Abstract
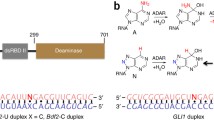
The Z-DNA-binding domain of human double-stranded RNA adenosine deaminase I (hZαADAR1) can specifically recognize the left-handed Z-DNA which preferentially occurs at alternating purine-pyrimidine repeats, especially the CG-repeats. The interactions of hZαADAR1 and Z-DNAs in different sequence contexts can affect many important biological functions including gene regulation and chromatin remodeling. Therefore it is of great necessity to fully understand their recognition mechanisms. However, most existing studies are aimed at the standard CG-repeat Z-DNA rather than the non-CG-repeats, and whether the molecular basis of hZαADAR1 binding to various Z-DNAs are identical or not is still unclear on the atomic level. Here, based on the recently determined crystal structures of three representative non-CG-repeat Z-DNAs (d(CACGTG)2, d(CGTACG)2 and d(CGGCCG)2) in complex with hZαADAR1, 40 ns molecular dynamics simulation together with binding free energy calculation were performed for each system. For comparison, the standard CG-repeat Z-DNA (d(CGCGCG)2) complexed with hZαADAR1 was also simulated. The consistent results demonstrate that nonpolar interaction is the driving force during the protein-DNA binding process, and that polar interaction mainly from helix α3 also provides important contributions. Five common hot-spot residues were identified, namely Lys169, Lys170, Asn173, Arg174 and Tyr177. Hydrogen bond analysis coupled with surface charge distribution further reveal the interfacial information between hZαADAR1 and Z-DNA in detail. All of the analysis illustrate that four complexes share the common key features and the similar binding modes irrespective of Z-DNA sequences, suggesting that Z-DNA recognition by hZαADAR1 is conformation-specific rather than sequence-specific. Additionally, by analyzing the conformational changes of hZαADAR1, we found that the binding of Z-DNA could effectively stabilize hZαADAR1 protein. Our study can provide some valuable information for better understanding the binding mechanism between hZαADAR1 or even other Z-DNA-binding protein and Z-DNA.









Similar content being viewed by others
References
Rich A, Nordheim A, Wang AH (1984) The chemistry and biology of left-handed Z-DNA. Annu Rev Biochem 53:791–846
Ho PS (1994) The non-B-DNA structure of d (CA/TG)n does not differ from that of Z-DNA. Proc Natl Acad Sci U S A 91:9549–9553
Wang G, Vasquez KM (2007) Z-DNA, an active element in the genome. Front Biosci 12:4424–4438
Rothenburg S, Koch-Nolte F, Rich A, Haag F (2001) A polymorphic dinucleotide repeat in the rat nucleolin gene forms Z-DNA and inhibits promoter activity. Proc Natl Acad Sci U S A 98:8985–8990
Liu H, Mulholland N, Fu H, Zhao K (2006) Cooperative activity of BRG1 and Z-DNA formation in chromatin remodeling. Mol Cell Biol 26:2550–2559
Herbert A, Rich A (1996) The biology of left-handed Z-DNA. J Biol Chem 271:11595–11598
Herbert A, Rich A (1999) Left-handed Z-DNA: structure and function. Genetica 106:37–47
Ha SC, Kim D, Hwang HY, Rich A, Kim YG, Kim KK (2008) The crystal structure of the second Z-DNA binding domain of human DAI (ZBP1) in complex with Z-DNA reveals an unusual binding mode to Z-DNA. Proc Natl Acad Sci U S A 105:20671–20676
Ha SC (2004) A poxvirus protein forms a complex with left-handed Z-DNA: crystal structure of a Yatapoxvirus Zalpha bound to DNA. Proc Natl Acad Sci U S A 101:14367–14372
Schwartz T, Lowenhaupt K, Heinemann U, Rich A (2001) Structure of the DLM-1–Z-DNA complex reveals a conserved family of Z-DNA-binding proteins. Nat Struct Biol 8:761–765
Schade M, Turner CJ, Kuhne R, Schmieder P, Lowenhaupt K, Herbert A, Rich A, Oschkinat H (1999) The solution structure of the Zalpha domain of the human RNA editing enzyme ADAR1 reveals a prepositioned binding surface for Z-DNA. Proc Natl Acad Sci U S A 96:12465–12470
Schwartz T (1999) Crystal structure of the Z domain of the human editing enzyme ADAR1 bound to left-handed Z-DNA. Science 284:1841–1845
Herbert A, Schade M, Lowenhaupt K, Alfken J, Schwartz T, Shlyakhtenko LS, Lyubchenko YL, Rich A (1998) The Zalpha domain from human ADAR1 binds to the Z-DNA conformer of many different sequences. Nucleic Acids Res 26:3486–3493
Herbert AG, Rich A (1993) A method to identify and characterize Z-DNA binding proteins using a linear oligodeoxynucleotide. Nucleic Acids Res 21:2669–2672
Herbert A, Alfken J, Kim YG, Mian IS, Nishikura K, Rich A (1997) A Z-DNA binding domain present in the human editing enzyme, double-stranded RNA adenosine deaminase. Proc Natl Acad Sci U S A 94:8421–8426
Kim YG, Lowenhaupt K, Maas S, Herbert A, Schwartz T, Rich A (2000) The zab domain of the human RNA editing enzyme ADAR1 recognizes Z-DNA when surrounded by B-DNA. J Biol Chem 275:26828–26833
Kang YM, Bang J, Lee EH, Ahn HC, Seo YJ, Kim KK, Kim YG, Choi BS, Lee JH (2009) NMR spectroscopic elucidation of the B-Z transition of a DNA double helix induced by the Zalpha domain of human ADAR1. J Am Chem Soc 131:11485–11491
Lee YM, Kim HE, Park CJ, Lee AR, Ahn HC, Cho SJ, Choi KH, Choi BS, Lee JH (2012) NMR study on the B-Z junction formation of DNA duplexes induced by Z-DNA binding domain of human ADAR1. J Am Chem Soc 134:5276–5283
Lee EH, Seo YJ, Ahn HC, Kang YM, Kim HE, Lee YM, Choi BS, Lee JH (2010) NMR study of hydrogen exchange during the B-Z transition of a DNA duplex induced by the Zalpha domains of yatapoxvirus E3L. FEBS Lett 584:4453–4457
Ha SC, Choi J, Hwang HY, Rich A, Kim YG, Kim KK (2008) The structures of non-CG-repeat Z-DNAs co-crystallized with the Z-DNA-binding domain, hZ ADAR1. Nucleic Acids Res 37:629–637
Seo YJ, Ahn HC, Lee EH, Bang J, Kang YM, Kim HE, Lee YM, Kim K, Choi BS, Lee JH (2010) Sequence discrimination of the Zalpha domain of human ADAR1 during B-Z transition of DNA duplexes. FEBS Lett 584:4344–4350
Pan Y, Nussinov R (2006) Structural Basis for p53 binding-induced DNA bending. J Biol Chem 282:691–699
Wan H, Hu JP, Li KS, Tian XH, Chang S (2013) Molecular dynamics simulations of DNA-free and DNA-bound TAL effectors. PLoS One 8:e76045
Habtemariam B, Anisimov VM, MacKerell AD Jr (2005) Cooperative binding of DNA and CBFbeta to the Runt domain of the CBFalpha studied via MD simulations. Nucleic Acids Res 33:4212–4222
Case DA, Cheatham TE 3rd, Darden T, Gohlke H, Luo R, Merz KM Jr, Onufriev A, Simmerling C, Wang B, Woods RJ (2005) The Amber biomolecular simulation programs. J Comput Chem 26:1668–1688
Jorgensen WL, Chandrasekhar J, Madura JD, Impey RW, Klein ML (1983) Comparison of simple potential functions for simulating liquid water. J Chem Phys 79:926–935
Hornak V, Abel R, Okur A, Strockbine B, Roitberg A, Simmerling C (2006) Comparison of multiple Amber force fields and development of improved protein backbone parameters. Proteins 65:712–725
Ryckaert JP, Ciccotti G, Berendsen HJC (1977) Numerical integration of the cartesian equations of motion of a system with constraints: molecular dynamics of n-alkanes. J Comput Phys 23:327–341
Essmann U, Perera L, Berkowitz ML, Darden T, Lee H, Pedersen LG (1995) A smooth particle mesh Ewald method. J Chem Phys 103:8577–8593
Shen M, Zhou S, Li Y, Pan P, Zhang L, Hou T (2013) Discovery and optimization of triazine derivatives as ROCK1 inhibitors: molecular docking, molecular dynamics simulations and free energy calculations. Mol BioSyst 9:361–374
Hou T, Li N, Li Y, Wang W (2012) Characterization of domain-peptide interaction interface: prediction of SH3 domain-mediated protein-protein interaction network in yeast by generic structure-based models. J Proteome Res 11:2982–2995
Liu H, Yao X, Wang C, Han J (2010) In silico identification of the potential drug resistance sites over 2009 influenza A (H1N1) virus neuraminidase. Mol Pharm 7:894–904
Yang Y, Liu H, Yao X (2012) Understanding the molecular basis of MK2–p38α signaling complex assembly: insights into protein–protein interaction by molecular dynamics and free energy studies. Mol BioSyst 8:2106
Xue W, Qi J, Yang Y, Jin X, Liu H, Yao X (2012) Understanding the effect of drug-resistant mutations of HIV-1 intasome on raltegravir action through molecular modeling study. Mol BioSyst 8:2135
Tsui V, Case DA (2000) Theory and applications of the generalized Born solvation model in macromolecular simulations. Biopolymers 56:275–291
Hou T, Wang J, Li Y, Wang W (2011) Assessing the performance of the MM/PBSA and MM/GBSA methods. 1. The accuracy of binding free energy calculations based on molecular dynamics simulations. J Chem Inf Model 51:69–82
Hou T, Wang J, Li Y, Wang W (2011) Assessing the performance of the molecular mechanics/Poisson Boltzmann surface area and molecular mechanics/generalized Born surface area methods. II. The accuracy of ranking poses generated from docking. J Comput Chem 32:866–877
Srinivasan J, Cheatham TE, Cieplak P, Kollman PA, Case DA (1998) Continuum solvent studies of the stability of DNA, RNA, and phosphoramidate − DNA helices. J Am Chem Soc 120:9401–9409
Xu L, Sun H, Li Y, Wang J, Hou T (2013) Assessing the performance of MM/PBSA and MM/GBSA methods. 3. The impact of force fields and ligand charge models. J Phys Chem B 117:8408–8421
Sitkoff D, Sharp KA, Honig B (1994) Accurate calculation of hydration free energies using macroscopic solvent models. J Phys Chem 98:1978–1988
Cui Q, Sulea T, Schrag JD, Munger C, Hung MN, Naim M, Cygler M, Purisima EO (2008) Molecular dynamics-solvated interaction energy studies of protein-protein interactions: the MP1-p14 scaffolding complex. J Mol Biol 379:787–802
Gohlke H, Kiel C, Case DA (2003) Insights into protein-protein binding by binding free energy calculation and free energy decomposition for the Ras-Raf and Ras-RalGDS complexes. J Mol Biol 330:891–913
Chong LT, Duan Y, Wang L, Massova I, Kollman PA (1999) Molecular dynamics and free-energy calculations applied to affinity maturation in antibody 48G7. Proc Natl Acad Sci U S A 96:14330–14335
Shao J, Tanner SW, Thompson N, Cheatham TE (2007) Clustering molecular dynamics trajectories. 1. Characterizing the performance of different clustering algorithms. J Chem Theory Comput 3:2312–2334
Lou H, Cukier RI (2006) Molecular dynamics of apo-adenylate kinase: a principal component analysis. J Phys Chem B 110:12796–12808
Prompers JJ, Bruschweiler R (2002) Dynamic and structural analysis of isotropically distributed molecular ensembles. Proteins 46:177–189
Wang X, Xu X, Zhu S, Xiao Z, Ma Z, Li Y, Wang Y (2012) Molecular dynamics simulation of conformational heterogeneity in transportin 1. Proteins 80:382–397
Hu C, Zhang Y, Huang G, Zhang Q, Gui J (2004) Molecular cloning and characterization of a fish PKR-like gene from cultured CAB cells induced by UV-inactivated virus. Fish Shellfish Immunol 17:353–366
Kim YG, Muralinath M, Brandt T, Pearcy M, Hauns K, Lowenhaupt K, Jacobs RL, Rich A (2003) A role for Z-DNA binding in vaccinia virus pathogenesis. Proc Natl Acad Sci U S A 10:6974–6979
Kim YG, Lowenhaupt K, Oh DB, Kim KK, Rich A (2004) Evidence that vaccinia virulence factor E3L binds to Z-DNA in vivo: implications for development of a therapy for poxvirus infection. Proc Natl Acad Sci U S A 101:1514–1518
Garvie CW, Wolberger C (2001) Recognition of specific DNA sequences. Mol Cell 8:937–946
Rohs R, West SM, Sosinsky A, Liu P, Mann RS, Honig B (2009) The role of DNA shape in protein-DNA recognition. Nature 461:1248–1253
Acknowledgments
This work was supported by the National Natural Science Foundation of China (Grant No 21175063) and the Natural Science Foundation of Gansu Province, China (Grant No: 1208RJYA034).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(DOC 83 kb)
Rights and permissions
About this article
Cite this article
Wang, Q., Li, L., Wang, X. et al. Understanding the recognition mechanisms of Zα domain of human editing enzyme ADAR1 (hZαADAR1) and various Z-DNAs from molecular dynamics simulation. J Mol Model 20, 2500 (2014). https://doi.org/10.1007/s00894-014-2500-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00894-014-2500-5