Abstract
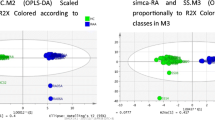
The nuclear magnetic resonance (NMR)-based metabolomic approach was used as analytical methodology to study the urine samples of chronic inflammatory rheumatic disease (CIRD) patients. The urine samples of CIRD patients were compared to the ones of both healthy subjects and patients with multiple sclerosis (MS), another immuno-mediated disease. Urine samples collected from 39 CIRD patients, 25 healthy subjects, and 26 MS patients were analyzed using 1H NMR spectroscopy, and the NMR spectra were examined using partial least squares-discriminant analysis (PLS-DA). PLS-DA models were validated by a double cross-validation procedure and randomization tests. Clear discriminations between CIRD patients and healthy controls (average diagnostic accuracy 83.5 ± 1.9%) as well as between CIRD patients and MS patients (diagnostic accuracy 81.1 ± 1.9%) were obtained. Leucine, alanine, 3-hydroxyisobutyric acid, hippuric acid, citric acid, 3-hydroxyisovaleric acid, and creatinine contributed to the discrimination; all of them being in a lower concentration in CIRD patients as compared to controls or to MS patients. The application of NMR metabolomics to study these still poorly understood diseases can be useful to better clarify the pathologic mechanisms; moreover, as a holistic approach, it allowed the detection of, by means of anomalous metabolic traits, the presence of other pathologies or pharmaceutical treatments not directly connected to CIRDs, giving comprehensive information on the general health state of individuals.

NMR-based metabolomic approach as a tool to study urine samples in CIRD patients with respect to MS patients and healthy controls




Similar content being viewed by others
Abbreviations
- CIRDs:
-
Chronic inflammatory rheumatic diseases
- COSY:
-
Correlation spectroscopy
- CSF:
-
Cerebrospinal fluid
- HMBC:
-
Heteronuclear multiple bond correlation
- HPLC:
-
High-performance liquid chromatography
- HSQC:
-
Heteronuclear single quantum coherence
- MS:
-
Multiple sclerosis
- NMR:
-
Nuclear magnetic resonance
- PLS-DA:
-
Partial least squares-discriminant analysis
- TMSP:
-
Trimethylsilyl propionate
- UTIs:
-
Urinary tract infections
- VIP:
-
Variable importance in projection
References
Wishart DS, Jewison T, Guo AC, Wilson M, Knox C, Liu Y, et al. HMDB 3.0—the Human Metabolome Database in 2013. Nucleic Acids Res. 2013;41:D801–7. doi:10.1093/nar/gks1065.
Slupsky CM, Rankin KN, Wagner J, Fu H, Chang D, Weljie AM, et al. Investigations of the effects of gender, diurnal variation, and age in human urinary metabolomic profiles. Anal Chem. 2007;79:6995–7004. doi:10.1021/ac0708588.
Liquid state 1H high field NMR in food analysis. http://www.sciencedirect.com/science/article/pii/S0079656512000209. Accessed 7 July 2016.
Pelantová H, Bártová S, Anýž J, Holubová M, Železná B, Maletínská L, et al. Metabolomic profiling of urinary changes in mice with monosodium glutamate-induced obesity. Anal Bioanal Chem. 2015;408:567–78. doi:10.1007/s00216-015-9133-0.
Assfalg M, Bertini I, Colangiuli D, Luchinat C, Schäfer H, Schütz B, et al. Evidence of different metabolic phenotypes in humans. Proc Natl Acad Sci. 2008;105:1420–4. doi:10.1073/pnas.0705685105.
Bernini P, Bertini I, Luchinat C, Nepi S, Saccenti E, Schäfer H, et al. Individual human phenotypes in metabolic space and time. J Proteome Res. 2009;8:4264–71. doi:10.1021/pr900344m.
Ghini V, Saccenti E, Tenori L, Assfalg M, Luchinat C. Allostasis and resilience of the human individual metabolic phenotype. J Proteome Res. 2015. doi:10.1021/acs.jproteome.5b00275.
Jones DP, Park Y, Ziegler TR. Nutritional metabolomics: progress in addressing complexity in diet and health. Annu Rev Nutr. 2012;32:183–202. doi:10.1146/annurev-nutr-072610-145159.
Bertini I, Calabrò A, De Carli V, Luchinat C, Nepi S, Porfirio B, et al. The metabonomic signature of celiac disease. J Proteome Res. 2008;8:170–7. doi:10.1021/pr800548z.
Priori R, Scrivo R, Brandt J, Valerio M, Casadei L, Valesini G, et al. Metabolomics in rheumatic diseases: the potential of an emerging methodology for improved patient diagnosis, prognosis, and treatment efficacy. Autoimmun Rev. 2013;12:1022–30. doi:10.1016/j.autrev.2013.04.002.
Tenori L, Oakman C, Morris PG, Gralka E, Turner N, Cappadona S, et al. Serum metabolomic profiles evaluated after surgery may identify patients with oestrogen receptor negative early breast cancer at increased risk of disease recurrence. Results from a retrospective study. Mol Oncol. 2015;9:128–39. doi:10.1016/j.molonc.2014.07.012.
Bernini P, Bertini I, Luchinat C, Tenori L, Tognaccini A. The cardiovascular risk of healthy individuals studied by NMR metabonomics of plasma samples. J Proteome Res. 2011;10:4983–92. doi:10.1021/pr200452j.
Wang L, Wu L-F, Lu X, Mo X-B, Tang Z-X, Lei S-F, et al. Integrated analyses of gene expression profiles digs out common markers for rheumatic diseases. PLoS ONE. 2015;10:e0137522. doi:10.1371/journal.pone.0137522.
Bernini P, Bertini I, Luchinat C, Nincheri P, Staderini S, Turano P. Standard operating procedures for pre-analytical handling of blood and urine for metabolomic studies and biobanks. J Biomol NMR. 2011;49:231–43. doi:10.1007/s10858-011-9489-1.
Bouatra S, Aziat F, Mandal R, Guo AC, Wilson MR, Knox C, et al. The human urine metabolome. PLoS ONE. 2013;8:e73076. doi:10.1371/journal.pone.0073076.
Dieterle F, Ross A, Schlotterbeck G, Senn H. Probabilistic quotient normalization as robust method to account for dilution of complex biological mixtures. Application in 1H NMR metabonomics. Anal Chem. 2006;78:4281–90. doi:10.1021/ac051632c.
Ihaka R, Gentleman R. R: a language for data analysis and graphics. J Comput Stat Graph. 1996;5:299–314.
Kruskal WH, Wallis WA. Use of ranks in one-criterion variance analysis. J Am Stat Assoc. 1952;47:583. doi:10.2307/2280779.
Benjamini Y, Hochberg Y. Controlling the false discovery rate: a practical and powerful approach to multiple testing. J R Stat Soc Ser B Methodol. 1995;57:289–300.
Hassan-Smith G, Wallace GR, Douglas MR, Sinclair AJ. The role of metabolomics in neurological disease. J Neuroimmunol. 2012;248:48–52. doi:10.1016/j.jneuroim.2012.01.009.
Young SP, Kapoor SR, Viant MR, Byrne JJ, Filer A, Buckley CD, et al. The impact of inflammation on metabolomic profiles in patients with arthritis. Arthritis Rheum. 2013;65:2015–23. doi:10.1002/art.38021.
Zabek A, Swierkot J, Malak A, Zawadzka I, Deja S, Bogunia-Kubik K, et al. Application of 1H NMR-based serum metabolomic studies for monitoring female patients with rheumatoid arthritis. J Pharm Biomed Anal. 2016;117:544–50. doi:10.1016/j.jpba.2015.10.007.
Nevedomskaya E, Pacchiarotta T, Artemov A, Meissner A, van Nieuwkoop C, van Dissel JT, et al. 1H NMR-based metabolic profiling of urinary tract infection: combining multiple statistical models and clinical data. Metabolomics. 2012;8:1227–35. doi:10.1007/s11306-012-0411-y.
Puntis D, Malik S, Saravanan V, Rynne M, Heycock C, Hamilton J, et al. Urinary tract infections in patients with rheumatoid arthritis. Clin Rheumatol. 2012;32:355–60. doi:10.1007/s10067-012-2129-7.
Munro R, Capell H. Prevalence of low body mass in rheumatoid arthritis: association with the acute phase response. Ann Rheum Dis. 1997;56:326–9.
Willer B, Stucki G, Hoppeler H, Brühlmann P, Krähenbühl S. Effects of creatine supplementation on muscle weakness in patients with rheumatoid arthritis. Rheumatol Oxf Engl. 2000;39:293–8.
Krähenbühl S, Willer B, Brühlmann P, Hoppeler H, Stucki G. Carnitine homeostasis in patients with rheumatoid arthritis. Clin Chim Acta. 1999;279:35–45. doi:10.1016/S0009-8981(98)00161-2.
van Wietmarschen HA, Dai W, van der Kooij AJ, Reijmers TH, Schroën Y, Wang M, et al. Characterization of rheumatoid arthritis subtypes using symptom profiles, clinical chemistry and metabolomics measurements. PLoS ONE. 2012;7:e44331. doi:10.1371/journal.pone.0044331.
Kapoor SR, Filer A, Fitzpatrick MA, Fisher BA, Taylor PC, Buckley CD, et al. Metabolic profiling predicts response to anti-tumor necrosis factor α therapy in patients with rheumatoid arthritis. Arthritis Rheum. 2013;65:1448–56. doi:10.1002/art.37921.
Mount D, Sayegh MH, Singh AK. Core concepts in the disorders of fluid, electrolytes and acid–base balance. New York: Springer Science & Business Media; 2012.
Dickens AM, Larkin JR, Griffin JL, Cavey A, Matthews L, Turner MR, et al. A type 2 biomarker separates relapsing-remitting from secondary progressive multiple sclerosis. Neurology. 2014;83:1492–9.
Tavazzi B, Batocchi AP, Amorini AM, Nociti V, D’Urso S, Longo S, et al. Serum metabolic profile in multiple sclerosis patients. Mult Scler Int. 2011;2011:e167156. doi:10.1155/2011/167156.
Reinke SN, Broadhurst DI, Sykes BD, Baker GB, Catz I, Warren KG, et al. Metabolomic profiling in multiple sclerosis: insights into biomarkers and pathogenesis. Mult Scler J. 2014;20:1396–400. doi:10.1177/1352458513516528.
Nicholson JK, Holmes E, Wilson ID. Gut microorganisms, mammalian metabolism and personalized health care. Nat Rev Microbiol. 2005;3:431–8.
Acknowledgements
This study has been developed within the “Unità di Metabolomica: Studi su Alimenti, Nutraceutici e Fluidi biologici” of the Sapienza University of Rome. This work has been partially supported by Fondazione Veronesi that granted L.T. through the Post-Doctoral Fellowship 2015 and by Ateneo 2015 (Sapienza University of Rome prot C26A15CJ98).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The study received the approval by the local ethics committee in accordance with local requirements, and at the time of the collection, informed consent was obtained from each participant enrolled in the study.
Conflict of interest
The authors declare that they have no competing interests.
Additional information
Alessia Vignoli, Donatella Maria Rodio, Anna Bellizzi and Anatoly Petrovich Sobolev contributed equally to this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(PDF 352 kb)
Rights and permissions
About this article
Cite this article
Vignoli, A., Rodio, D.M., Bellizzi, A. et al. NMR-based metabolomic approach to study urine samples of chronic inflammatory rheumatic disease patients. Anal Bioanal Chem 409, 1405–1413 (2017). https://doi.org/10.1007/s00216-016-0074-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00216-016-0074-z