Abstract
Aims
To determine the (1) incidence of adverse drug events (ADEs) in 10 emergency department (EDs) of general hospitals in the Regione Campania (southern Italy), (2) rate of ADE-related hospital admissions, (3) drug classes most frequently involved, and (4) the types of ADEs and their frequency.
Methods
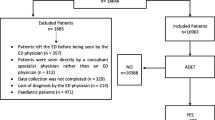
We performed a cohort study of all patients attending the EDs. This study was carried out in two observational periods of 10 days each in 10 EDs. Demographic, clinical, and pharmacological data about all patients admitted to EDs were collected by trained and qualified monitors. Records related to ADEs were analyzed and validated by a specific scientific committee.
Results
Of 7,861 ED visits, 96 were ADE-related. The incidence of hospitalization was higher in patients who had taken medication than in patients with a negative drug history (24.9 vs. 16.4%). ADEs were significantly more frequent in women. Patients aged between 60 and 69 years and between 30 and 39 years were significantly more likely to experience an ADE. Serious ADEs were identified in 20 ED visits (20.8% of total sample). Antibiotics, NSAIDs, and agents acting on the renin-angiotensin system were the drugs most often involved in ADEs. In multivariate analyses, the adjusted odds ratio was 3.4 (95% CI: 1.07–2.84) for patients taking NSAIDs, 4.78 (95% CI: 2.26–10.12) for those taking β2-adrenergic-receptor agonists, and 6.20 (95%CI: 2.74–14.06) for those taking β-lactam antibiotics.
Conclusion
This study shows that ADEs are an important problem in industrialized countries. Moreover, it shows that ADEs affect hospital admission rates and reinforces the importance of drug-induced disease as a public health problem.

Similar content being viewed by others
References
Prince BS, Goetz CM, Rihn TL, Olsky M (1992) Drug-related emergency department visits and hospital admissions. Am J Hosp Pharm 49(7):1696–1700
Einarson TR (1993) Drug-related hospital admissions. Ann Pharmacother 27(7–8):832–840
Roughead EE, Gilbert AL, Primrose JG, Sansom LN (1998) Drug-related hospital admissions: a review of Australian studies published 1988–1996. Med J Aust 168(8):405–408
Schneeweiss S, Hasford J, Göttler M, Hoffmann A, Riethling AK, Avorn J (2002) Admissions caused by adverse drug events to internal medicine and emergency departments in hospitals: a longitudinal population-based study. Eur J Clin Pharmacol 58(4):285–291
Bergman U, Wiholm BE (1981) Drug-related problems causing admission to a medical clinic. Eur J Clin Pharmacol 20(3):193–200
Mitchell AS, Henry DA, Sanson-Fisher R, O’Connell DL (1988) Patients as a direct source of information on adverse drug reactions. BMJ 297(6653):891–893
Mannesse CK, Derkx FH, de Ridder MA, Man in ‘t Veld AJ, van der Cammen TJ (2000) Contribution of adverse drug reactions to hospital admission of older patients. Age Ageing 29(1):35–39
Stoukides CA, D’Agostino PR, Kaufman MB (1993) Adverse drug reaction surveillance in an emergency room. Am J Hosp Pharm 50(4):712–714
Dennehy CE, Kishi DT, Louie C (1996) Drug-related illness in emergency department patients. Am J Health Syst Pharm 53(12):1422–1426
Schneitman-McIntire O, Farnen TA, Gordon N, Chan J, Toy WA (1996) Medication misadventures resulting in emergency department visits at an HMO medical center. Am J Health Syst Pharm 53(12):1416–1422
Smith KM, McAdams JW, Frenia ML, Todd MW (1997) Drug-related problems in emergency department patients. Am J Health Syst Pharm 54(3):295–298
Raschetti R, Morgutti M, Menniti-Ippolito F, Belisari A, Rossignoli A, Longhini P, Guidara C (1999) Suspected adverse drug events requiring emergency department visits or hospital admission. Eur J Clin Pharmacol 54:959–963
Malhotra S, Jain S, Pandhi P (2001) Drug-related visits to the medical emergency department: a prospective study from India. Int J Clin Pharmacol Ther 39(1):12–18
Hafner JW, Belknap SM, Squillante MD, Bucheit KA (2002) Adverse drug events in emergency department patients. Ann Emerg Med 39(3):258–267
Capuano A, Motola G, Russo F, Avolio A, Filippelli A, Rossi F, Mazzeo F (2004) Adverse drug events in two emergency departments in Naples, Italy: an observational study. Pharmacol Res 50(6):631–636
Hohl CM, Dankoff J, Colacone A, Afilalo M (2001) Polypharmacy, adverse drug-related events, and potential adverse drug interactions in elderly patients presenting to an emergency department. Ann Emerg Med 38(6):666–671
Wasserfallen J, Livio F, Buclin T, Tillet L, Yersin B, Biollaz J (2001) Rate, type, and cost of adverse drug reactions in emergency department admissions. Eur J Intern Med 12(5):442–447
Patel P, Zed PJ (2002) Drug-related visits to the emergency department: how big is the problem? Pharmacotherapy 22(7):915–923
Queneau P, Bannwarth B, Carpentier F, Guliana JM, Bouget J, Trombert B, Leverve X, Lapostolle F, Borron SW, Adnet F, Association Pédagogique Nationale pour l’Enseignement de la Thérapeutique (APNET) (2007) Emergency department visits caused by adverse drug events: results of a French survey. Drug Safety 30(1):81–88
Gurwitz JH, Field TS, Judge J, Rochon P, Harrold LR, Cadoret C, Lee M, White K, LaPrino J, Erramuspe-Mainard J, De Florio M, Gavendo L, Auger J, Bates DW (2005) The incidence of adverse drug events in two large academic long-term care facilities. Am J Med 118(3):251–258
Olivier P, Boulbés O, Tubery M, Lauque D, Montastruc J-L, Lapeyre-Mestre M (2002) Assessing the feasibility of using an adverse drug reaction preventability scale in clinical practice: a study in a French emergency department. Drug Safety 25(14):1035–1044
Wasserfallen JB, Livio F, Buclin T, Tillet L, Yersin B, Biollaz J (2001) Rate, type, and cost of diverse drug reactions in emergency department admissions. Eur J Intern Med 12:442–447
Nebeker JR, Barach P, Samore MH (2004) Clarifying adverse drug events: a clinician’s guide to terminology, documentation, and reporting. Ann Intern Med 140(10):795–801
Schumock GT, Thornton JP (1992) Focusing on the preventability of adverse drug reactions. Hosp Pharm 27:538
Pouyanne P, Haramburu F, Imbs JL, Begaud B (2000) Admissions to hospital caused by adverse drug reactions: cross sectional incidence study. French Pharmacovigilance Centres. BMJ 320(7241):1036
Hallas J, Harvald B, Gram LF, Grodum E, Brosen K, Haghfelt T, Damsbo N (1990) Drug related hospital admissions: the role of definitions and intensity of data collection, and the possibility of prevention. J Intern Med 228(2):83–90
Lazarou J, Pomeranz BH, Corey PN (1998) Incidence of adverse drug reactions in hospitalized patients: a meta-analysis of prospective studies. JAMA 279(15):1200–1205
Trifirò G, Calogero G, Ippolito FM, Cosentino M, Giuliani R, Conforti A, Venegoni M, Mazzaglia G, Caputi AP (2005) Adverse drug events in emergency department population: a prospective Italian study. Pharmacoepidemiol Drug Saf 14(5):333–340
Field TS, Gurwitz JH, Harrold LR, Rothschild J, DeBellis KR, Seger AC, Auger JC, Garber LA, Cadoret C, Fish LS, Garber LD, Kelleher M, Bates DW (2004) Risk factors for adverse drug events among older adults in the ambulatory setting. J Am Geriatr Soc 52(8):1349–1354
Gandhi TK, Bates DW, Burstin HR et al (1998) Drug complications in outpatients. J Gen Intern Med 13(suppl 1):46
Gwee MC (1993) Prevention of adverse drug reactions: role of the patient (consumer). Ann Acad Med Singap 22(1):90–93
Grymonpre RE, Mitenko PA, Sitar DS et al (1988) Drug-associated hospital admissions in older medical patients. J Am Geriatr Soc 36:1092–1098
Gerety MB, Cornell JE, Plichta DT et al (1993) Adverse events related to drugs and drug withdrawal in nursing home residents. J Am Geriatr Soc 41:1326–1332
Mazzaglia G, Caputi AP, Rossi A et al (2003) Exploring patient- and doctor-related variables associated with antibiotic prescribing for respiratory infections in primary care. Eur J Clin Pharmacol 59:651–657
Traversa G, Bianchi C, Da Cas R, Abraha I, Menniti-Ippolito F, Venegoni M (2003) Cohort study of hepatotoxicity associated with nimesulide and other non-steroidal anti-inflammatory drugs. Br Med J 327:18–22
Benichou C, Danan G, Flahault A (1993) Causality assessment of adverse reactions to drugs–II. An original model for validation of drug causality assessment methods: case reports with positive rechallenge. J Clin Epidemiol 46(11):1331–1336
Acknowledgements
The authors thank the monitors for data collection and the physicians of the 10 hospital EDs involved in the study: Azienda Ospedaliera (A.O.) A Cardarelli, Napoli, A.O. S. Maria degli Incurabili di Napoli, Presidio Ospedaliero (P.O) Loreto Mare di Napoli, A.O. Pellegrini di Napoli, A.O. San Paolo di Napoli, A.O. San Giuseppe Moscati di Avellino, A.O. G. Rummo di Benevento, A.O. San Sebastiano di Caserta, AA.OO.RR. San Giovanni di Dio e Ruggi d’Aragona di Salerno, and Ospedale San Leonardo di Castellammare di Stabia. We are grateful to Jean Ann Gilder for text editing. This study was supported by grants from Regione Campania (Delibera Giunta Regionale no. 6458 del 31/11/2001).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Capuano, A., Irpino, A., Gallo, M. et al. Regional surveillance of emergency-department visits for outpatient adverse drug events. Eur J Clin Pharmacol 65, 721–728 (2009). https://doi.org/10.1007/s00228-009-0641-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00228-009-0641-8