Summary
We have studied the pharmacokinetics of oxybutynin (Ditropan) after single oral (5 mg) and intravenous administration (1 and 5 mg), and after repeated oral administration in healthy volunteers.
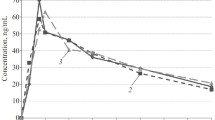
Oxybutynin was rapidly absorbed, maximum plasma concentrations (8 ng·ml−1) being reached in less than 1 h. The absolute systemic availability averaged 6% and the tablet and solution forms displayed similar relative systemic availability.
Plasma concentrations of oxybutynin fell biexponentially, the elimination half-life being about 2 h. There was a large interindividual variation in oxybutynin plasma concentrations. Almost no intact drug could be recovered in the urine. During repeated oral administration steady-state was reached after eight days of treatment.
The low absolute systemic availability of oxybutynin, the large interindividual variability in its plasma concentrations, and the apparent absence of intact oxybutynin in the urine suggest that its major pathway of elimination is hepatic metabolism.
Similar content being viewed by others
References
Lish PM, La Budde JA, Peeters EL, Robbins SI (1967) Oxybutynin, a musculotropic antispasmodic drug with moderate anticholinergic action. Arch Int Pharmacodyn Ther 156: 467–488
Fredericks CM, Anderson GF, Kreulen DJ (1975) Study of the anticholinergic and antispasmodic activity of oxybutynin (Ditropan) on rabbit detrusor. Invest Urol 12: 317–319
Diokno AC, Lapides J (1972) Oxybutynin: A new drug with analgesic and anticholinergic properties. J Urol 108: 307–309
Lindeke B, Brotell H, Karlen B, Rietz G, Vietorisz A (1981) Determination of oxybutynin (4-diethylaminobut-2 ynyl-2-cyclo-hexyl-2-phenylglycolate) in serum and urine by gas chromatography/mass spectrometry with single ion detection. Acta Pharm Suec 18: 25–34
Aaltonen L, Allonen H, Iisalo E, Juhakoski A, Kleimola T, Sellman R (1984) Antimuscarinic activity of oxybutynin in the human plasma quantitated by a radioreceptor assay. Acta Pharmacol Toxicol 55: 100–103
Society of Actuaries (1959) Build and blood pressure study. Society of Actuaries, vol 1, Chicago
Mahgoub A, Idle JR, Dring LG, Lancaster R, Smith RL (1977) Polymorphic hydroxylation of debrisoquine in man. Lancet 2: 584–586
Gangji D, Juvent M, Douchamps J, Herchuelz A (1985) Oxidation of nifedipine and debrisoquine: Two distinct types of genetic polymorphism. Joint meeting of the Belgian, Dutch and German Pharmacological and Toxicological Societies. Aachen, FRG, Sept 23–26
Loo JCK, Riegelman S (1970) Assessment of pharmacokinetic constants from post infusion blood curves obtained after i.v. infusion. J Pharm Sci 59: 53–55
Westlake WJ (1976) Symmetrical confidence intervals for bioequivalence trials. Biometrics 32: 741–744
Lindeke B, Hallstrom G, Johansson C, Ericsson A, Olsson L-I, Stromberg S (1981) Metabolism of oxybutynin: Establishment of desethyloxybutynin and oxybutynin N-oxide formation in rat liver preparations using deuterium substitution and gas chromatographic mass spectrometric analysis. Biomed Mass Spectrometry 8: 506–513
Kupfer A, Preisig R (1984) Pharmacogenetics of mephenytoin: A new drug hydroxylation polymorphism in man. Eur J Clin Pharmacol 26: 753–759
Waring RH, Mitchell SC, Shah RR, Idle JR, Smith RL (1982) Polymorphic sulphoxidation of S-carboxymethyl-L-cysteine in man. Biochem Pharmacol 31: 3151–3154
Scott J, Poffenbarger PL (1979) Pharmacogenetics of tolbutamide metabolism in man. Diabetes 28: 41–54
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Douchamps, J., Derenne, F., Stockis, A. et al. The pharmacokinetics of oxybutynin in man. Eur J Clin Pharmacol 35, 515–520 (1988). https://doi.org/10.1007/BF00558247
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00558247