Abstract
Rationale
Although passive administration of heroin to drug-naive rats increases extracellular dopamine (DA) in the nucleus accumbens (NAc), its ability to do so also after active drug exposure (self-administration) is debated.
Objectives
This study investigated by repeated microdialysis sampling the inter- and intrasession changes in the responsiveness of the NAc shell and core DA and the behavioral effects of active and passive heroin exposure in the intravenous self-administration/yoked paradigm.
Materials and methods
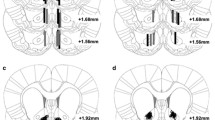
Rats were implanted with jugular catheters and bilateral intracerebral chronic guide cannulae. Nose poking in the active hole by master rats resulted in heroin administration to the same subjects and to their yoked mates. Concentric microdialysis probes were inserted daily in the guide cannulae, and changes in dialysate DA in response to heroin exposure (0.05 mg/kg) were monitored in the same subject for 90 min for 4 weeks. Behavior associated with heroin exposure, distinguished into nonstereotyped and stereotyped, was also recorded.
Results
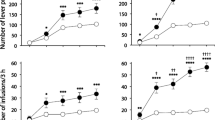
Dialysate DA increased preferentially in the shell of master rats from the first session (+112%) and throughout the 4 weeks of self-administration (+130–140%). In yoked rats, a preferential but lesser increase in DA in the shell was observed only on the first session (+60%), as the DA response in the NAc core increased progressively (+25–118%), so that within a week, the shell/core ratio was reversed, and this pattern was maintained for the following 2 weeks. Yoked rats showed a progressive and larger increase in stereotyped behaviors than master rats.
Conclusions
Chronic heroin self-administration increases extracellular DA preferentially in the NAc shell. Response-noncontingent heroin administration is particularly prone, compared to response-contingent administration, to induce behavioral and biochemical sensitization.








Similar content being viewed by others
References
Bassareo V, Di Chiara G (1997) Differential influence of associative and nonassociative learning mechanisms on the responsiveness of prefrontal and accumbal dopamine transmission to food stimuli in rats fed ad libitum. J Neurosci 17:851–861
Bassareo V, Di Chiara G (1999) Differential responsiveness of dopamine transmission to food-stimuli in nucleus accumbens shell/core compartments. Neuroscience 89:637–641
Bassareo V, De Luca MA, Di Chiara G (2002) Differential expression of motivational stimulus Properties by dopamine in nucleus accumbens shell versus core and prefrontal cortex. J Neurosci 22:4709–4719
Bordi F, Carr KD, Meller E (1989) Stereotypies elicited by injection of N-propylnorapomorphine into striatal subregions and nucleus accumbens. Brain Res 489:205–215
Bozarth MA, Wise RA (1981a) Heroin reward is dependent on a dopaminergic substrate. Life Sci 29:1881–1886
Bozarth MA, Wise RA (1981b) Intracranial self-administration of morphine into the ventral tegmental area in rats. Life Sci 28:551–555
Cadoni C, Di Chiara G (1999) Reciprocal changes in dopamine responsiveness in the nucleus accumbens shell and core and in the dorsal caudate-putamen in rats sensitized to morphine. Neuroscience 90:447–455
Cameron DL, Crocker AD (1988) Alkylation of striatal dopamine receptors abolishes stereotyped behavior but has no effect on dopamine stimulated adenylate cyclase activity. Neurosci Lett 90:165–171
Czoty PW, Justice JB Jr, Howell LL (2000) Cocaine-induced changes in extracellular dopamine determined by microdialysis in awake squirrel monkeys. Psychopharmacology (Berl) 148:299–306
David V, Cazala P (1994) A comparative study of self-administration of morphine into the amygdala and the ventral tegmental area in mice. Behav Brain Res 65:205–211
Davis MD, Heffner TG, Cooke LW (1997) Dopamine agonist-induced inhibition of neurotransmitter release from the awake squirrel monkey putamen as measured by microdialysis. J Neurochem 68:659–666
Devine DP, Wise RA (1994) Self-administration of morphine, DAMGO, and DPDPE into the ventral tegmental area of rats. J Neurosci 14:1978–1984
Di Chiara G (1998) A motivational learning hypothesis of the role of mesolimbic dopamine in compulsive drug use. J Psychopharmacol 12:54–67
Di Chiara G, Imperato A (1988) Opposite effects of mu and kappa opiate agonists on dopamine release in the nucleus accumbens and in the dorsal caudate of freely moving rats. J Pharmacol Exp Ther 244:1067–1080
Di Chiara G, Tanda G, Carboni E (1996) Estimation of in-vivo neurotransmitter release by brain microdialysis: the issue of validity. Behav Pharmacol 7:640–657
Dickinson A, Balleine B (1994) Motivational control of goal-directed action. Anim Learn Behav 22:1–18
Dworkin SI, Mirkis S, Smith JE (1995) Response-dependent versus response-independent presentation of cocaine: differences in the lethal effects of the drug. Psychopharmacology (Berl) 117:262–266
Elmer GI, Pieper JO, Rubinstein M, Low MJ, Grandy DK, Wise RA (2002) Failure of intravenous morphine to serve as an effective instrumental reinforcer in dopamine D2 receptor knock-out mice. J Neurosci 22:RC224
Fenu S, Spina L, Rivas E, Longoni R, Di Chiara G (2006) Morphine-conditioned single-trial place preference: role of nucleus accumbens shell dopamine receptors in acquisition, but not expression. Psychopharmacology (Berl) 187:143–153
Fumero B, Guadalupe T, Valladares F, Mora F, O'Neill RD, Mas M, Gonzalez-Mora JL (1994) Fixed versus removable microdialysis probes for in vivo neurochemical analysis: implications for behavioral studies. J Neurochem 63:1407–1415
Georgieva J, Luthman J, Mohringe B, Magnusson O (1993) Tissue and microdialysate changes after repeated and permanent probe implantation in the striatum of freely moving rats. Brain Res Bull 31:463–470
Gerrits MA, Ramsey NF, Wolterink G, van Ree JM (1994) Lack of evidence for an involvement of nucleus accumbens dopamine D1 receptors in the initiation of heroin self-administration in the rat. Psychopharmacology (Berl) 114:486–494
Hemby SE, Martin TJ, Co C, Dworkin SI, Smith JE (1995) The effects of intravenous heroin administration on extracellular nucleus accumbens dopamine concentrations as determined by in vivo microdialysis. J Pharmacol Exp Ther 273:591–598
Hemby SE, Co C, Koves TR, Smith JE, Dworkin SI (1997) Differences in extracellular dopamine concentrations in the nucleus accumbens during response-dependent and response-independent cocaine administration in the rat. Psychopharmacology (Berl) 133:7–16
Hnasko TS, Sotak BN, Palmiter RD (2005) Morphine reward in dopamine-deficient mice. Nature 438:854–857
Kalivas PW, Pierce RC, Cornish J, Sorg BA (1998) A role for sensitization in craving and relapse in cocaine addiction. J Psychopharmacol 12:49–53
Lecca D, Cacciapaglia F, Valentini V, Gronli J, Spiga S, Di Chiara G (2006a) Preferential increase of extracellular dopamine in the rat nucleus accumbens shell as compared to that in the core during acquisition and maintenance of intravenous nicotine self-administration. Psychopharmacology (Berl) 184:435–446
Lecca D, Cacciapaglia F, Valentini V, Di Chiara G (2006b) Monitoring extracellular dopamine in the rat nucleus accumbens shell and core during acquisition and maintenance of intravenous WIN 55,212-2 self-administration. Psychopharmacology (Berl) 188:63–74
Lecca D, Cacciapaglia F, Valentini V, Acquas E, Di Chiara G (2007) Differential neurochemical and behavioral adaptation to cocaine after response contingent and noncontingent exposure in the rat. Psychopharmacology (Berl) 191:653–667
Li Y, Hu XT, Berney TG, Vartanian AJ, Stine CD, Wolf ME, White FJ (1999a) Both glutamate receptor antagonists and prefrontal cortex lesions prevent induction of cocaine sensitization and associated neuroadaptations. Synapse 34:169–180
Li Y, Wolf ME, White FJ (1999b) The expression of cocaine sensitization is not prevented by MK-801 or ibotenic acid lesions of the medial prefrontal cortex. Behav Brain Res 104:119–125
Longoni R, Spina L, Mulas A, Carboni E, Garau L, Melchiorri P, Di Chiara G (1991) (D-Ala2)deltorphin II: DI-dependent stereotypies and stimulation of dopamine release in the nucleus accumbens. J Neurosci 11:1565–1576
Maldonado R, Saiardi A, Valverde O, Samad TA, Roques BP, Borrelli E (1997) Absence of opiate rewarding effects in mice lacking dopamine D2 receptors. Nature 388:586–589
Martin-Fardon R, Sandillon F, Thibault J, Privat A, Vignon J (1997) Long-term monitoring of extracellular dopamine concentration in the rat striatum by a repeated microdialysis procedure. J Neurosci Methods 72:123–135
O’Brien (2001) In: Hardman JG, Oilman AG, Limbird LE (eds) Goodman and Oilman’s the pharmacological basis of therapeutics. McGraw-Hill, New York
Paxinos G, Watson C (1998) The rat brain in stereotaxic coordinates. Academic, Sydney
Pettit HO, Ettenberg A, Bloom FE, Koob GF (1984) Destruction of dopamine in the nucleus accumbens selectively attenuates cocaine but not heroin self-administration in rats. Psychopharmacology (Berl) 84:167–173
Pontieri FE, Tanda G, Di Chiara G (1995) Intravenous cocaine, morphine, and amphetamine preferentially increase extracellular dopamine in the "shell" as compared with the “core” of the rat nucleus accumbens. Proc Natl Acad Sci USA 92:12304–12308
Robinson TE, Berridge KC (1993) The neural basis of drug craving: an incentive-sensitization theory of addiction. Brain Res Brain Res Rev 18:247–291
Spyraki C, Fibiger HC, Phillips AG (1983) Attenuation of heroin reward in rats by disruption of the mesolimbic dopamine nsystem. Psychopharmacology (Berl) 79:278–83
Tanda G, Pontieri FE, Di Chiara G (1997) Cannabinoid and heroin activation of mesolimbic dopamine transmission by a common mu1 opioid receptor mechanism. Science 276:2048–2050
van Ree JM, Ramsey N (1987) The dopamine hypothesis of opiate reward challenged. Eur J Pharmacol 134:239–243
Volkow ND, Fowler JS, Wang GJ (2002) Role of dopamine in drug reinforcement and addiction in humans: results from imaging studies. Behav Pharmacol 13:355–366
Wise RA, Leone P, Rivest R, Leeb K (1995) Elevations of nucleus accumbens dopamine and DOPAC levels during intravenous heroin self-administration. Synapse 21:140–148
Acknowledgments
The authors gratefully acknowledge the supply of the self-administration cages by Dr. Steve Goldberg and Dr. Gianluigi Tanda. This study was supported by funds from Ministero dell’Università e della Ricerca, progetti di Ricerca Nazionale Bando 2003, from the Centre of Excellence for Studies on the Neurobiology of the Addictions, and from the European Commission, NIDE project.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lecca, D., Valentini, V., Cacciapaglia, F. et al. Reciprocal effects of response contingent and noncontingent intravenous heroin on in vivo nucleus accumbens shell versus core dopamine in the rat: a repeated sampling microdialysis study. Psychopharmacology 194, 103–116 (2007). https://doi.org/10.1007/s00213-007-0815-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-007-0815-y