Abstract
The intracellular protease from Pyrococcus horikoshii (PhpI) is a member of the DJ-1/ThiJ/PfpI superfamily, which is suggested to be involved in cellular protection against environmental stresses. In this study, flexible docking approach was employed to dock the ligand into the active site of PhpI. By analyzing the results, active site architecture and certain key residues responsible for substrate specificity were identified on the enzyme. Our docking result indicates that Glu12 plays an important role in substrate binding. The kinetic experiment conducted by Zhan shows that the E12T mutant is more stable than that of the wild-type. We also predict that Glu15, Lys43, and Tyr46 may be important in the catalytic efficiency and thermostability of enzyme. The new structural and mechanistic insights obtained from computational study should be valuable for detailed structures and mechanisms of the member of the DJ-1 superfamily.

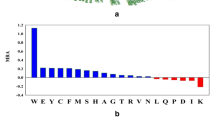
a AAFR-AMC in the a E12T b WT











Similar content being viewed by others
References
Malki A, Caldas T, Abdallah J, Kern R, Eckey V, Kim SJ, Cha SS, Mori H, Richarme G (2005) J Biol Chem 280:14420–14426
Mujacic M, Bader MW, Baneyx F (2004) Mol Micro 51:849–859
Quigley PM, Korotkov K, Baneyx F, Hol Wim GJ (2004) Protein Sci 13:269–272
Quigley PM, Korotkov K, Baneyx F, Hol Wim GJ (2003) Proc Natl Acad Sci USA 100:3137–3142
Du XL, Choi IG, Kim R, Wang W, Jancarik J, Yokota H, Kim SH (2000) Proc Natl Acad Sci USA 97:14079–14084
Abdallah J, Kern R, Malki A, Eckey, Richarme G (2006) Protein Expression Purif 47:455–460
Bonifati V, Rizzu P, Baren MJ, Schaap O, Breedveld GJ, Krieger E, Dekker MCJ, Squitieri F, Ibanez P, Joosse M, Dongen JW, Vanacore N, Swieten JC, Brice A, Meco G, Duijn CM, Oostra BA, Heutink P (2003) Science 299:256–259
Canet-Avilés RM, Wilson MA, Miller DW, Ahmad R, McLendon C, Bandyopadhyay S, Baptista MJ, Ringe D, Petsko GA, Cookson MR (2004) Proc Natl Acad Sci USA 101:9103–9108
Wilson MA, Collins JL, Hod Y, Ringe D, Petsko GA (2004) Proc Natl Acad Sci USA 100:9256–9261
Lee SJ, Kim SJ, Kim IK, Koo J, Jeong CS, Kim GH, Park C, Kang SO, Suh PG, Lee HS, Cha SS (2003) J Biol Chem 278:44552–44559
Lakshminarasimhan M, Maldonado MT, Zhou W, Fink AL, Wilson MA (2008) Biochemistry 47:1381–1392
Kazuya H, Nobuo NS, Masataka H, Takeshi N, Takahiro T, Hiroyoshi A, Fuyuhiko I (2003) J Biol Chem 278:31380–31384
Fioravanti E, Dura MA, Lascoux D, Micossi E, Franzetti B, McSweeney S (2008) Biochemistry 47:11581–11589
Bandyopadhyay S, Cookson MR (2004) BMC Evol Biol 4:1–9
Wilson MA, Amour CV, Collins JL, Ringe D, Petsko GA (2004) Proc Natl Acad Sci USA 101:1531–1536
Wei Y, Ringe D, Wilson MA, Ondrechen MJ (2007) PLoS Com Biol 3:0120–0126
da Costa C (2007) Curr Mol Med 7:650–657
Insight II, version 2000. San Diego, Accelrys Inc 2000
Affinity User Guide, Accelrys Inc, San Diego, USA 1999
Amber 9 users’ manual (2007) University of California, San Francisco
Jorgensen MJ, Chandrasekhar J, Madura JD, Impey RW, Klein ML (1983) J Chem Phys 79:926–935
Ryckaert JP, Ciccotti G, Berendsen HJC (1977) J Comput Phys 23:327–341
Bershein S, Tawfik DS (2008) Chem Biol 12151-158
Tkuriki N, Tawfik DS (2009) Science 324:203–207
Acknowledgments
This work was supported by the National High Technology Program of China (863 Program, 2006A020203) and the basic research fund of Jilin University (No. 200810019). We thank Dr. Dave Case for his kindness in offering us the Amber9 program (amber.scripps.edu) as a freeware.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Weiwei Han and Dongling Zhan contributed equally to this work.
Rights and permissions
About this article
Cite this article
Zhan, D., Han, W. & Feng, Y. Experimental and computational studies indicate the mutation of Glu12 to increase the thermostability of oligomeric protease from Pyrococcus horikoshii . J Mol Model 17, 1241–1249 (2011). https://doi.org/10.1007/s00894-010-0819-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00894-010-0819-0