Abstract
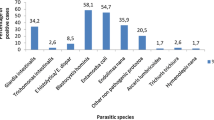
Direct wet mount examination and concentration are the most commonly used methods for detecting intestinal parasites from fecal samples. Concentration methods are used when there are fewer protozoan cyst, coccidian oocyst, microsporidial spore, helminth egg, and larvae in the fecal samples. Early detection of the causative intestinal parasites plays a significant role in implementing timely and correct treatment, which relieves the patients' symptoms and also prevents recurrences. Formalin-ethyl acetate concentration (FEAC) is believed to be a gold standard method to detect most intestinal parasites. Thus, in this study, we evaluated the diagnostic value of Feconomics® [manufactured by Salubris Inc, Boston, USA. Patent application number (TR): 2010/07549] which is a simple, new, and rapid fecal concentration method for the detection of the intestinal parasites in human beings. We also compared the FEAC with Feconomics® and direct wet mount examination. A total of 918 fecal samples were collected from the patients suspected to have intestinal parasitic infection. Samples were examined with the direct wet mount, FEAC, and Feconomics® methods. Different parasite species 15.9 % (146/918) with Feconomics®, 13.3 % (122/918) with FEAC, and 9.8 % (90/918) with direct wet mount examination, Feconomics® > FEAC > direct wet mount examinations were detected. They were statistically compared considering FEAC as the gold standard for parasitological diagnosis; the sensitivity and specificity of Feconomics® were calculated as 96 and 97 %, respectively. Blastocystis hominis was found to be the most common parasite, followed by Giardia lamblia with direct wet mount examination, FEAC, and Feconomics® methods. Feconomics® proved to be better than not only FEAC in concentrating parasite egg and cyst forms as well as in maintaining characteristic morphology but it is also better in direct wet mount examination. Feconomics® eliminates the need for centrifugation by using absorbent beads that help the homogenization and concentration of the sample. Feconomics® in this study was considerably better than FEAC in detecting the trophozoites of Giardia lamblia. We suggest that Feconomics® be used for the routine diagnosis of intestinal parasitic infection in rural areas of developing countries due to the fact that a centrifuge is not required and it eliminates large stool particles.


Similar content being viewed by others
References
Ahmedi NA, Damraj F (2009) A field evaluation of formalin-gasoline technique in the concentration of stool for detection of intestinal parasites. Parasitol Res 104:553–557
Cheesbrough M (1998) District laboratory practice in tropical countries, part 1. Cambridge University Press, Cambridge, pp 209–235
Coplu N, Gozalan A, Akın L (2007) Gaitada parazit incelenmesinde kullanılan yogunlaştırma yöntemlerinin karsılastırılması. Turk Parazitol Derg 31(2):123–128
Erdman D (1981) Clinical comparison of ethylacetate and diethyl ether in the formalin ether sedimentation technique. J Clin Microbiol 14(5):483–485
Faust EC, D’Antoni JS, Odom V, Miller MJ, Peres C, Sawitz W et al (1938) A critical study of clinical laboratory technicians for the diagnosis of protozoan cysts and helminth eggs in feces. Am J Trop Med 18:169–183
Garcia LS, Bruckner DA (1999) Practical guide to diagnostic parasitology American Society for Microbiology 9-82
Harp JA (2003) Parasitic infections of the gastrointestinal tract. Curr Opin Gastroenterol 29:31–36
Katagiri S, Oliveira-Sequeira TC (2010) Comparison of three concentration methods for the recovery of canine intestinal parasites from stool samples. Exp Parasitol 126(2):214–216
Kucik CJ, Martin GL, Sortor BV (2004) Common intestinal parasites. Am Fam Physician 69:161–168
Kurt O, Akyar I, Gorgun S, Kocagoz T, Ozbilgin A (2012) Feconomics®: a simple, novel and fast technique for stool concentration in parasitology laboratory Kafkas Univ Vet Fak Derg 18 (Suppl-A): A161-A165
Long EG, Tsin AT, Robinson BA (1985) Comparison of the fecal con-trade system with the formalin-ethyl acetate technique for detection of intestinal parasites. J Clin Microbiol 22(2):210–211
Markell EK, John DT, Krotoski WA (1999) Markell and Voge’s medical parasitology, 8th edition. W.B. Saunders Co, Philadelphia, p 501
Mendoza D, Nunaz FA, Escobedo AA, Pelavo L, Fernandez M, Torres D, Cordovi RA (2003) Usefullness of 2 coproparasitological methods and their utilization in an antigiardiasis therapeutic trial. Rev Cubana Med Trop 55(3):174–178
Methanitikorn R, Sukontason K, Sukontason KL, Piangiai S (2003) Evaluation of the formalin-tween concentration technique for parasitic detection. Rev Inst Med Trop Sao Paulo 45(5):289–291
Ok UZ, Yereli K (1996) Parazitoloji laboratuvarında sık kullanılan dışkı inceleme yöntemlerinin değerlendirilmesi. Türk Parazitol Derg 20:77–84
Perry JL, Matthews JS, Miller GR (1990) Parasite detection efficiencies of five stool concentration systems. J Clin Microbiol 20:1094–1097
Ritchie LS (1948) An ether sedimentation technique for routine stool examinations. Bull US Army Med Dept 8:326
Sing S (1990) Laboratory diagnosis of intestinal parasitosis. Indian J Pediatr 57:789–792
Young KH, Bullock SL, Melvin DM, Spruill CL (1979) Ethyl acetate as a substitute for diethyl ether in the formalin ether sedimentation technique. J Clin Microbiol 10:852–853
Zierdt WS (1984) Concentration and identification of Cryptosporidium sp. by use of a parasite concentrator. J Clin Microbiol 20(5):860–861
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Koltas, I.S., Akyar, I., Elgun, G. et al. Feconomics®; a new and more convenient method, the routine diagnosis of intestinal parasitic infections. Parasitol Res 113, 2503–2508 (2014). https://doi.org/10.1007/s00436-014-3899-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-014-3899-4