Abstract.
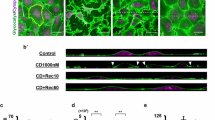
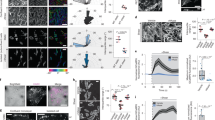
Clostridium botulinum C2 toxin (C2 toxin) and purified ADP-ribosylated-α-actin (ADP-r-α-actin) cause specific actin depolymerisation in living cells. This effect was used to investigate the actin microfilament system with particular emphasis on cell-cell adhesion and plasma membrane integrity in endothelial cells. C2 toxin caused time- and dose-dependent (15–100 ng/ml) changes in endothelial surface morphology (investigated by atomic force microscopy), intercellular gap formation and cell detachment under shear stress. Low concentrations of C2 toxin (1.5 ng/ml), however, did not induce cell detachment but inhibited shear stress-dependent cell alignment. Gap formation as well as cell loss under shear stress was also observed in cells microinjected with purified ADP-r-α-actin. Intercellular gap formation was mediated by increased α-catenin solubility (40%) due to actin filament depolymerisation. Disintegration of plasma membranes (measured by LDH release) and cell fragmentation during simultaneous exposure to shear stress and C2 toxin were due to a loss of more than 50% of membrane-associated actin. These data show that small disturbances in actin dynamics inhibit shear stress-dependent cell alignment; that depolymerisation of actin filaments increases the solubility of α-catenin, thus resulting in cell dissociation and that actin filaments of the membrane cytoskeleton are required to protect the cells from haemodynamic injury such as shear stress. Together, the study shows a heterogeneous regulation of actin filament dynamics at subcellular locations. Junction-associated actin filaments displayed the highest sensitivity whereas stress fibres were far more stable.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Electronic Publication
Rights and permissions
About this article
Cite this article
Schnittler, HJ., Schneider, S.W., Raifer, H. et al. Role of actin filaments in endothelial cell-cell adhesion and membrane stability under fluid shear stress. Pflügers Arch - Eur J Physiol 442, 675–687 (2001). https://doi.org/10.1007/s004240100589
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s004240100589