Abstract
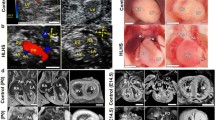
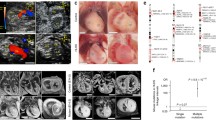
Hypoplastic left heart syndrome (HLHS) is a severe congenital heart defect (CHD) that necessitates staged, single ventricle surgical palliation. An increased frequency of bicuspid aortic valve (BAV) has been observed among relatives. We postulated number of mutant alleles as a molecular basis for variable CHD expression in an extended family comprised of an HLHS proband and four family members who underwent echocardiography and whole-genome sequencing (WGS). Dermal fibroblast-derived induced pluripotent stem cells (iPSC) were procured from the proband–parent trio and bioengineered into cardiomyocytes. Cardiac phenotyping revealed aortic valve atresia and a slit-like left ventricular cavity in the HLHS proband, isolated bicuspid pulmonary valve in his mother, BAV in a maternal 4° relative, and no CHD in his father or sister. Filtering of WGS for rare, functional variants that segregated with CHD and were compound heterozygous in the HLHS proband identified NOTCH1 as the sole candidate gene. An unreported missense mutation (P1964L) in the cytoplasmic domain, segregating with semilunar valve malformation, was maternally inherited and a rare missense mutation (P1256L) in the extracellular domain, clinically silent in the heterozygous state, was paternally inherited. Patient-specific iPSCs exhibited diminished transcript levels of NOTCH1 signaling pathway components, impaired myocardiogenesis, and a higher prevalence of heterogeneous myofilament organization. Extended, phenotypically characterized families enable WGS-derived variant filtering for plausible Mendelian modes of inheritance, a powerful strategy to discover molecular underpinnings of CHD. Identification of compound heterozygous NOTCH1 mutations and iPSC-based functional modeling implicate mutant allele burden and impaired myogenic potential as mechanisms for HLHS.




Similar content being viewed by others
References
Abecasis GR, Cherny SS, Cookson WO, Cardon LR (2002) Merlin-rapid analysis of dense genetic maps using sparse gene flow trees. Nat Genet 30:97–101. doi:10.1038/ng786
Adzhubei IA, Schmidt S, Peshkin L, Remensky VE, Gerasimova A, Bork P, Kondrashov AS, Sunyaev SR (2010) A method and server for predicting damaging missense mutations. Nat Method 7:248–249. doi:10.1038/nmeth0410-248
Danacek P, Auton A, Abecasis G, Albers CA, Banks E, DePristo MA, Handsaker RE, Lunter G, Marth GT, Sherry ST, McVean G, Durbin R, 1000 Genomes Project Analysis Group (2011) The variant call format and VCF tools. Bioinformatics 27:2156–2158. doi:10.1093/bioinformatics/btr330
DePristo MA, Banks E, Poplin RE, Garimella KV, Maguire JR, Hartl C, Philippakis AA, del Angel G, Rivas MA, Hanna M, McKenna A, Fennell TJ, Kernytsky AM, Sivachenko AY, Cibulskis K, Gabriel SB, Altshuler D, Daly MJ (2011) A framework for variation discovery and genotyping using next-generation DNA sequencing data. Nat Genet 43:491–498. doi:10.1038/ng.806
Ehebauer MT, Chirgadze DY, Hayward P, Martinez Arias A, Blundell TL (2005) High-resolution crystal structure of the human Notch 1 ankyrin domain. Biochem J 392:13–20. doi:10.1042/BJ20050515
Exome Aggregation Consortium (ExAC), Cambridge, MA (2015). http://exac.broadinstitute.org
Exome Variant Server (2014) NHLBI GO Exome Sequencing Project (ESP), Seattle, WA. http://evs.gs.washington.edu/EVS
Foffa I, Ait Ali L, Panesi P, Mariani M, Festa P, Botto N, Vecoli C, Andreassi MG (2014) Sequencing of NOTCH1, GATA5, TGFBR1 and TGFBR2 genes in familial cases of bicuspid aortic valve. BMC Med Genet 16:1–9. doi:10.1186/1471-2350-14-44
Garg V, Muth AN, Ransom JF, Schluterman MK, Barnes R, King IN, Grossfeld PD, Srivastava D (2005) Mutations in NOTCH1 cause aortic valve disease. Nature 437:270–274. doi:10.1038/nature03940
Gelb BD, Chung WK (2014) Complex genetics and etiology of human congenital heart disease. Cold Spring Harb Perspect Med 4:1–12. doi:10.1101/cshperspect.a013953
Grego-Bessa J, Luna-Zurita L, del Monte G, Bolos V, Melgar P, Arandilla A, Garratt AN, Zang H, Mukouyama Y, Chen H, Shou W, Ballestar E, Esteller M, Rojas A, Perez-Pomares JM, de la Pompa JL (2007) Notch signaling is essential for ventricular chamber development. Dev Cell 12:415–429. doi:10.1016/j.devcel.2006.12.011
High FA, Epstein JA (2008) The multifaceted role of Notch in cardiac development and disease. Nature Rev 9:49–61. doi:10.1038/nrg2279
Iascone M, Ciccone R, Galletti L, Marchetti D, Seddio F, Lincesso AR, Pezzoli L, Vetro A, Barachetti D, Boni L, Federici D, Soto AM, Comas JV, Ferrazzi P, Zuffardi O (2012) Identification of de novo mutation and rare variants in hypoplastic left heart syndrome. Clin Genet 81:542–554. doi:10.1111/j.1399-0004.2011.01674
Jiang Y, Habibollah S, Tilgner K, Collin J, Barta T, Al-Aama JY, Tesarov L, Hussain R, Trafford AW, Kirkwood G, Sernagor E, Eleftheriou CG, Przyborski S, Stojkovic M, Lako M, Keavney B, Armstrong L (2014) An induced pluripotent stem cell model of hypoplastic left heart syndrome reveals multiple expression and functional differences in HLHS-derived cardiac myocytes. Stem Cells Transl Med 3:416–423. doi:10.5966/sctm.2013-0105
Kobayashi J, Yoshida M, Tarui S, Hirata M, Negai Y, Kasahara S, Naruse K, Ito H, Sano S, Oh H (2014) Directed differentiation of patient-specific induced pluripotent stem cells identifies the transcriptional repression and epigenetic modification of NKX2-5, HAND1, and NOTCH1 in hypoplastic left heart syndrome. PLoS One 9:1–14. doi:10.1371/journal.pone.0102796
Loffredo CA, Chokkalingam A, Sill AM, Boughman JA, Clark EB, Scheel J, Brenner JI (2004) Prevalence of congenital cardiovascular malformations among relatives of infants with hypoplastic left heart, coarctation of the aorta, and d-transposition of the great arteries. Am J Med Genet A 124A:225–230. doi:10.1002/ajmg.a.20366
Matise TC, Chen F, Chen W, De La Vega FM, Hansen M, He C, Hyland FCL, Kennedy GC, Kong X, Murray SS, Ziegle JS, Stewart WCL, Buyske S (2007) A second-generation combined linkage-physical map of the human genome. Genome Res 17:1783–1786. doi:10.1101/gr.7156307
McBride KL, Pignatelli R, Lewin M, Ho T, Fernbach S, Menesses A, Lam W, Leal SM, Kaplan N, Schliekelman P, Towbin JA, Belmont JW (2005) Inheritance analysis of congenital left ventricular outflow tract obstruction malformations: segregation, multiplex relative risk, and heritability. Am J Med Genet A 134A:180–186. doi:10.1002/ajmg.a.30602
McBride KL, Riley MF, Zender GA, Fitzgerald-Butt SM, Towbin JA, Belmont JW, Cole SE (2008) NOTCH1 mutations in individuals with left ventricular outflow tract malformations reduce ligand-induced signaling. Hum Mol Genet 17:2886–2893. doi:10.1093/hmg/ddn187
McKellar SH, Tester DJ, Yagubyan M, Majumdar R, Ackerman MJ, Sundt TM 3rd (2007) Novel NOTCH1 mutations in patients with bicuspid aortic valve disease and thoracic aortic aneurysms. J Thorac Cardiovasc Surg 134:290–296. doi:10.1016/j.jtcvs.2007.02.041
McKenna A, Hanna M, Banks E, Sivachenko A, Cibulskis K, Kernytsky A, Garimella K, Altshuler D, Gabriel S, Daly M, DePristo MA (2010) The genome analysis toolkit: a MapReduce framework for analyzing next-generation DNA sequencing data. Genome Res 20:1297–1303. doi:10.1101/gr.107524.110
Mohamed SA, Aherrahrou Z, Liptau H, Erasmi AW, Hagemann C, Wrobel S, Borzym K, Schunkert H, Sievers HH, Erdmann J (2006) Novel missense mutations (p. T596M and p.P1797H) in NOTCH1 in patients with bicuspid aortic valve. Biochem Biophys Res Commun 345:1460–1465. doi:10.1016/j.bbrc.2006.05.046
Ng PC, Henikoff S (2001) Predicting deleterious amino acid substitutions. Genome Res 11:863–874. doi:10.1101/gr.176601
Norwood WI, Lang P, Hansen DD (1983) Physiologic repair of aortic atresia-hypoplastic left heart syndrome. N Engl J Med 308:23–26. doi:10.1056/NEJM198301063080106
Petrovski S, Wang Q, Heinzen EL, Allen AS, Goldstein DB (2013) Genic intolerance to functional variation and the interpretation of personal genomes. PLoS Genet 9:1–13. doi:10.1371/journal.pgen.1003709
Purcell S, Neale B, Todd-Brown K, Thomas L, Ferreira MAR, Bender D, Maller J, Sklar P, de Bakker PIW, Dal MJ, Sham PC (2007) PLINK: a tool set for whole-genome association and population-based linkage analysis. Am J Hum Genet 81:559–575. doi:10.1086/519795
Raney BJ, Dreszer TR, Barber GP, Clawson H, Fujita PA, Wang T, Nguyen N, Paten B, Zweig AS, Karolchik D, Kent WJ (2014) Track data hubs enable visualization of user-defined genome-wide annotations on the UCSC genome browser. Bioinformatics 30:1003–1005. doi:10.1093/bioinformatics/btt637
Riley MF, McBride KL, Cole SE (2011) NOTCH1 missense alleles associated with left ventricular outflow tract defects exhibit impaired receptor processing and defective EMT. Biochim Biophys Acta 1812:121–129. doi:10.1016/j.bbadis.2010.10.002
Robinson JT, Thorvaldsodottir Winckler W, Guttman M, Lander ES, Getz G, Mesirov JP (2011) Integrative genomics viewer. Nat Biotechnol 33:24–26. doi:10.1038/nbt.1754
Sun N, Yazawa M, Liu J, Han L, Sanchez-Freire V, Abilez OJ, Navarrete EG, Hu S, Wang L, Lee A, Pavlovic A, Lin S, Chen R, Hajjar RJ, Snyder MP, Dolmetsch RE, Butte MJ, Ashley EA, Longaker MT, Robbins RC, Wu JC (2012) Patient-specific induced pluripotent stem cells as a model for familial dilated cardiomyopathy. Sci Transl Med 4:130ra47. doi:10.1126/scitranslmed.3003552
Swiatek PJ, Lindsell CE, del Amo FF, Weinmaster G, Gridley T (1994) Notch1 is essential for postimplantation\development in mice. Genes Dev 8:707–719. doi:10.1101/gad.8.6.707
The 1000 Genomes Consortium (2012) An integrated map of genetic variation from 1092 human genomes. Nature 491:56–65. doi:10.1038/nature11632
Timmerman LA, Grego-Bessa J, Raya A, Bertran E, Perez-Pomares JM, Diez J, Aranda S, Palomo S, McCormick F, Izpisua-Belmonte JC, de la Pompa JL (2004) Notch promotes epithelial-mesenchymal transition during cardiac development and oncogenic transformation. Genes Dev 18:99–115. doi:10.1101/gad.276304
Wessels MW, Berger RM, Frohn-Mulder IM, Roos-Hesselink JW, Hoogeboom JJ, Mancini GS, Bartelings MM, Krijger RD, Wladimiroff JW, Niermeijer MF, Grossfeld P, Willems PJ (2005) Autosomal dominant inheritance of left ventricular outflow tract obstruction. Am J Med Genet A 134A:171–179. doi:10.1002/ajmg.a.30601
Yu X, Zou J, Ye Z, Hammond H, Chen G, Tokunaga A, Mali P, Li YM, Civin C, Gaiano N, Cheng L (2008) Notch signaling activation in human embryonic stem cells is required for embryonic, but not trophoblastic, lineage commitment. Cell Stem Cell 2:461–471. doi:10.1016/j.stem.2008.03.001
Acknowledgments
The authors gratefully acknowledge the patients and family members who participated in this study.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Informed consent
Informed consent was obtained from all individual participants included in the study.
Funding
Todd and Karen Wanek Family Program for Hypoplastic Left Heart Syndrome, Mayo Clinic Foundation.
Conflict of interest
Mayo Clinic and Timothy J. Nelson have a Financial Conflict of Interest managed according to established policies in the context of licensed technology to ReGen Theranostics, Inc.
Rights and permissions
About this article
Cite this article
Theis, J.L., Hrstka, S.C.L., Evans, J.M. et al. Compound heterozygous NOTCH1 mutations underlie impaired cardiogenesis in a patient with hypoplastic left heart syndrome. Hum Genet 134, 1003–1011 (2015). https://doi.org/10.1007/s00439-015-1582-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00439-015-1582-1