Abstract
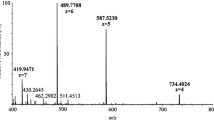
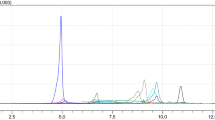
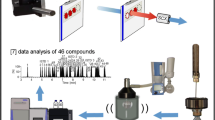
In the present study, a new type of mass spectrometer combining a quadrupole mass filter, a higher collision dissociation (HCD) cell and an Orbitrap detector, was evaluated for the analysis of dried blood spots (DBS) in doping controls. DBS analysis is characterized by the necessity to detect prohibited compounds in sub-nanogram-per-milliliter levels with high identification capacity. After extraction of DBS with an organic solvent and liquid chromatographic separation (using a regular C18-RP-analytical UHPLC-column) of target analytes, mass spectrometry is performed with a high-resolution full scan in positive and negative mode by means of electrospray ionisation. Single-product ion mass spectra are acquired using the data-dependent analysis mode (employing an inclusion list) for previously selected precursors of known prohibited compounds with fixed retention time ranges. Besides, a sensitive screening in a targeted approach, non-targeted analysis for retrospective data evaluation is thus possible. The chosen experimental design enables the determination of various drugs from different classes with one generic sample preparation which is shown for 26 selected model compounds (Δ9-tetrahydrocannabinol (THC), tetrahydrocannabinol-9-carboxylic acid (THC-COOH), methylhexaneamine, methylphenidate, cocaine, nikethamide, 3,4-methylenedioxyamphetamine, N-methyl-3,4-methylenedioxyamphetamine, strychnine, mesocarb, salbutamol, formoterol, clenbuterol, metandienone, stanozolol, bisoprolol, propranolol, metoprolol, anastrazole, clomiphene, exemestane, dexamethasone, budesonide, selective androgen receptor modulator (SARM) S4 (andarine), SARM S1, hydrochlorothiazide). Generally, only qualitative result interpretation was focussed upon, but for target analytes with deuterium-labelled internal standards (salbutamol, clenbuterol, cocaine, dexamethasone, THC-COOH and THC) quantitative analysis was also possible. Especially the most challenging analytes, THC and its carboxy metabolite, were detected in DBS at relevant concentrations (<0.5 ng/mL) using targeted HCD experiments. The method was validated for the parameters: specificity, linearity (0–20 ng/mL), precision (<25%), recovery (mean 60%), limit of detection/quantification, ion suppression, stability and accuracy (80–120%). Six isotope-labelled analogues used as internal standards facilitate a quantitative result interpretation which is of utmost importance especially for in-competition drug sports testing.



Similar content being viewed by others
References
Danaceau JP, Scott Morrison M, Slawson MH (2008) J Mass Spectrom 43:993–1000
Moulard Y, Bailly-Chouriberry L, Boyer S, Garcia P, Popot MA, Bonnaire Y (2011) Anal Chim Acta 700:126–136
Clarke A, Scarth J, Teale P, Pearce C, Hillyer L (2011) Drug Test Anal 3:74–87
Jiwan JL, Wallemacq P, Herent MF (2011) Clin Biochem 44:136–147
Thomas A, Guddat S, Kohler M, Krug O, Schänzer W, Petrou M, Thevis M (2010) Rapid Commun Mass Spectrom 24:1124–1132
Thomas A, Höppner S, Geyer H, Schänzer W, Petrou M, Kwiatkowska D, Pokrywka A, Thevis M (2011) Anal Bioanal Chem 401:507–516
Thomas A, Kohler M, Mester J, Geyer H, Schänzer W, Petrou M, Thevis M (2010) Drug Test Anal 2:144–148
Peters FT (2011) Clin Biochem 44:54–65
Maurer HH (2011) Ther Drug Monit 32:324–327
Thevis M, Makarov AA, Horning S, Schänzer W (2005) Rapid Commun Mass Spectrom 19:3369–3378
Michalski A, Damoc E, Hauschild JP, Lange O, Wieghaus A, Makarov A, Nagaraj N, Cox J, Mann M, Horning S Mol Cell Proteomics 10: doi:10.1074/mcp.M111.011015
Scigelova M, Hornshaw M, Giannakopulos A, Makarov A (2011) Mol Cell Proteomics 10: doi: 10.1074/mcp.M111.009431
Thomas A, Geyer H, Guddat S, Schänzer W, Thevis M Drug Test Anal doi:10.1002/dta.342
Abu-Rabie P, Spooner N (2009) Anal Chem 81:10275–10284
Aldenhoven M, de Koning TJ, Verheijen FW, Prinsen BH, Wijburg FA, van der Ploeg AT, de Sain-van der Velden MG, Boelens J (2010) Biol Blood Marrow Transplant 16:701–704
Carducci C, Santagata S, Leuzzi V, Artiola C, Giovanniello T, Battini R, Antonozzi I (2006) Clin Chim Acta 364:180–187
Clavijo CF, Hoffman KL, Thomas JJ, Carvalho B, Chu LF, Drover DR, Hammer GB, Christians U, Galinkin JL (2011) Anal Bioanal Chem 400:715–728
Deglon J, Lauer E, Thomas A, Mangin P, Staub C (2010) Anal Bioanal Chem 396:2523–2532
Deglon J, Thomas A, Cataldo A, Mangin P, Staub C (2009) J Pharm Biomed Anal 49:1034–1039
Li W, Tse FL (2010) Biomed Chromatogr 24:49–65
Mather J, Rainville PD, Spooner N, Evans CA, Smith NW, Plumb RS (2011) Bioanalysis 3:411–420
McDade TW, Williams S, Snodgrass JJ (2007) Demography 44:899–925
Parker DR, Bargiota A, Cowan FJ, Corrall RJ (1997) Clin Endocrinol (Oxf) 47:679–683
Patel P, Tanna S, Mulla H, Kairamkonda V, Pandya H, Lawson G (2010) J Chromatogr B Analyt Technol Biomed Life Sci 878:3277–3282
Therrell BL, Hannon WH, Pass KA, Lorey F, Brokopp C, Eckman J, Glass M, Heidenreich R, Kinney S, Kling S, Landenburger G, Meaney FJ, McCabe ER, Panny S, Schwartz M, Shapira E (1996) Biochem Mol Med 57:116–124
Wong P, Pham R, Bruenner BA, James CA (2010) Bioanalysis 2:1787–1789
Thevis M, Thomas A, Fusshöller G, Beuck S, Geyer H, Schänzer W (2010) Rapid Commun Mass Spectrom 24:2245–2254
Thevis M, Geyer H, Kamber M, Schänzer W (2009) Drug Test Anal 1:387–392
Matuszewski BK, Constanzer ML, Chavez-Eng CM (2003) Anal Chem 75:3019–3030
Brenneisen R, Meyer P, Chtioui H, Saugy M, Kamber M (2010) Anal Bioanal Chem 396:2493–2502
König S, Aebi B, Lanz S, Gasser M, Weinmann W (2011) Anal Bioanal Chem 400:9–16
Schwope DM, Scheidweiler KB, Huestis MA (2011) Anal Bioanal Chem 401:1273–1283
Zhang H, Zhu M, Ray KL, Ma L, Zhang D (2008) Rapid Commun Mass Spectrom 22:2082–2088
http://www.wada-ama.org/en/World-Anti-Doping-Program/Sports-and-Anti-Doping-Organizations/International-Standards/Prohibited-List/ (2011) Vol. 2011, WADA
Saugy M, Robinson N, Saudan C (2009) Drug Test Anal 1:474–478
http://www.wada-ama.org/Documents/World_Anti-Doping_Program/WADP-IS-Laboratories/WADA_Int.Standard_Laboratories_2009_EN.pdf (2011) Vol. 2011, WADA
Thevis M (2010) Mass spectrometry in sports drug testing. Wiley, New Jersey
Spooner N, Ramakrishnan Y, Barfield M, Dewit O, Miller S (2011) Bioanalysis 2:1515–1522
Denniff P, Spooner N (2011) Bioanalysis 2:1385–1395
Abu-Rabie P, Spooner N (2011) Bioanalysis 2:1373–1384
Youhnovski N, Bergeron A, Furtado M, Garofolo F (2011) Rapid Commun Mass Spectrom 25:2951–2958
Thevis M, Thomas A, Kohler M, Beuck S, Schänzer W (2009) J Mass Spectrom 44:442–460
Thevis M, Schänzer W (2007) Mini Rev Med Chem 7:531–537
Acknowledgements
The study was carried out with support of Antidoping Switzerland (Berne, Switzerland), the Manfred Donike Institute for Doping Analysis, Cologne, and the Federal Ministry of the Interior of the Federal Republic of Germany.
Author information
Authors and Affiliations
Corresponding author
Additional information
Published in the special issue High-Resolution Mass Spectrometry with guest editors Hans H. Maurer and David C. Muddiman.
Rights and permissions
About this article
Cite this article
Thomas, A., Geyer, H., Schänzer, W. et al. Sensitive determination of prohibited drugs in dried blood spots (DBS) for doping controls by means of a benchtop quadrupole/Orbitrap mass spectrometer. Anal Bioanal Chem 403, 1279–1289 (2012). https://doi.org/10.1007/s00216-011-5655-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00216-011-5655-2