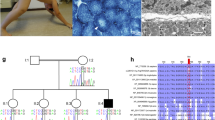
Abstract
Congenital myopathies are a heterogeneous group of muscle disorders that are often genetically determined. Here, we investigated a boy with congenital myopathy, deafness, and neuropathy from a consanguineous Kurdish family by autozygosity mapping and whole exome sequencing. We found a homozygous nonsense mutation in SPTBN4 [c.1597C>T, NM_020971.2; p.(Q533*), NP_066022.2; ClinVar SUB2292235] encoding βIV-spectrin, a non-erythrocytic member of the β-spectrin family. Western blot confirmed the absence of the full-length 288 kDa isoform in muscle and of a specific 72 kDa isoform in fibroblasts. Clinical symptoms of the patient largely corresponded to those described for the quivering mouse, a loss-of-function animal model. Since the human phenotype of βIV-spectrin deficiency included a myopathy with incomplete congenital fiber-type disproportion, we investigated muscle of the quivering (qv4J) mouse and found complete absence of type 1 fibers (fiber-type 2 uniformity). Immunohistology confirmed expression of βIV-spectrin in normal human and mouse muscle at the sarcolemma and its absence in patient and quivering (qv4J) mouse. SPTBN4 mRNA-expression levels in healthy skeletal muscle were found in the range of other regulatory proteins. More patients have to be described to confirm the triad of congenital myopathy, neuropathy and deafness as the defining symptom complex for βIV-spectrin deficiency.



Similar content being viewed by others
References
Bock GR, Steel KP (1983) Inner ear pathology in the deafness mutant mouse. Acta Otolaryngol (Stockh) 96:39–47. doi:10.3109/00016488309132873
Clarke NF (2011) Congenital fiber-type disproportion. Semin Pediatr Neurol 18:264–271. doi:10.1016/j.spen.2011.10.008
Deol MS, Frank MP, Steel KP, Bock GR (1983) Genetic deafness of central origin. Brain Res 258:177–179. doi:10.1016/0006-8993(83)91248-9
DePristo MA, Banks E, Poplin R et al (2011) A framework for variation discovery and genotyping using next-generation DNA sequencing data. Nat Genet 43:491–498. doi:10.1038/ng.806
Devaux JJ (2010) The C-terminal domain of βIV-spectrin is crucial for KCNQ2 aggregation and excitability at nodes of Ranvier. J Physiol 588:4719–4730. doi:10.1113/jphysiol.2010.196022
Dobin A, Davis CA, Schlesinger F et al (2013) STAR: ultrafast universal RNA-seq aligner. Bioinformatics 29:15–21. doi:10.1093/bioinformatics/bts635
Hedstrom KL, Rasband MN (2006) Intrinsic and extrinsic determinants of ion channel localization in neurons. J Neurochem 98:1345–1352. doi:10.1111/j.1471-4159.2006.04001.x
Hund TJ, Koval OM, Li J et al (2010) A βIV-spectrin/CaMKII signaling complex is essential for membrane excitability in mice. J Clin Investig 120:3508–3519. doi:10.1172/JCI43621
Komada M, Soriano P (2002) βIV-spectrin regulates sodium channel clustering through ankyrin-G at axon initial segments and nodes of Ranvier. J Cell Biol 156:337–348. doi:10.1083/jcb.200110003
Li H (2013) Aligning sequence reads, clone sequences and assembly contigs with BWA-MEM. http://arxiv.org/pdf/1303.3997.pdf. Accessed Sept 3 2015
Maggi L, Scoto M, Cirak S et al (2013) Congenital myopathies—Clinical features and frequency of individual subtypes diagnosed over a 5-year period in the United Kingdom. Neuromuscul Disord 23:195–205. doi:10.1016/j.nmd.2013.01.004
North KN, Wang CH, Clarke N et al (2014) Approach to the diagnosis of congenital myopathies. Neuromuscul Disord 24:97–116. doi:10.1016/j.nmd.2013.11.003
Parkinson NJ, Olsson CL, Hallows JL et al (2001) Mutant β-spectrin 4 causes auditory and motor neuropathies in quivering mice. Nat Genet 29:61–65. doi:10.1038/ng710
Pfaffl MW (2001) A new mathematical model for relative quantification in real-time RT–PCR. Nucleic Acids Res 29:e45–e45. doi:10.1093/nar/29.9.e45
Relizani K, Mouisel E, Giannesini B et al (2014) Blockade of ActRIIB signaling triggers muscle fatigability and metabolic myopathy. Mol Ther 22:1423–1433. doi:10.1038/mt.2014.90
Schwarz JM, Cooper DN, Schuelke M, Seelow D (2014) MutationTaster2: mutation prediction for the deep-sequencing age. Nat Methods 11:361–362. doi:10.1038/nmeth.2890
Seelow D, Schuelke M (2012) HomozygosityMapper2012–bridging the gap between homozygosity mapping and deep sequencing. Nucleic Acids Res 40:W516–W520. doi:10.1093/nar/gks487
Trapnell C, Roberts A, Goff L et al (2012) Differential gene and transcript expression analysis of RNA-seq experiments with TopHat and Cufflinks. Nat Protoc 7:562–578. doi:10.1038/nprot.2012.016
Tse WT, Tang J, Jin O et al (2001) A new spectrin, βIV, has a major truncated isoform that associates with promyelocytic leukemia protein nuclear bodies and the nuclear matrix. J Biol Chem 276:23974–23985. doi:10.1074/jbc.M009307200
Yang Y, Lacas-Gervais S, Morest DK et al (2004) βIV Spectrins are essential for membrane stability and the molecular organization of nodes of ranvier. J Neurosci 24:7230–7240. doi:10.1523/JNEUROSCI.2125-04.2004
Yao Z, Snider L, Balog J et al (2014) DUX4-induced gene expression is the major molecular signature in FSHD skeletal muscle. Hum Mol Genet 23:5342–5352. doi:10.1093/hmg/ddu251
Acknowledgements
The authors would like to thank the patient and his parents for participation in the study.
Author information
Authors and Affiliations
Contributions
MS, EK phenotyped the patient and gathered clinical information and material; SMG performed cell culture experiments; EG, FS performed molecular genetics experiments; WS performed muscle histology; SDU, TJH provided and prepared the muscle samples of the quivering (qv4J) mouse; MS performed homozygosity mapping, sequence alignment, and bioinformatic analysis of the WES data. EK analyzed and validated the results of the WES data. EK, MS co-authored the first draft of the manuscript and contributed funding. All authors read the final version of the manuscript for intellectual content and gave their permission for publication.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Funding statement
The project was funded by the Charité-Universitätsmedizin Berlin via the “Rahel-Hirsch” Program to EK, the Deutsche Forschungsgemeinschaft (SFB 665 TP C4) to MS, and the NeuroCure Center of Excellence (Exc 257) to MS.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Knierim, E., Gill, E., Seifert, F. et al. A recessive mutation in beta-IV-spectrin (SPTBN4) associates with congenital myopathy, neuropathy, and central deafness. Hum Genet 136, 903–910 (2017). https://doi.org/10.1007/s00439-017-1814-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00439-017-1814-7