Abstract
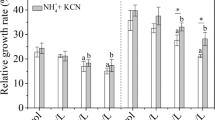
In plants, glutamine synthetase (GS) is the enzyme that is mainly responsible for the assimilation of ammonium. Conversely, in microorganisms such as bacteria and Ascomycota, NADP(H)-dependent glutamate dehydrogenase (GDH) and GS both have important roles in ammonium assimilation. Here, we report the changes in nitrogen assimilation, metabolism, growth, and grain yield of rice plants caused by an ectopic expression of NADP(H)-GDH (gdhA) from the fungus Aspergillus niger in the cytoplasm. An investigation of the kinetic properties of purified recombinant protein showed that the fungal gdhA had 5.4–10.2 times higher V max value and 15.9–43.1 times higher K m value for NH4 +, compared with corresponding values for rice cytosolic GS as reported in the literature. These results suggested that the introduction of fungal GDH into rice could modify its ammonium assimilation pathway. We therefore expressed gdhA in the cytoplasm of rice plants. NADP(H)-GDH activities in the gdhA-transgenic lines were markedly higher than those in a control line. Tracer experiments by feeding with 15NH4 + showed that the introduced gdhA, together with the endogenous GS, directly assimilated NH4 + absorbed from the roots. Furthermore, in comparison with the control line, the transgenic lines showed an increase in dry weight and nitrogen content when sufficient nitrogen was present, but did not do so under low-nitrogen conditions. Under field condition, the transgenic line examined showed a significant increase in grain yield in comparison with the control line. These results suggest that the introduction of fungal gdhA into rice plants could lead to better growth and higher grain yield by enhancing the assimilation of ammonium.





Similar content being viewed by others
Abbreviations
- CE–MS:
-
Capillary electrophoresis–mass spectrometry
- GDH:
-
Glutamate dehydrogenase
- GOGAT:
-
Glutamate synthase
- GS:
-
Glutamine synthetase
- MSX:
-
Methionine sulfoximine
- 2-OG:
-
2-Oxoglutarate
References
Abiko T, Obara M, Ushioda A, Hayakawa T, Hodges M, Yamaya T (2005) Localization of NAD-isocitrate dehydrogenase and glutamate dehydrogenase in rice roots: candidates for providing carbon skeletons to NADH-glutamate synthase. Plant Cell Physiol 46:1724–1734
Ameziane R, Bernhard K, Lightfoot D (2000) Expression of the bacterial gdhA gene encoding a NADPH glutamate dehydrogenase in tobacco affects plant growth and development. Plant Soil 221:47–57
Bradford M (1976) A rapid and sensitive method for the quantification of microgram quantities of protein utilizing the principle of protein dye binding. Anal Biochem 72:248–254
Chichkova S, Arellano J, Vance CP, Hernández G (2001) Transgenic tobacco plants that overexpress alfalfa NADH-glutamate synthase have higher carbon and nitrogen content. J Exp Bot 52:2079–2087
Fuentes SI, Allen DJ, Ortiz-Lopez A, Hernández G (2001) Over-expression of cytosolic glutamine synthetase increases photosynthesis and growth at low nitrogen concentrations. J Exp Bot 52:1071–1081
Hiei Y, Ohta S, Komari T, Kumashiro T (1994) Efficient transformation of rice (Oryza sativa L.) mediated by Agrobacterium and sequence analysis of the boundaries of the T-DNA. Plant J 6:271–282
Hirel B, Gadal P (1980) Glutamine synthetase in rice. Plant Physiol 66:619–623
Hodges M (2002) Enzyme redundancy and the importance of 2-oxoglutarate in plant ammonium assimilation. J Exp Bot 53:905–916
Ireland RJ, Lea PJ (1999) The enzymes of glutamine, glutamate, asparagine, and aspartate metabolism. In: Singh BK (ed) Plant amino acids: biochemistry and biotechnology. Marcel Dekker, New York, pp 49–109
Ishiyama K, Inoue E, Watanabe-Takahashi A, Obara M, Yamaya T, Takahashi H (2004a) Kinetic properties and ammonium-dependent regulation of cytosolic isoenzymes of glutamine synthetase in Arabidopsis. J Biol Chem 16(279):16598–16605
Ishiyama K, Inoue E, Tabuchi M, Yamaya T, Takahashi H (2004b) Biochemical background and compartmentalized functions of cytosolic glutamine synthetase for active ammonium assimilation in rice roots. Plant Cell Physiol 45:1640–1647
Kamachi K, Yamaya T, Hayakawa T, Mae T, Ojima K (1992) Vascular bundle-specific localization of cytosolic glutamine synthetase in rice leaves. Plant Physiol 99:1481–1486
Kinghorn JR, Pateman JA (1973) NAD and NADP l-glutamate dehydrogenase activity and ammonium regulation in Aspergillus nidulans. J Gen Microbiol 78:39–46
Kisaka H, Kida T (2007) Transgenic tomato plant carrying a gene for NADP-dependent glutamate dehydrogenase (gdhA) from Aspergillus nidulans. Plant Sci 164:35–42
Kiyomiya S, Nakanishi H, Uchida H, Tsuji A, Nishiyama S, Futatsubashi M, Tsukada H, Ishioka NS, Watanabe S, Ito T, Mizuniwa C, Osa A, Matsuhashi S, Hashimoto S, Sekine T, Mori S (2001) Real time visualization of 13N-translocation in rice under different environmental conditions using positron emitting tracer imaging system. Plant Physiol 125:1743–1753
Labboun S, Tercé-Laforgue T, Roscher A, Bedu M, Restivo FM, Velanis CN, Skopelitis DS, Moshou PN, Roubelakis-Angelakis KA, Suzuki A, Hirel B (2009) Resolving the role of plant glutamate dehydrogenase. I. In vivo real time nuclear magnetic resonance spectroscopy experiments. Plant Cell Physiol 50:1761–1773
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227:680–685
Lea PJ, Miflin BJ (1974) Alternative route for nitrogen assimilation in higher plants. Nature 18:614–616
Lea PJ, Thurman DA (1972) Intracellular location and properties of plant l-glutamate dehydrogenases. J Exp Bot 23:440–449
Macheda ML, Hynes MJ, Davis MA (1999) The Aspergillus nidulans gltA gene encoding glutamate synthase is required for ammonium assimilation in the absence of NADP-glutamate dehydrogenase. Curr Genet 34:467–471
Mae T, Ohira K (1981) The remobilization of nitrogen related to leaf growth and senescence in rice plants (Oryza sativa L.). Plant Cell Physiol 22:1067–1074
Magalhães JR, Ju GC, Rich PJ, Rhodes D (1990) Kinetics of 15NH4 + assimilation in Zea mays. Plant Physiol 94:647–656
Melo-Oliveira R, Oliveira IC, Coruzzi GM (1996) Arabidopsis mutant analysis and gene regulation define a nonredundant role for glutamate dehydrogenase in nitrogen assimilation. Proc Natl Acad Sci 14:4718–4723
Miyashita Y, Good AG (2008) NAD(H)-dependent glutamate dehydrogenase is essential for the survival of Arabidopsis thaliana during dark-induced carbon starvation. J Exp Bot 59:667–680
Mungur R, Glass AD, Goodenow DB, Lightfoot DA (2005) Metabolite fingerprinting in transgenic Nicotiana tabacum altered by the Escherichia coli glutamate dehydrogenase gene. J Biomed Biotechnol 30:198–214
Noor S, Punekar NS (2005) Allosteric NADP-glutamate dehydrogenase from aspergilli: purification, characterization and implications for metabolic regulation at the carbon–nitrogen interface. Microbiology 151:1409–1419
Obara M, Sato T, Yamaya T (2000) High contents of glutamine synthetase does not accompany a high activity of the enzyme in rice (Oryza sativa) leaves of indica cultivars. Physiol Plant 108:11–18
Obara M, Tamura W, Ebitani T, Yano M, Sato T, Yamaya T (2010) Fine-mapping of qRL6.1, a major QTL for root length of rice seedlings grown under a wide range of NH4 + concentrations in hydroponic conditions. Theor Appl Genet. doi:10.1007/s00122-010-1328-3
Oliveira IC, Brears T, Knight TJ, Clark A, Coruzzi GM (2002) Overexpression of cytosolic glutamine synthetase. Relation to nitrogen, light, and photorespiration. Plant Physiol 129:170–180
Paczek V, Dubois F, Sangwan R, Morot-Gaudry JF, Roubelakis-Angelakis KA, Hirel B (2003) Cellular and subcellular localisation of glutamine synthetase and glutamate dehydrogenase in grapes gives new insights on the regulation of carbon and nitrogen metabolism. Planta 216:245–254
Purnell MP, Botella JR (2007) Tobacco isoenzyme 1 of NAD(H)-dependent glutamate dehydrogenase catabolizes glutamate in vivo. Plant Physiol 143:530–539
Purnell MP, Skopelitis DS, Roubelakis-Angelakis KA, Botella JR (2005) Modulation of higher-plant NAD(H)-dependent glutamate dehydrogenase activity in transgenic tobacco via alteration of beta subunit levels. Planta 222:167–180
Sambrook J, Fritsch EF, Maniatis T (1989) Molecular cloning: a laboratory manual, 2nd edn. Cold Spring Harbor Laboratory Press, New York
Sato S, Soga T, Nishioka T, Tomita M (2004) Simultaneous determination of the main metabolites in rice leaves using capillary electrophoresis mass spectrometry and capillary electrophoresis diode array detection. Plant J 40:151–163
Skopelitis DS, Paranychianakis NV, Kouvarakis A, Spyros A, Stephanou EG, Roubelakis-Angelakis KA (2007) The isoenzyme 7 of tobacco NAD(H)-dependent glutamate dehydrogenase exhibits high deaminating and low aminating activities in vivo. Plant Physiol 145:1726–1734
Tabuchi M, Sugiyama K, Ishiyama K, Inoue E, Sato T, Takahashi H, Yamaya T (2005) Severe reduction in growth rate and grain filling of rice mutants lacking OsGS1;1, a cytosolic glutamine synthetase1;1. Plant J 42:641–651
Temple SJ, Knight TJ, Unkefer PJ, Sengupta-Gopalan C (1993) Modulation of glutamine synthetase gene expression in tobacco by the introduction of an alfalfa glutamine synthetase gene in sense and antisense orientation: molecular and biochemical analysis. Mol Gen Genet 236:315–325
Tercé-Laforgue T, Dubois F, Ferrario-Méry S, de Crecenzo MA, Sangwan R, Hirel B (2004) Glutamate dehydrogenase of tobacco is mainly induced in the cytosol of phloem companion cells when ammonia is provided either externally or released during photorespiration. Plant Physiol 136:4308–4317
Wakayama M, Abiko T, Asano T, Kawakami A, Egami T, Aoki N, Sasaki H, Ohsugi R (2008) Analysis of carboxylic acids and amino acids in plant extracts by using the capillary zone electrophoresis/tandem mass spectroscopy. In: Conference abstract book, 5th international conference on plant metabolomics, 15–18 July 2008, Yokohama, Japan, pp 162–163
Wootton JC (1983) Re-assessment of ammonium-ion affinities of NADP-specific glutamate dehydrogenases. Biochem J 209:527–531
Yamaya T, Oaks A, Matsumoto H (1984) Characteristics of glutamate dehydrogenase in mitochondria prepared from corn shoots. Plant Physiol 76:1009–1013
Yamaya T, Hayakawa T, Tanasawa K, Kamachi K, Mae T, Ojima K (1992) Tissue distribution of glutamate synthase and glutamine synthetase in rice leaves. Plant Physiol 100:1427–1432
Yamaya T, Obara M, Nakajima H, Sasaki S, Hayakawa T, Sato T (2002) Genetic manipulation and quantitative-trait loci mapping for nitrogen recycling in rice. J Exp Bot 53:917–925
Acknowledgments
We thank Dr. Tatsuro Hirose for provision of the antibody of susy2, Dr. Nakagawa for provision of the pGWB5 vector, and Dr. Masaru Fujimoto for assistance with confocal laser microscopy. The field trial in South Korea was practiced in cooperation with Inplanta Innovations Inc. (Yokohama, Japan). This work was supported by a grant from the PROBRAIN (Program for promotion of Basic Research Activities for Innovative Biosciences) project of the Ministry of Agriculture, Forestry, and Fisheries of Japan.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Abiko, T., Wakayama, M., Kawakami, A. et al. Changes in nitrogen assimilation, metabolism, and growth in transgenic rice plants expressing a fungal NADP(H)-dependent glutamate dehydrogenase (gdhA). Planta 232, 299–311 (2010). https://doi.org/10.1007/s00425-010-1172-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00425-010-1172-3