Abstract
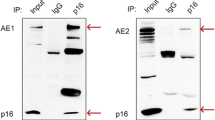
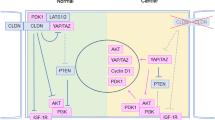
Human anion exchanger 2 (AE2) is a plasma membrane protein that regulates intracellular pH and cell volume. AE2 contributes to transepithelial transport of chloride and bicarbonate in normal colon and other epithelial tissues. We now report that AE2 overexpression in colon cancer cells is correlated with expression of the nuclear proliferation marker, Ki67. Survival analysis of 24 patients with colon cancer in early stage or 33 patients with tubular adenocarcinoma demonstrated that expression of AE2 is correlated with poor prognosis. Cellular and molecular experiments indicated that AE2 expression promoted proliferation of colon cancer cells. In addition, we found that transcription factor EGR1 underlies AE2 upregulation and the AE2 sequester p16INK4a (P16) in the cytoplasm of colon cancer cells. Cytoplasmic P16 enhanced ERK phosphorylation and promoted proliferation of colon cancer cells. Gastrin inhibited proliferation of colon cancer cells by suppressing expression of EGR1 and AE2 and by blocking ERK phosphorylation. Taken together, our data describe a novel EGR1/AE2/P16/P-ERK signaling pathway in colon carcinogenesis, with implications for pathologic prognosis and for novel therapeutic approaches.






Similar content being viewed by others
References
Saif MW, Chu E (2010) Biology of colorectal cancer. Cancer J 16:196–201. doi:10.1097/PPO.0b013e3181e076af
Plotnikov A, Chuderland D, Karamansha Y, Livnah O, Seger R (2011) Nuclear extracellular signal-regulated kinase 1 and 2 translocation is mediated by casein kinase 2 and accelerated by autophosphorylation. Mol Cell Biol 31:3515–3530. doi:10.1128/MCB.05424-11
Kang YH, Yang IJ, Shin HM (2011) Herbal formula HMC05 prevents human aortic smooth muscle cell migration and proliferation by inhibiting the ERK1/2 MAPK signaling cascade. J Nat Med. doi:10.1007/s11418-011-0573-3
She QB, Halilovic E, Ye Q, Zhen W, Shirasawa S, Sasazuki T, Solit DB, Rosen N (2010) 4E-BP1 is a key effector of the oncogenic activation of the AKT and ERK signaling pathways that integrates their function in tumors. Cancer Cell 18:39–51. doi:10.1016/j.ccr.2010.05.023
Alper SL (2006) Molecular physiology of SLC4 anion exchangers. Exp Physiol 91:153–161. doi:10.1113/expphysiol.2005.031765
Alper SL (2009) Molecular physiology and genetics of Na+-independent SLC4 anion exchangers. J Exp Biol 212:1672–1683. doi:10.1242/jeb.029454
Rossmann H, Alper SL, Nader M, Wang Z, Gregor M, Seidler U (2000) Three 5′-variant mRNAs of anion exchanger AE2 in stomach and intestine of mouse, rabbit, and rat. Ann NY Acad Sci 915:81–91
Yang Y, Wu PP, Wu J, Shen WW, Wu YL, Fu AF, Zheng L, Jin XL, Fu GH (2008) Expression of anion exchanger 2 in human gastric cancer. Exp Oncol 30:81–87
Wu TT, Hsieh YH, Wu CC, Tsai JH, Hsieh YS, Huang CY, Liu JY (2006) Overexpression of anion exchanger 2 in human hepatocellular carcinoma. Chin J Physiol 49:192–198
Hwang JM, Kao SH, Hsieh YH, Li KL, Wang PH, Hsu LS, Liu JY (2009) Reduction of anion exchanger 2 expression induces apoptosis of human hepatocellular carcinoma cells. Mol Cell Biochem 327:135–144. doi:10.1007/s11010-009-0051-3
Gawenis LR, Ledoussal C, Judd LM, Prasad V, Alper SL, Stuart-Tilley A, Woo AL, Grisham C, Sanford LP, Doetschman T et al (2004) Mice with a targeted disruption of the AE2 Cl−/HCO3− exchanger are achlorhydric. J Biol Chem 279:30531–30539. doi:10.1074/jbc.M403779200
Frische S, Zolotarev AS, Kim YH, Praetorius J, Alper S, Nielsen S, Wall SM (2004) AE2 isoforms in rat kidney: immunohistochemical localization and regulation in response to chronic NH4Cl loading. Am J Physiol Renal Physiol 286:F1163–F1170. doi:10.1152/ajprenal.00409.2003
Tian H, Zhang N, Suo WH, Wang T, Song LJ, Wu J, Liu Q, Shen WW, Fu GH (2010) Gastrin suppresses the interdependent expression of p16 and anion exchanger 1 favoring growth inhibition of gastric cancer cells. Int J Cancer 127:1462–1474. doi:10.1002/ijc.25124
Shen WW, Wu J, Cai L, Liu BY, Gao Y, Chen GQ, Fu GH (2007) Expression of anion exchanger 1 sequestrates p16 in the cytoplasm in gastric and colonic adenocarcinoma. Neoplasia 9:812–819
Fu GH, Wang Y, Xi YH, Shen WW, Pan XY, Shen WZ, Jiang XS, Chen GQ (2005) Direct interaction and cooperative role of tumor suppressor p16 with band 3 (AE1). FEBS Lett 579:2105–2110. doi:10.1016/j.febslet.2005.02.063
Naik S, Dothager RS, Marasa J, Lewis CL, Piwnica-Worms D (2009) Vascular endothelial growth factor receptor-1 is synthetic lethal to aberrant {beta}-catenin activation in colon cancer. Clin Cancer Res 15:7529–7537. doi:10.1158/1078-0432.CCR-09-0336
Munne A, Fabre M, Marinoso ML, Gallen M, Real FX (1999) Nuclear beta-catenin in colorectal tumors: to freeze or not to freeze? Colon cancer team at IMAS. J Histochem Cytochem 47:1089–1094
Zhou C, Tiberi M, Liang B, Alper SL, Baltz JM (2009) HCO3(−)/Cl(−) exchange inactivation and reactivation during mouse oocyte meiosis correlates with MEK/MAPK-regulated Ae2 plasma membrane localization. PLoS One 4:e7417. doi:10.1371/journal.pone.0007417
Vares G, Uehara Y, Ono T, Nakajima T, Wang B, Taki K, Matsumoto T, Oghiso Y, Tanaka K, Ichinohe K et al (2011) Transcription factor-recognition sequences potentially involved in modulation of gene expression after exposure to low-dose-rate gamma-rays in the mouse liver. J Radiat Res (Tokyo) 52:249–256
Vindis C, Seguelas MH, Lanier S, Parini A, Cambon C (2001) Dopamine induces ERK activation in renal epithelial cells through H2O2 produced by monoamine oxidase. Kidney Int 59:76–86. doi:10.1046/j.1523-1755.2001.00468.x
Baron V, Adamson ED, Calogero A, Ragona G, Mercola D (2006) The transcription factor Egr1 is a direct regulator of multiple tumor suppressors including TGFbeta1, PTEN, p53, and fibronectin. Cancer Gene Ther 13:115–124. doi:10.1038/sj.cgt.7700896
Zagurovskaya M, Shareef MM, Das A, Reeves A, Gupta S, Sudol M, Bedford MT, Prichard J, Mohiuddin M, Ahmed MM (2009) EGR-1 forms a complex with YAP-1 and upregulates Bax expression in irradiated prostate carcinoma cells. Oncogene 28:1121–1131. doi:10.1038/onc.2008.461
Hong Y, Ho KS, Eu KW, Cheah PY (2007) A susceptibility gene set for early onset colorectal cancer that integrates diverse signaling pathways: implication for tumorigenesis. Clin Cancer Res 13:1107–1114. doi:10.1158/1078-0432.CCR-06-1633
Wilson AJ, Chueh AC, Togel L, Corner GA, Ahmed N, Goel S, Byun DS, Nasser S, Houston MA, Jhawer M et al (2010) Apoptotic sensitivity of colon cancer cells to histone deacetylase inhibitors is mediated by an Sp1/Sp3-activated transcriptional program involving immediate-early gene induction. Cancer Res 70:609–620. doi:10.1158/0008-5472.CAN-09-2327
Mahalingam D, Natoni A, Keane M, Samali A, Szegezdi E (2010) Early growth response-1 is a regulator of DR5-induced apoptosis in colon cancer cells. Br J Cancer 102:754–764. doi:10.1038/sj.bjc.6605545
Gawenis LR, Bradford EM, Alper SL, Prasad V, Shull GE (2010) AE2 Cl−/HCO3− exchanger is required for normal cAMP-stimulated anion secretion in murine proximal colon. Am J Physiol Gastrointest Liver Physiol 298:G493–G503. doi:10.1152/ajpgi.00178.2009
Currid A, Ortega B, Valverde MA (2004) Chloride secretion in a morphologically differentiated human colonic cell line that expresses the epithelial Na+ channel. J Physiol 555:241–250. doi:10.1113/jphysiol.2003.059295
Rajendran VM, Binder HJ (2000) Characterization and molecular localization of anion transporters in colonic epithelial cells. Ann NY Acad Sci 915:15–29
Pientong C, Ekalaksananan T, Swadpanich U, Kongyingyoes B, Kritpetcharat O, Yuenyao P, Ruckait N (2003) Immunocytochemical detection of p16INK4a protein in scraped cervical cells. Acta Cytol 47:616–623
Liu Q, Song LJ, Xu WQ, Zhao L, Zheng L, Yan ZW, Fu GH (2009) Expression of cytoplasmic p16 and anion exchanger 1 is associated with the invasion and absence of lymph metastasis in gastric carcinoma. Mol Med Report 2:169–174. doi:10.3892/mmr_00000079
Emig R, Magener A, Ehemann V, Meyer A, Stilgenbauer F, Volkmann M, Wallwiener D, Sinn HP (1998) Aberrant cytoplasmic expression of the p16 protein in breast cancer is associated with accelerated tumour proliferation. Br J Cancer 78:1661–1668
Feng XL, Li L, Gao YN, Zhang JJ, Xiao T, Ying JM, Gao JD, Sun YT, Cheng SJ (2011) Overexpression of c-erbB-2 and loss of p16 have molecular diagnostic relevance but no prognostic value in lung cancer. Med Oncol 28:336–341. doi:10.1007/s12032-010-9452-0
Kudahetti SC, Fisher G, Ambroisine L, Prowse D, Kattan MW, Foster CS, Moller H, Oliver T, Fletcher A, Cooper C et al (2010) Immunohistochemistry for p16, but not Rb or p21, is an independent predictor of prognosis in conservatively treated, clinically localised prostate cancer. Pathology 42:519–523. doi:10.3109/00313025.2010.508788
Pintus G, Tadolini B, Posadino AM, Sanna B, Debidda M, Carru C, Deiana L, Ventura C (2003) PKC/Raf/MEK/ERK signaling pathway modulates native-LDL-induced E2F-1 gene expression and endothelial cell proliferation. Cardiovasc Res 59:934–944
Fenton JI, Hursting SD, Perkins SN, Hord NG (2006) Interleukin-6 production induced by leptin treatment promotes cell proliferation in an Apc (Min/+) colon epithelial cell line. Carcinogenesis 27:1507–1515. doi:10.1093/carcin/bgl018
Acknowledgments
The authors thank China Zhenjiang First People’s Hospital for providing the 202 paraffin-embedded colon cancer specimens. pcDNA3.0-EGR1 construct were kindly provided by Ian de Belle (Laval University, Canada). This work was supported in part by the National Natural Science Foundation of China (NO30570697; NO30770960), National High Technology Research and Development Program of China (863 Program; NO2008AA02Z120), Shanghai Natural Science Foundation (NO11ZR1419700).
Conflict of interest
The authors declare no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
Ling-Jun Song and Rui-Jun Liu contributed equally to this work.
Rights and permissions
About this article
Cite this article
Song, LJ., Liu, RJ., Zeng, Z. et al. Gastrin inhibits a novel, pathological colon cancer signaling pathway involving EGR1, AE2, and P-ERK. J Mol Med 90, 707–718 (2012). https://doi.org/10.1007/s00109-011-0851-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00109-011-0851-2