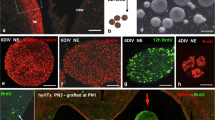
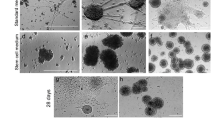
Abstract
Fetal onset hydrocephalus and abnormal neurogenesis are two inseparable phenomena turned on by a cell junction pathology first affecting neural stem/progenitor cells (NSPCs) and later the multiciliated ependyma. The neurological impairment of children born with hydrocephalus is not reverted by derivative surgery. NSPCs and neurosphere (NE) grafting into the cerebrospinal fluid (CSF) of hydrocephalic fetuses thus appears as a promising therapeutic procedure. There is little information about the cell lineages actually forming the NE as they grow throughout their days in vitro (DIV). Furthermore, there is no information on how good a host the CSF is for grafted NE. Here, we use the HTx rat, a model with hereditary hydrocephalus, with the mutation expressed in about 30% of the litter (hyHTx), while the littermates develop normally (nHTx). The investigation was designed (i) to establish the nature of the cells forming 4 and 6-DIV NE grown from NSPCs collected from PN1/nHTx rats and (ii) to study the effects on these NEs of CSF collected from nHTx and hyHTx. Immunofluorescence analyses showed that 90% of cells forming 4-DIV NEs were non-committed multipotential NSPCs, while in 6-DIV NE, 40% of the NSPCs were already committed into neuronal, glial and ependymal lineages. Six-DIV NE further cultured for 3 weeks in the presence of fetal bovine serum, CSF from nHTx or CSF from hyHTx, differentiated into neurons, astrocytes and βIV-tubulin+ multiciliated ependymal cells that were joined together by adherent junctions and displayed synchronized cilia beating. This supports the possibility that ependymal cells are born from subpopulations of NSC with their own time table of differentiation. As a whole, the findings indicate that the CSF is a supportive medium to host NE and that NE grafted into the CSF have the potential to produce neurons, glia and ependyma.






Similar content being viewed by others
Abbreviations
- AJ:
-
adherent junctions
- AQP4:
-
aquaporin 4
- BrdU:
-
bromodeoxyuridine
- CSF:
-
cerebrospinal fluid
- DIV:
-
days in vitro
- EGF:
-
epidermal growth factor
- FBS:
-
fetal bovine serum
- FGF:
-
fibroblast growth factor
- GFAP:
-
glial fibrillary acidic protein
- GJ:
-
gap junctions
- hyHTx:
-
hydrocephalic Texas rat
- NSPCs:
-
neural stem and progenitor cells
- NE:
-
neurospheres
- nHTX:
-
non-hydrocephalic Texas rat
- PN:
-
postnatal
- SVZ:
-
subventricular zone
- VZ:
-
ventricular zone
References
Ahn YS, Chang YS, Sung DK, Sung SI, Yoo HS, Lee HJ, Oh WI, Park WS (2013) Mesenchymal stem cells prevent hydrocephalus after severe intraventricular hemorrhage. Stroke. https://doi.org/10.1161/STROKEAHA.112.679092
Armstrong RJ, Svendsen N (2000) Neural stem cells: from cell biology to cell replacement. Cell Transplant 9:139–152
Bai H, Suzuki Y, Noda T, Wu S, Kataoka K, Kitada K, Ohta M, Chou H, Ide C (2003) Dissemination and proliferation of neural stem cells on injection into the fourth ventricle of the rat: a transplantation. J Neurosci Methods 124:181–187
Bátiz LF, Jimenez A, Guerra M, Rodríguez-Perez LM, Toledo CD, Vio K, Páez P, Pérez- Fígares JM, Rodrígue EM (2011) New ependymal cells are born postnatally in two discrete regions of the mouse brain and support ventricular enlargement in hydrocephalus. Acta Neuropathol 121:721–735. https://doi.org/10.1007/s00401-011-0799-x
Bez A, Corsini E, Curti D, Biggiogera M, Colombo A, Nicosia R, Pagano S, Parati A (2003) Neurosphere and neurosphere-forming cells: morphological and ultrastructural characterization. Brain Res 993:18–29. https://doi.org/10.1016/j.brainres.2003.08.061
Bonaguidi MA, Song J, Ming G, Song H (2012) A unifying hypothesis on mammalian neural stem cell properties in the adult hippocampus. Curr Opin Neurobiol 22:754–761. https://doi.org/10.1016/j.conb.2012.03.013
Buddensiek J, Dressel A, Kowalski M, Runge U, Schroeder H, Hermann A, Kirsch M, Storch A, Sabolek M (2010) Cerebrospinal fluid promotes survival and astroglial differentiation of adult human neural progenitor cells but inhibits proliferation and neuronal differentiation. BMC Neurosci 11:48. https://doi.org/10.1186/1471-2202-11-48
Bueno D, Garcia-Fernández J (2016) Evolutionary development of embryonic cerebrospinal fluid composition and regulation: an open research field with implications for brain development and function. Fluids Barriers CNS 13:5. https://doi.org/10.1186/s12987-016-0029-y
Del Bigio MR (2001) Pathophysiologic consequences of hydrocephalus. Neurosurg Clin N Am 12:639–649
Del Bigio MR (2010) Neuropathology and structural changes in hydrocephalus. Devel Dis Res Rev 16:16–22. https://doi.org/10.1002/ddrr.94
Fuchs E, Segre JA (2000) Stem cells: a new lease on life. Cell 100:143–155. https://doi.org/10.1016/S0092-8674(00)81691-8
Gato A, Alonso MI, Martin C, Carnicero E, Moro JA, De la Mano A, Fernandez JM, Lamus F, Desmond ME (2014) Embryonic cerebrospinal fluid in brain development: neural progenitor control. Croat Med J 55:299–305. https://doi.org/10.3325/cmj
Gato A, Moro JA, Alonso MI, Bueno D, De La Mano A, Martín C (2005) Embryonic cerebrospinal fluid regulates neuroepithelial survival, proliferation, and neurogenesis in chick embryos. Anat Rec A Discov Mol Cell Evol Biol 284:475–484. https://doi.org/10.1002/ar.a.20185
Ge D, Song K, Guan S, Dai M, Ma X, Liu T, Cui Z (2012) Effect of the neurosphere size on the viability and metabolism of neural stem/progenitor cells. African J Biotech 11:3976–3985. https://doi.org/10.5897/AJB11.3324
Gil-Perotín S, Duran-Moreno M, Cebrián-Silla A, Ramírez M, García-Belda P, García-Verdugo JM (2013) Adult neural stem cells from the subventricular zone: a review of the neurosphere assay. Anat Rec 296:1435–1452. https://doi.org/10.1002/ar.22746
Guerra M, Henzi R, Ortloff A, Lichtin N, Vío K, Jiménez A, Dominguez-Pinos MD, González C, Jara MC, Hinostroza F, Rodríguez S, Jara M, Ortega E, Guerra F, Sival DA, den Dunnen W, Pérez-Figares JM, McAllister JP, Johanson CE, Rodríguez EM (2015a) A cell junction pathology of neural stem cells is associated with ventricular zone disruption, hydrocephalus, and abnormal neurogenesis. J Neuropathol Exp Neurol 74:653–671. https://doi.org/10.1097/NEN.0000000000000203
Guerra MM, González C, Caprile T, Jara M, Vío K, Muño RI, Rodríguez S, Rodríguez EM (2015b) Understanding how the subcommissural organ and other periventricular secretory structures contribute via the cerebrospinal fluid to neurogenesis. Front Cell Neurosc 9:480. https://doi.org/10.3389/fncel.2015.00480
Jones HC, Bucknall RM (1987) Changes in cerebrospinal fluid pressure and outflow from the lateral ventricles during development of congenital hydrocephalus in the H-Tx rat. Exper Neurol 98:573–583. https://doi.org/10.1016/0014-4886(87)90266-4
Kim DE, Schellingerhout D, Ishii K, Shah K, Weissleder R (2004) Imaging of stem cell recruitment to ischemic infarcts in a murine model. Stroke 35:952–957. https://doi.org/10.1161/01.STR.0000120308.21946.5D
Lehtinen MK, Zappaterra MW, Chen X, Yang YJ, Hil A, Lun M, Maynard T, Gonzalez D, Kim S, Ye P, D’Ercole AJ, Wong ET, LaMantia AS, Walsh CA (2011) The cerebrospinal fluid provides a proliferative niche for neural progenitor cells. Neuron 69:893–905. https://doi.org/10.1016/j.neuron.2011.01.023
Lobo M, Alonso F, Redondo C, López-Toledano M, Caso E, Herranz A, Paíno C, Reimers D, Bazán E (2003) Cellular characterization of epidermal growth factor-expanded free-floating neurospheres. J Histoch Cytoch 51:89–103. https://doi.org/10.1177/002215540305100111
Marshall GP, Ross HH, Suslov O, Zheng T, Steindler D, Laywell E (2008) Production of neurospheres from CNS tissue. Meth Mol Biol 438:135–150 doi:https://doi.org/10.1007/978-1-59745-133-812
McAllister JP (2012) Pathophysiology of congenital and neonatal hydrocephalus. Sem Fet Neonat Med 17:285–294. https://doi.org/10.1016/j.siny.2012.06.004
McAllister JP, Guerra MM, Castaneyra Ruiz L, Jimenez A, Dominguez-Pinos MD, Sival D, den Dunnen W, Morales D, Schmidt R, Rodríguez EM, Limbrick D (2017) Ventricular zone disruption in human neonates with intraventricular hemorrhage. J Neurop Exper Neurol 76:358–375. https://doi.org/10.1093/jnen/nlx017
Mokrý J, Karbanová J (2006) Foetal mouse neural stem cells give rise to ependymal cells in vitro. Fol Biol (Praha) 52:149–155
Mollenhauer HH (1964) Plastic embedding mixtures for use in electron microscopy. Stain Technol 39:111–114
Monni E, Congiu T, Massa D, Nat R, Diana A (2011) Human neurospheres: from stained sections to three-dimensional assembly. Transl Neurosci 2:43–48. https://doi.org/10.2478/s13380-011-0007-4
Neuhuber B, Barshinger AL, Paul C, Shumsky JS, Mitsui T, Fischer I (2008) Stem cell delivery by lumbar puncture as a therapeutic alternative to direct injection into injured spinal cord. J Neurosurg Spine 9:390–399. https://doi.org/10.3171/SPI.2008.9.10.390
Ohta M, Yoshihisa S, Toru N, Kazuya K, Hirotomi C, Namiko I, Masaaki K, Matsumoto N, Dezawa M, Suzuki S, Ide C (2004) Implantation of neural stem cells via cerebrospinal fluid into the injured root. Neuroreport 15:1249–1253. https://doi.org/10.1097/01.wnr.0000129998.72184.e1
Ortega E, Muñoz RI, Luza N, Guerra F, Guerra M, Vio K, Henzi R, Jaque RS, McAllister JP, Rodrigue EM (2016) The value of early and comprehensive diagnoses in a human fetus with hydrocephalus and progressive obliteration of the aqueduct of sylvius: case report. BBMC Neurology 16:45. https://doi.org/10.1186/s12883-016-0566-7
Ortloff AR Vío K, Guerra M, Jaramillo K, Kaehne T, Jones H, McAllister JP, Rodríguez EM (2013) Role of the subcommissural organ in the pathogenesis of congenital hydrocephalus in the HTx rat. Cell Tiss Res 352:707–725. https://doi.org/10.1007/s00441-013-1615-9
Páez P, Bátiz LF, Roales-Buján R, Rodríguez-Pérez LM, Rodríguez S, Jiménez AJ, Rodríguez EM, Pérez-Fígare JM (2007) Patterned neuropathologic events occurring in hyh congenital hydrocephalic mutant mice. J Neuropathol Exper Neurol 66:1082–1092. https://doi.org/10.1097/nen.0b013e31815c1952
Pandamooz S, Mohammad N, Alinezhad F, Zarghami A, Pourghasem M (2013) The influence of cerebrospinal fluid on epidermal neural crest stem cells may pave the path for cell-based therapy. Stem Cell Res Ther 4(4):84. https://doi.org/10.1186/scrt235
Parada C, Gato A, Aparicio M, Bueno D (2006) Proteome analysis of chick embryonic cerebrospinal fluid. Proteomics 6:312–320. https://doi.org/10.1002/pmic.200500085
Pluchino S, Quattrini A, Brambilla E, Gritti E, Salani G, Dina G, Galli R, Del Carro U, Amadio S, Bergami A, Furlan R, Comi G, Vescovi AL, Martino G (2003) Injection of adult neurospheres induces recovery in a chronic model of multiple sclerosis. Nature 422:688–694. https://doi.org/10.1038/nature01552
Reynolds BA, Weiss S (1992) Generation of neurons and astrocytes from isolated cells of the adult mammalian central nervous system. Science (New York, NY) 255:1707–1710. https://doi.org/10.1126/science.1553558
Reynolds BA, Weiss (1996) Clonal and population analyses demonstrate that an EGF-responsive mammalian embryonic CNS precursor is a stem cell. Dev Biol 175(1):1–13. https://doi.org/10.1006/dbio.1996.0090
Reynolds BA, Rietze R (2005) Neural stem cells and neurospheres. Re-evaluating the relationship. Nature Meth 2:333–336. https://doi.org/10.1038/nmeth58
Rietze RL, Valcanis H, Brooker GF, Thomas T, Voss K, Bartlett PF (2001) Purification of a pluripotent neural stem cell from the adult mouse brain. Nature 412:736–739. https://doi.org/10.1038/35089085
Rodriguez EM (1976) The cerebrospinal fluid as a pathway in neuroendocrine integration. J Endocrinol 71:407–443. https://doi.org/10.1677/JOE.0.0710407
Rodriguez EM, Guerra MM (2017) Neural stem cells and foetal-onset hydrocephalus. Pediatr Neurosurg 52:446–461. https://doi.org/10.1159/000453074
Rodríguez EM, Guerra MM, Vío K, González C, Ortloff A, Bátiz LF, Rodríguez S, Jara MC, Munoz RI, Ortega E, Jaque J, Guerra F, Sival DA, den Dunnen WFA, Jimenez A, Dominguez-Pinos MD, Pérez-Figares JM, McAllister JP, Johanson C (2012) A cell junction pathology of neural stem cells leads to abnormal neurogenesis and hydrocephalus. Biol Res 45:231–241. https://doi.org/10.4067/S0716-97602012000300005
Satake K, Lou J, Lenke LG (2004) Migration of mesenchymal stem cells through cerebrospinal fluid into injured spinal cord tissue. Spine 29:1971–1979. https://doi.org/10.1097/01.brs.0000138273.02820.0a
Seaberg RM, van der Kooy D (2002) Adult rodent neurogenic regions: the ventricular subependyma contains neural stem cells, but the dentate gyrus contains restricted progenitors. Journal Neurosc 22:1784–93 doi:22/5/1784 [pii] ET - 2002/03/07
Sival DA, Guerra MM, den Dunnen WFA, Bátiz LF, Alvial G, Castañeyra-Perdomo A, Rodríguez EM (2011) Neuroependymal denudation is in progress in full-term human foetal spina bifida aperta. Brain Pathol 21:163–179. https://doi.org/10.1111/j.1750-3639.2010.00432.x
Slovinska L, Szekiova E, Blasko J, Devaux S, Salzet M, Cizkova D (2015) Comparison of dynamic behavior and maturation of neural multipotent cells derived from different spinal cord developmental stages: an in vitro study. Acta Neurobiol Exp 75:107–114
Suslov ON, Kukekov VG, Ignatova TN, Steindler DA (2002) Neural stem cell heterogeneity demonstrated by molecular phenotyping of clonal neurospheres. Proc Nat Acad Sci 99:14506–14511. https://doi.org/10.1073/pnas.212525299
Talaverón R, Fernández P, Escamilla R, Pastor AM, Matarredona ER, Sáez JC (2015) Neural progenitor cells isolated from the subventricular zone present hemichannel activity and form functional gap junctions with glial cells. Front Cell Neurosci 9:411–422. https://doi.org/10.3389/fncel.2015.00411
Temple S (2001) The development of neural stem cells. Nature 414:112–117
Vio K, Rodríguez S, Yulis CR, Oliver C, Rodriguez EM (2008) The subcommissural organ of the rat secretes Reissner’s fiber glycoproteins and CSF-soluble proteins reaching the internal and external CSF compartments. Cerebr Fluid Res 5:1–14. https://doi.org/10.1186/1743-8454-5-3
Wagner C, Batiz LF, Rodriguez S, Jiménez AJ, Páez P, Tomé M, Pérez-Fígares JM, Rodríguez EM (2003) Cellular mechanisms involved in the stenosis and obliteration of the cerebral aqueduct of hyh mutant mice developing congenital hydrocephalus. J Neuropathol Exp Neurol 62:1019–1040
Wang TY, Sen A, Behie LA, Kallos MS (2006) Dynamic behavior of cells within neurospheres in expanding populations of neural precursors. Brain Res 1107:82–96. https://doi.org/10.1016/j.brainres.2006.05.110
Weiss S, Reynolds BA, Vescovi A, Morshead C, Craig CG, van der Kooy D (1996) Is there a neural stem cell in the mammalian forebrain? Trends Neurosc 19:387–393. https://doi.org/10.1016/S0166-2236(96)10035-7
Williams MA, McAllister JP, Walker ML (2007) Priorities for hydrocephalus research: report from a National Institutes of Health-sponsored workshop. J Neurosurg 107:345–357
Wu S, Suzuki Y, Noda Y, Bai H, Kitada M, Kataoka K, Nishimura Y, Ide C (2002) Immunohistochemical and electron microscopic study of invasion and differentiation in spinal cord lesion of neural stem cells grafted through cerebrospinal fluid in rat. J Neurosc Res 69:940–945. https://doi.org/10.1002/jnr.10346
Zappaterra MD, Lisgo SN, Lindsay S, Gygi SP, Walsh CA, Ballif BA (2007) A comparative proteomic analysis of human and rat embryonic cerebrospinal fluid. J Prot Res 6:3537–3548. https://doi.org/10.1021/Pr07047w2
Zhu M, Feng Y, Dangelmajer F, Guerrero-Cazares H, Chaichana KL, Smith CL, Levchenko A, Lei T, Quinones-Hinojosa A (2015) Human cerebrospinal fluid regulates proliferation and migration of stem cells through insulin-like growth factor-1. Stem Cells Dev 24:160–171. https://doi.org/10.1089/scd.2014.0076
Acknowledgements
The authors wish to acknowledge the valuable technical support of Mrs Clara Jara, and Confocal and Electron Microscopy Core Facilities of Universidad Austral de Chile. Monoclonal antibodies against nestin and BrdU were obtained from the Developmental Studies Hybridoma Bank developed under the auspices of the NICHD and maintained by The University of Iowa, Department of Biological Sciences, Iowa City, Iowa.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supported by Fondecyt 1111018 to EMR; Hydrocephalus Association Established Investigator Award No. 51002705 to PM, EMR, CJ; Doctoral CONICYT Fellowship (Chile) to RH.
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Henzi, R., Guerra, M., Vío, K. et al. Neurospheres from neural stem/neural progenitor cells (NSPCs) of non-hydrocephalic HTx rats produce neurons, astrocytes and multiciliated ependyma: the cerebrospinal fluid of normal and hydrocephalic rats supports such a differentiation. Cell Tissue Res 373, 421–438 (2018). https://doi.org/10.1007/s00441-018-2828-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00441-018-2828-8