooled OR for the allele contrast in time. The differential magnitude of effect in large versus small studies (Ioannidis et al. 2003; Zintzaras et al. 2005) for the allele contrast was tested using the Egger regression test for funnel plot asymmetry (Egger et al. 1997) and the Begg-Mazumdar test, which is based on Kendall’s tau (Begg and Mazumdar 1994). Whether the OR in the first study versus the pooled OR of the subsequent studies differed beyond chance (P<0.05) was assessed using the Z statistic (Ioannidis et al. 2003).
The meta-analysis consisted of the main (overall) analysis, which includes all available data, and the sensitivity analysis, which examines the effect of excluding specific studies such as studies with the controls not in Hardy-Weinberg equilibrium. Subgroup analyses for the GCA cases and for each ethnicity were also performed. However, the consistency of genetic effects across these traditionally defined racial groups does not necessarily mean that race-specific genetic effects are exactly the same. Analyses were performed using Meta-Analyst (Joseph Lau, Boston, Massachusetts, USA 1998), and CVF90 with IMLS library (Zintzaras and Hadjigeorgiou 2005; Zintzaras et al. 2005; Zintzaras and Stefanidis 2005).
The distribution of the genotypes in the control group was tested for Hardy-Weinberg equilibrium using an exact test implemented by GDA software (Weir 1996). Studies with the controls not in Hardy-Weinberg equilibrium were subjected to a sensitivity analysis (Zintzaras et al 2006), i.e., the effect of excluding specific studies was examined.
Results
Eligible studies
A total of 13 published articles reported on the relationship between any of the C677T and A1298C polymorphisms and GC. However, data from only eight articles met the inclusion criteria. The studies that investigated A1298C also investigated C677T (four studies). The studies were published between 2002 and 2005. In all studies, the cases were histologically confirmed (or historically based on pathology), and the controls were free of GC cancer (two studies, Sarbia et al. 2005 and Graziano et al. 2006, reported the use of healthy controls). Six studies stated that the controls were age-sex matched (Stolzenberg et al. 2003; Kim et al. 2005; Wang et al. 2005; Shen et al. 2005; Lacasana et al. 2005; Graziano et al. 2006). Studies were conducted in various populations of different ethnicities: five studies were conducted in populations of East Asian ethnicity (Miao et al. 2002; Stolzenberg et al. 2003; Kim et al. 2005; Wang et al. 2005; Shen et al. 2005), two studies involved Caucasians (Sarbia et al. 2005; Graziano et al. 2005), and one study Mexicans (Lacasana et al. 2005). A list of all the details abstracted from these studies is provided in Tables 1 and 2. For the determination of the genetic polymorphisms of C677T and A1298C, validated genotyping methods were used in all studies: PCR-RFLP (Miao et al. 2002; Kim et al. 2005; Wang et al. 2005; Shen et al. 2005; Lacasana et al. 2005; Graziano et al. 2006), real-time PCR with allele-specific oligonucleotide method (Stolzenberg et al. 2003), or real-time fluorescence PCR and melting-curve analysis (Sarbia et al. 2005).
Summary statistics
The studies provided 1,584/2,785 cases/controls for C677T and 760/1,624 cases/controls for A1298C. In cases and controls, the allele C and the CT genotype were the most common for C677T. The allele A was the most common for A1298C, and the frequency of the AA genotype was the highest in both cases and controls, while that for genotype CC was the lowest (Table 2).
In three studies (Kim et al. 2005; Shen et al. 2005; Lacasana et al. 2005) investigating the C677T polymorphism, and in two studies (Stolzenberg et al. 2003; Miao et al. 2002) of the A1298C polymorphism, the distribution of the genotypes in control groups was not in Hardy-Weinberg equilibrium (P<0.05), indicating genotyping errors and/or population stratification (Zintzaras and Hadjigeorgiou 2004; Xu et al. 2002). Therefore, a sensitivity analysis was carried out for these studies. None of these studies reported linkage disequilibrium for C677T and A1298C polymorphisms.
Main results, subgroup and sensitivity analyses
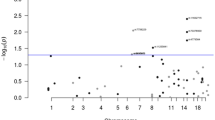
The heterogeneity results and the effect of the association between the polymorphisms and the risk of GC for the genetic contrasts under investigation are shown in Fig. 1 and Table 3.
C677T and A1298C MTHFR polymorphisms and the risk of gastric cancer: contrast of allele T against C for C677T, and C against A for A1298C. Each study is shown with an odds ratio (OR) estimate and the corresponding 95% confidence interval (95% ci). The random effects pooled odds ratios are shown. The horizontal axis is plotted on a log scale. HWE Hardy-Weinberg equilibrium, GCA gastric cancer adenocarcinoma
Overall, there was moderate heterogeneity (P=0.12, I2=40%) among the eight studies, and the C677T allele T was associated with a 27% increased risk of GC compared with the C allele: the RE pooled OR was significant [OR=1.27 95% confidence interval in parenthesis (1.13–1.44)]. In subgroup analysis, the RE pooled OR was significant: OR=1.28 (1.14–1.44) for the East Asians. However, for those studies involving Caucasians the RE pooled OR was not significant: OR=1.11 (0.38–3.20). Regarding the GCA, the analysis detected an association for the allele contrast in East Asians: FE OR=1.36 (1.18–1.56). The contrast of homozygotes (TT vs. CC) produced the same pattern of results as the allele contrast. The recessive model for the effect of T allele produced significant results overall in GC and in GCA: FE OR=1.47 (1.26–1.72) and RE OR=1.53 (1.13–2.05), respectively. In East Asians, the association was significant both for GC and GCA: FE OR=1.61 (1.32–1.96) and FE OR=1.70 (1.36–2.12), respectively. The sensitivity analysis for the above contrasts did not alter the pattern of results. The dominant model for the effect of T allele produced no significant associations, except for the main analysis; however, the sensitivity analysis showed non-significance.
Concerning the A1298C polymorphism, the association was significant for GCA in East Asians: the FE pooled OR for the allele contrast (C vs. A) was 1.38 (1.18–1.62). The contrast of homozygotes (CC vs. AA) and the recessive model for the effect of allele C showed highly significant associations: FE OR=1.71 (1.25–2.32) and FE OR=1.62 (1.28–2.06). The sensitivity analysis did not change the pattern of results.
Potential bias
Two studies (Miao 2002; Stolzenberg 2003) reported that genotyping was performed with blinding to case/control status. Overall, the cumulative and recursive meta-analyses for the allelic contrast (C677T T vs. C) showed that RE pooled OR had a downward trend in the period 2002–2005 (OR=1.44 in 2002, first study; OR=1.34 in 2003; and OR=1.23 in 2005), and then increased to 1.27 in 2006 (−12% relative change from 2002). There is no statistical difference between the OR (1.44) of the first study versus the pooled RE OR of the subsequent studies (1.25) (Z=0.90, P=0.37). The cumulative and recursive meta-analyses for the allelic contrast (A1298C C vs. A) showed that RE pooled OR remained nonsignificant and fairly constant over the period 2002–2005: OR=1.02 in 2002 (first study), OR=0.98 in 2003, and OR=1.00 in 2005.
The Egger test and the Begg-Mazumdar test were based on the allele contrast of C677T polymorphism, and they indicated that there is no differential magnitude of effect in large versus small studies (i.e., no source of bias) (P=0.45 and 0.72, respectively). However, these results might not be so reliable since the number of studies is relatively small.
Discussion
Regarding the MTHFR polymorphisms and their association with cancers, definite conclusions cannot be drawn. Some studies reported an increased risk of breast (Zintzaras 2006), endometrium (Estellar et al. 1997), and oesophagus (Song et al. 2001) cancers, whereas others reported decreased risk of leukemia (Skibola et al. 1999) and colorectal cancer (Chen et al. 2005) associated with these polymorphisms when folate supply was sufficient (Wang et al. 2005). The studies investigating the association with stomach cancer have indicated increased risk or no association. This is the first meta-analysis to examine the C677T and A1298C polymorphisms of the MTHFR gene and their relationship to susceptibility for GC. The meta-analysis involved eight studies that investigated the C677T polymorphism and four studies the A1298C polymorphism. The studies provided 1,584/2,785 cases/controls for C677T, and 760/1,624 cases/controls for A1298C. The strength of the present analysis is based on the aggregation of data from the published studies, thus there is more information for estimating the effect of association (Muncher et al. 2002; Zintzaras and Hadjigeorgiou 2004). In the present meta-analysis the effect of allele contrast, the contrast of homozygotes, and the contrast for the dominant and recessive models for alleles C677T T and A1298C C were estimated. In addition, the consistency of genetic effects across populations of different racial descent (ethnicity) was investigated.
The overall result of the present meta-analysis demonstrated that MTHFR C677T polymorphism could be a risk factor for GC, and GCA, under a recessive model for allele T. This association holds true especially in subjects of East Asian descent—the results on the Caucasian population have shown no association, but the number of studies on Caucasians was limited. It seems that MTHFR A1298C is a risk factor for GCA in East Asian cases under a recessive model for allele C, however, more studies are required for definite conclusions. The sensitivity analysis for the studies not in Hardy-Weinberg equilibrium did not alter the pattern of association. In investigating the allelic or genotypic contrasts, quantitative heterogeneity varied from no to high heterogeneity. The meta-analysis showed no potential bias, since there was no differential magnitude of effect in large versus small studies.
A major limitation of using a meta-analytic approach for population-based observational studies is the confounding factors (e.g., age, sex, and life style) that influence the estimates of associations. In addition, studies on various ethnicities may help to elucidate the genetics of GC further. In this meta-analysis, only the unadjusted pooled ORs could be calculated since data for each level of possible confounding factors were not provided. The cases and controls of each study were well defined with similar inclusion criteria, although they unavoidably cover a spectrum of disease in terms of clinical, demographic and dietary data. In addition, the risk effect may depend on gene methylation, which is determined by folate intake during development, and on the interaction with other risk factors that modulate the development of cancer, such as lifestyle (including smoking, alcohol consumption and body mass index). Folic acid consumption is essential for maintaining or altering the effect of the polymorphic variants such as MTHFR (Ma et al. 1997). However, evidence for the relation between folate intake and GC is insufficient to lead to specific public health interventions (Choumenkovitch et al. 2002; Bailey et al. 2003).
The present meta-analysis provided evidence that TT and CC genotypes of the MTHFR C677T and A1298C polymorphisms, respectively, contribute to susceptibility of GC. GC is a complex disease with multifactorial etiology and therefore, the contributing pathogenetic role of lifestyle factors and dietary intake should also be considered. The existence of gene–nutrient interactions may explain the discrepancy in results of individual genetic association studies, and therefore, clinical trials of folic acid with a sufficiently large number of participants are warranted.
References
Bailey LB (2003) Folate, methyl-related nutrients, alcohol, and the MTHFR 677C→T polymorphism affect cancer risk: intake recommendations. J Nutr 133:3748S–3753S
Begg CB, Mazumdar M (1994) Operating characteristics of a rank correlation test for publication bias. Biometrics 50:1088–1101
Blount BC, Mack MM, Wehr CM, MacGregor JT, Hiatt RA, Wang G, Wickramasinghe SN, Everson RB, Ames BN (1997) Folate deficiency causes uracil misincorporation into human DNA and chromosome breakage: implications for cancer and neuronal damage. Proc Natl Acad Sci USA 7:3290–3295
Chen K, Jiang QT, He HQ (2005) Relationship between metabolic enzyme polymorphism and colorectal cancer. World J Gastroenterol 11:331–335
Choumenkovitch SF, Selhub J, Wilson PW, Rader JI, Rosenberg IH, Jacques PF (2002) Folic acid intake from fortification in United States exceeds predictions. J Nutr 132:2792–2798
Egger M, Davey SG, Schneider M, Minder C (1997) Bias in meta-analysis detected by a simple, graphical test. BMJ 315:629–634
Esteller M, Garcia A, Martinez-Palones JM, Xercavins J, Reventos J (1997) Germ line polymorphisms in cytochrome-P450 1A1 (C4887 CYP1A1) and methylenetetrahydrofolate reductase (MTHFR) genes and endometrial cancer susceptibility. Carcinogenesis 18:2307–2311
Frosst P, Blom HJ, Milos R, Goyette P, Sheppard CA, Matthews RG, Boers GJ, den Heijer M, Kluijtmans LA, van den Heuvel LP (1995) A candidate genetic risk factor for vascular disease: a common mutation in methylenetetrahydrofolate reductase. Nat Genet 10:111–113
Graziano F, Kawakami K, Ruzzo A, Watanabe G, Santini D, Pizzagalli F, Bisonni R, Mari D, Floriani I, Catalano V, Silva R, Tonini G, Torri V, Giustini L, Magnani M (2006) Methylenetetrahydrofolate reductase 677C/T gene polymorphism, gastric cancer susceptibility and genomic DNA hypomethylation in an at-risk Italian population. Int J Cancer 118:628–632
Higgins JP, Thompson SG (2002) Quantifying heterogeneity in a meta-analysis. Stat Med 21:1539–1558
Ioannidis JP, Trikalinos TA, Ntzani EE, Contopoulos-Ioannidis DG (2003) Genetic associations in large versus small studies: an empirical assessment. Lancet 361:567–571
Ioannidis JP, Ntzani EE, Trikalinos TA (2004) “Racial” differences in genetic effects for complex diseases. Nat Genet 36:1312–1318
Kim JK, Kim S, Han JH, Kim HJ, Chong SY, Hong SP, Hwang SG, Ahn JY, Cha KY, Oh D, Kim NK (2005) Polymorphisms of 5,10-methylenetetrahydrofolate reductase and risk of stomach cancer in a Korean population. Anticancer Res 25:2249–2252
Kim YI (1999) Folate and cancer prevention: a new medical application of folate beyond hyperhomocysteinemia and neural tube defects. Nutr Rev 57:314–321
La Vecchia C, Ferraroni M, D’Avanzo B, Decarli A, Franceschi S (1994) Selected micronutrient intake and the risk of gastric cancer. Cancer Epidemiol Biomarkers Prev 3:393–398
Lacasana-Navarro M, Galvan-Portillo M, Chen J, Lopez-Cervantes M, Lopez-Carrillo L (2005) Methylenetetrahydrofolate reductase 677C>T polymorphism and gastric cancer susceptibility in Mexico. Eur J Cancer 42(4):528–533
Lau J, Antman EM, Jimenez-Silva J, Kupelnick B, Mosteller F, Chalmers TC (1992) Cumulative meta-analysis of therapeutic trials for myocardial infarction. N Engl J Med 327:248–254
Lau J, Ioannidis JP, Schmid CH (1997) Quantitative synthesis in systematic reviews. Ann Intern Med 127:820–826
Ma J, Stampfer MJ, Christensen B, Giovannucci E, Hunter DJ, Chen J, Willett WC, Selhub J, Hennekens CH, Gravel R, Rozen R (1999) A polymorphism of the methionine synthase gene: association with plasma folate, vitamin B12, homocyst(e)ine, and colorectal cancer risk. Cancer Epidemiol Biomarkers Prev 8:825–829
Miao X, Xing D, Tan W, Qi J, Lu W, Lin D (2002) Susceptibility to gastric cardia adenocarcinoma and genetic polymorphisms in methylenetetrahydrofolate reductase in an at-risk Chinese population. Cancer Epidemiol Biomarkers Prev 11:1454–1458
Muncer S (2002) Response to: ‘Power dressing and meta-analysis: incorporating power analysis into meta-analysis’. J Adv Nurs 38:274–280
Parkin DM, Pisani P, Ferlay J (1999) Global cancer statistics. CA Cancer J Clin 49:33–64
Sarbia M, Geddert H, Kiel S, Kandemin Y, Schulz WA, Vossen S, Zotz RD, Willers R, Baldus SE, Schneider PM, Gabbert HE (2005) Methylenetetrahydrofolate reductase C677T polymorphism and risk of adenocarcinoma of the upper gastrointestinal tract. Scand J Gastroenterol 40:109–111
Shen H, Xu Y, Zheng Y, Qian Y, Yu R, Qin Y, Wang X, Spitz MR, Wei Q (2001) Polymorphisms of 5,10-methylenetetrahydrofolate reductase and risk of gastric cancer in a Chinese population: a case-control study. Int J Cancer 95:332–336
Shen H, Newmann AS, Hu Z, Zhang Z, Xu Y, Wang L, Hu X, Guo J, Wang X, Wei Q (2005) Methylenetetrahydrofolate reductase polymorphisms/haplotypes and risk of gastric cancer: a case-control analysis in China. Oncol Rep 13:355–360
Skibola CF, Smith MT, Kane E, Roman E, Rollinson S, Cartwright RA, Morgan G (1999) Polymorphisms in the methylenetetrahydrofolate reductase gene are associated with susceptibility to acute leukemia in adults. Proc Natl Acad Sci USA 96:12810–12815
Song C, Xing D, Tan W, Wei Q, Lin D (2001) Methylenetetrahydrofolate reductase polymorphisms increase risk of esophageal squamous cell carcinoma in a Chinese population. Cancer Res 61:3272–3275
Stolzenberg-Solomon RZ, Qiao YL, Abnet CC, Ratnasinghe DL, Dawsey SM, Dong ZW, Taylor PR, Mark SD (2003) Esophageal and gastric cardia cancer risk and folate- and vitamin B(12)-related polymorphisms in Linxian, China. Cancer Epidemiol Biomarkers Prev 12:1222–1226
Ueland PM, Hustad S, Schneede J, Refsum H, Vollset SE (2001) Biological and clinical implications of the MTHFR C677T polymorphism. Trends Pharmacol Sci 22:195–201
Wang LD, Guo RF, Fan ZM, He X, Gao SS, Guo HQ, Matsuo K, Yin LM, Li JL (2005) Association of methylenetetrahydrofolate reductase and thymidylate synthase promoter polymorphisms with genetic susceptibility to esophageal and cardia cancer in a Chinese high-risk population. Dis Esophagus 18:177–184
Weir BS (1996) Genetic data analysis II: methods for discrete population genetic data. Sinauer, Sunderland
Weisberg I, Tran P, Christensen B, Sibani S, Rozen R (1998) A second genetic polymorphism in methylenetetrahydrofolate reductase (MTHFR) associated with decreased enzyme activity. Mol Genet Metab 64:169–172
Whitehead A (1997) A prospectively planned cumulative meta-analysis applied to a series of concurrent clinical trials. Stat Med 16:2901–2913
Xu J, Turner A, Little J, Bleecker ER, Meyers DA (2002) Positive results in association studies are associated with departure from Hardy–Weinberg equilibrium: hint for genotyping error? Hum Genet 111:573–574
Zintzaras E (2006) Methylenetetrahydrofolate reductase (MTHFR) gene and susceptibility to breast cancer: a meta-analysis. Clin Genet 69:297–301
Zintzaras E, Hadjigeorgiou GM (2004) Association of paraoxonase 1 gene polymorphisms with risk of Parkinson’s disease: a meta-analysis. J Hum Genet 49:474–481
Zintzaras E, Hadjigeorgiou GM (2005) The role of G196A polymorphism in the brain-derived neurotrophic factor gene in the cause of Parkinson’s disease: a meta-analysis. J Hum Genet 50:560–566
Zintzaras E, Ioannidis JP (2005) Heterogeneity testing in meta-analysis of genome searches. Genet Epidemiol 28:123–137
Zintzaras E, Stefanidis I (2005) Association between the GLUT1 gene polymorphism and the risk of diabetic nephropathy: a meta-analysis. J Hum Genet 50:84–91
Zintzaras E, Chatzoulis DZ, Karabatsas CH, Stefanidis I (2005) The relationship between C677T methylenetetrahydrofolate reductase gene polymorphism and retinopathy in type 2 diabetes: a meta-analysis. J Hum Genet 50:267–275
Zintzaras E, Stefanidis I, Santos M, Vidal F (2006) Do alcohol-metabolizing enzyme gene polymorphisms increase the risk of alcoholism and alcoholic liver disease? Hepatology 43:127–132
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zintzaras, E. Association of methylenetetrahydrofolate reductase (MTHFR) polymorphisms with genetic susceptibility to gastric cancer: a meta-analysis. J Hum Genet 51, 618–624 (2006). https://doi.org/10.1007/s10038-006-0405-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10038-006-0405-6
Keywords
This article is cited by
-
CT genotype of 5,10-methylenetetrahydrofolate reductase (MTHFR) C677T polymorphism is protector factor of major depressive disorder in the Tunisian population: a case control study
Annals of General Psychiatry (2016)
-
The clinical significance of methylenetetrahydrofolate reductase (MTHFR) polymorphisms in acute lymphoblastic leukemia
Comparative Clinical Pathology (2012)
-
MTHFR polymorphisms in gastric cancer and in first-degree relatives of patients with gastric cancer
Tumor Biology (2010)
-
APOE gene polymorphisms and response to statin therapy
The Pharmacogenomics Journal (2009)
-
Investigation of inter-individual variability of the one-carbon folate pathway: a bioinformatic and genetic review
The Pharmacogenomics Journal (2009)