Summary
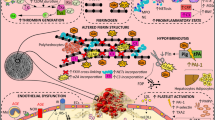
Numerous studies have shown that coagulation abnormalities occur in the course of diabetes mellitus, resulting in a state of thrombophilia. These observations are supported by epidemiological studies which demonstrate that thromboembolic events are more likely to occur in diabetic patients. The coagulation abnormalities observed in diabetic patients seem to be caused by the hyperglycaemia, which also constitutes the distinguishing feature of this disease. These data are also supported by in vitro studies which demonstrate how glucose can directly determine alterations in the coagulation system. The abnormalities observed involve all stages of coagulation, affecting both thrombus formation and its inhibition, fibrinolysis, platelet and endothelial function. The final result is an imbalance between thrombus formation and dissolution, favouring the former. Hyperglycaemia probably determines the onset of these abnormalities through three mechanisms which are, respectively, non-enzymatic glycation, the development of increased oxidative stress and a decrease in the levels of heparan sulphate. The first seems to affect the functionality of key molecules of coagulation in a negative sense. Oxidative stress constitutes an important pro-thrombotic stimulus, while the decrease in heparan sulphate determines a reduction in antithrombotic defenses. Good metabolic control could play a key role in controlling the coagulation irregularities in diabetes. However, considering the difficulties in achieving such an objective, it is possible that the use of drugs may represent a valid alternative. In fact, several drugs exist which are of potential interest. It is, however, necessary to perform long-term studies which demonstrate unequivocably that by controlling the coagulation abnormalities in diabetic patients, prolongation of life is possible.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Colwell JA (1993) Vascular thrombosis in type II diabetes mellitus. Diabetes 42: 8–11
Theroux P, Latour JG, Leger-Gauthier C, De Lara J (1987) Fibrinopeptide A and platelet factor levels in unstable angina pectoris. Circulation 75: 156–162
Kruskal JB, Commerford PJ, Franks JJ, Kirsch RE (1987) Fibrin and fibrinogen-related antigens in patients with stable and unstable coronary artery disease. N Engl J Med 317: 1361–1365
Kannel WB, Mc Gee DL (1979) Diabetes and cardiovascular disease. The Framingham study. JAMA 241: 2035–2038
Nossel HL, Yudelman J, Canfield RE et al. (1974) Measurement of fibrinopeptide A in human blood. J Clin Invest 54: 43–53
Rabie MJ, Blashill A, Furie BC (1986) Prothrombin fragment 1.2.3, a major product of prothrombin activation in human plasma. J Biol Chem 261: 13210–13215
Rosove MH, Frank HSL, Harving SSL (1984) Plasma betathromboglobulin, platelet factor 4, fibrinopeptide A, and other hemostatic functions during improved short-term glycemic control in diabetes mellitus. Diabetes Care 7: 174–179
Ceriello A, Giacomello R, Colatutto A, Taboga C, Gonano F (1992) Increased prothrombin fragment 1+2 in type I diabetic patients. Haemostasis 22: 50–51
Jones RL (1985) Fibrinopeptide A in diabetes mellitus: relation to levels of blood glucose, fibrinogen disappearance, and hemodynamic changes. Diabetes 34: 836–841
Ceriello A, Giugliano D, Quatraro A, Dello Russo P, Marchi E, Torella R (1989) Hyperglycemia may determine fibrinopeptide A plasma level increase in humans. Metabolism 38: 1162–1163
Jones RL, Peterson CM (1979) Reduced fibrinogen survival in diabetes mellitus: a reversible phenomenon. J Clin Invest 63: 485–493
Wilhelmsen L, Svardsudd K, Korsan-Bengtensen K, Larsson B, Welin L, Tibblin G (1984) Fibrinogen as a risk factor for stroke and myocardial infarction. N Engl J Med 311: 501–505
Ganda OP, Arkin CF (1992) Hyperfibrinogenemia. An important risk factor for vascular complications in diabetes. Diabetes Care 15: 1245–1250
Coller BS, Frank RN, Milton RC, Gralnick HR (1978) Plasma cofactors of platelet function: correlation with diabetic retinopathy and hemoglobin A1a-c. Ann Intern Med 88: 311–316
Meade JW, Brozovic M, Chakrabarti RR et al. (1986) Haemostatic function and ischemic heart disease: principal results of the Northwick Park Heart Study. Lancet II: 533–537
Ceriello A, Giugliano D, Quatraro A, Dello Russo P, Torella R (1988) Blood glucose may condition factor VII levels in diabetic and normal subjects. Diabetologia 31: 889–891
Balleisen L, Assmann G, Bailey J (1985) Epidemiological study on factor VII, factor VIII and fibrinogen in an industrial population. Baseline data on the relation to blood pressure, blood glucose, uric acid and lipid fractions. Thromb Haemostas 54: 721–723
Ceriello A, Quatraro A, Marchi E, Barbanti M, Dello Russo P, Lefebvre PJ (1990) The role of hyperglycaemia-induced alterations of antithrombin III and factor X activation in the thrombin hyperactivity of diabetes mellitus. Diabetic Med 7: 343–348
Ostermann H, van de Loo J (1986) Factors of the hemostatic system in diabetic patients. A survey of controlled studies. Haemostasis 16: 386–416
Ceriello A, Dello Russo P, Zuccotti C et al. (1983) Decreased antithrombin III activity in diabetes may be due to non-enzymatic glycosylation. A preliminary report. Thromb Haemostas 50: 633–634
Ceriello A, Giugliano D, Quatraro A et al. (1987) Daily rapid blood glucose variations may condition antithrombin III biologic activity but not its plasma concentration in insulin-dependent diabetes. Diabet Metab 13: 16–19
Ceriello A, Giugliano D, Quatraro A et al. (1987) Induced hyperglycemia alters antithrombin III activity but not its plasma concentration in healthy normal subjects. Diabetes 36: 320–323
Ceriello A, Giugliano D, Dello Russo P, Tirelli A, Passariello N, Sgambato S (1986) Metabolic control may alter antithrombin III activity but not its plasma concentration in diabetes: a possible role for nonenzymatic glycosylation. Diabetes Care 9: 32–35
Husted SE, Nielsen HK, Bak JF, Beck-Nielsen H (1989) Antithrombin III, von Willebrand factor antigen and platelet function in young diabetic patients treated with multiple insulin injections versus insulin pump treatment. Eur J Clin Invest 19: 90–94
Ceriello A, Giugliano D, Quatraro A, Marchi E, Barbanti M, Lefèbvre PJ (1990) Evidence for a hyperglycaemia-dependent decrease of antithrombin III-thrombin complex formation in humans. Diabetologia 33: 163–167
Ceriello A, Quatraro A, Dello Russo P, Marchi E, Milani MR, Giugliano D (1990) Evidence for a reduced heparin cofactor II biological activity in diabetes. Haemostasis 20: 357–361
Brownlee M (1976) Alpha-2-macroglobulin and reduced basement membrane degradation in diabetes. Lancet I: 779–780
Ceriello A, Giugliano D, Quatraro A, Marchi E, Barbanti M, Giugliano D (1989) Increased alpha-2-macroglobulin in diabetes: a hyperglycemia-related phenomenon associated with reduced antithrombin III activity. Acta Diabetol Lat 26: 147–154
Ceriello A, Quatraro A, Dello Russo P, Marchi E, Barbanti M, Giugliano D (1989) Hyperglycemia-conditioned increase in alpha-2-macroglobulin in healthy normal subjects: a phenomenon correlated with deficient antithrombin III activity. Acta Haemat 82: 61–63
Vukovich TC, Schernthaner G (1986) Decreased protein Clevels in patients with insulin-dependent type I diabetes mellitus. Diabetes 35: 617–619
Ceriello A, Quatraro A, Dello Russo P et al. (1990) Protein C deficiency in insulin-dependent diabetes: a hyperglycemia-related phenomenon. Thromb Haemostas 64: 104–107
De Fouwuj NS, Haverkate P, Bertina RM, Koopma T, van Wijngaarden A, von Hisbergh VW (1986) The cofactor role of protein S in the acceleration of whole blood clot lysis. Blood 67: 1192–1196
Ceriello A, Giugliano D, Quatraro A, Marchi E, Barbanti E, Lefebvre PJ (1990) Possible role for increased C4b-binding protein level in acquired protein S deficiency in type I diabetes. Diabetes 39: 447–449
Davis JW, Hartman CR, Davis RF, Kyner JL, Lewis HD, Phillips PE (1982) Platelet aggregate ratio in diabetes mellitus. Acta Haemat 67: 222–224
Burrows AW, Chavin SI, Hockaday TDR (1978) Plasma thromboglobulin concentrations in diabetes mellitus. Lancet I: 235–237
Daví G, Rini GB, Averna M et al. (1982) Enhanced platelet release reaction in insulin-dependent and insulin-independent diabetic patients. Haemostasis 12: 275–281
Alessandrini P, Mc Rae J, Feman S, Fitzgerald GA (1988) Thromboxane biosynthesis and platelet function in type I diabetes mellitus. N Engl J Med 319: 208–212
Davi G, Catalano I, Averna M, Notarbartolo A, Strano A, Ciabattoni G, Patrono C (1990) Thromboxane biosynthesis and platelet function in type II diabetes mellitus. N Engl J Med 322: 1769–1774
Mayfield RK, Haluskka PV, Wohltmann HS et al. (1985) Platelet function during continuous insulin infusion treatment in insulin-dependent diabetic patients. Diabetes 34: 1127–1133
Juhan I, Buonocore M, Jouve R, Vague P, Moulin JP, Vialettes B (1982) Abnormalities of erythrocyte deformability and platelet aggregation in insulin-dependent diabetics corrected by insulin in vivo and in vitro. Lancet I: 535–537
Collier A, Tymkewycz P, Armstrong R, Young RJ, Jones RL, Clarke BF (1986) Increased platelet thromboxane receptor sensitivity in diabetic patients with proliferative retinopathy. Diabetologia 29: 471–474
Di Minno G, Silver MJ, Cerbone AM, Riccardi G, Rivellese A, Mancini M (1986) Platelet fibrinogen binding in diabetes mellitus: differences between binding to platelets from retinopathic and retinopathic diabetic patients. Diabetes 35: 182–185
Betteridge DJ, El Tahir KEH, Reckless JPD, Williams KS (1982) Platelets from diabetic subjects show diminished sensitivity to prostacyclin. Eur J Clin Invest 12: 395–398
Winocour PD, Halushka PV, Colwell JA (1985) Platelet involvement in diabetes mellitus. In: Longenecker GL (ed) The platelets: physiology and pharmacology. Academic Press, New York, pp 341–366
Jones RL, Peterson CM (1981) Hematologic alterations in diabetes mellitus. Am J Med 70: 339–352
Ceriello A, Dello Russo P, Sgambato S, Giugliano D (1982) Glycosylated haemoglobin and reticulocyte count in diabetes. Diabetologia 22: 223 (Letter)
Graham JJ, Ryall RG, Wise PH (1980) Glycosylated haemoglobin and relative polycythaemia in diabetes mellitus. Diabetologia 18: 205–207
Ceriello A, Dello Russo P, Curcio F, Balsamo C, Pietrantuono C (1983) Red blood cell volume and glycaemic control in diabetes. Diabetologia 24: 397 (Letter)
Juhan-Vague I, Alessi MC, Vague P (1991) Increased plasma plasminogen activator inhibitor 1 levels. A possible link between insulin resistance and atherothrombosis. Diabetologia 34: 457–462
Walmsley D, Hampton KK, Grant PJ (1991) Contrasting fibrinolytic responses in type I (insulin dependent) and type II (non-insulin dependent) diabetes. Diabetic Med 8: 954–959
Ceriello A, Quatraro A, Marchi E, Barbanti M, Giugliano D (1993) Impaired fibrinolytic response to increased thrombin activation in type I diabetes mellitus: the effect of the glycosaminoglycan sulodexide. Diabet Metab 19: 225–229
Miles LA, Fless GM, Levin EG, Scanu AM, Plow EF (1989) A potential basis for the thrombotic risk associated with lipoprotein (a). Nature 339: 301–303
Ramirez LC, Arauz-Pacheco C, Lackner C, Albright G, Adams BV, Raskin P (1992) Lipoprotein (a) levels in diabetes mellitus: relationship to metabolic control. Ann Intern Med 117: 42–47
Stern DM, Esposito C, Gerlach H et al. (1991) Endothelium and regulation of coagulation. Diabetes Care 14 [Suppl. 1]: 160–166
Ono H, Umeda F, Inoguchi T, Ibayashi H (1988) Glucose inhibits prostacyclin production by cultured aortic endothelial cells. Thromb Haemostas 60: 174–177
Boeri D, Almus FE, Maiello M, Cagliero E, Rao LVM, Lorenzi M (1989) Modification of tissue-factor mRNA and protein response to thrombin and interleukin 1 by high glucose in cultured human endothelial cells. Diabetes 38: 212–218
Maiello M, Boeri D, Podestà F et al. (1992) Increased expression of tissue plasminogen activator and its inhibitor and reduced fibrinolytic potential of human endothelial cells cultured in elevated glucose. Diabetes 41: 1009–1015
Borch-Johnsen K, Kreiner S (1987) Proteinuria: value as a predictor of cardiovascular mortality in insulin-dependent diabetes mellitus. BMJ 294: 1651–1654
Mogensen CE (1984) Microalbuminuria predicts clinical proteinuria and early mortality in maturity-onset diabetes mellitus. N Engl J Med 310: 356–360
Stehouwer CDA, Nanta JJP, Zeldenrust GC, Hackeng WHL, Donker AJA, Den Ottolander GJH (1992) Urinary albumin excretion, cardiovascular disease, and endothelial dysfunction in non-insulin-dependent diabetes mellitus. Lancet II: 319–323
Jones SL, Close CF, Mattock MB, Jarrett RJ, Keen H, Viberti GC (1989) Plasma lipid and coagulation factor concentrations in insulin dependent diabetes with microalbuminuria. BMJ 298: 487–490
Villanueva GB, Allen N (1988) Demonstration of altered antithrombin III activity due to non enzymatic glycosylation at glucose concentration expected to be encountered in severely diabetic patients. Diabetes 37: 1103–1107
Ceriello A, Giugliano D, Quatraro A et al. (1986) Heparin preserves antithrombin III biological activity from hyperglycemia-induced alterations in insulin-dependent diabetics. Haemostasis 16: 458–464
Ceriello A, Marchi E, Palazzini E, Quatraro A, Giugliano D (1990) Low molecular weight heparin restores antithrombin III activity from hyperglycemia induced alterations. Diabet Metab 16: 86–92
Ceriello A, Marchi E, Barbanti M et al. (1990) Non-enzymatic glycation reduces heparin cofactor II anti-thrombin activity. Diabetologia 33: 205–207
Brownlee M, Vlassara H, Cerami A (1983) Nonenzymatic glycosylation reduces the susceptibility of fibrin to degradation by plasmin. Diabetes 32: 600–604
Winocour PD, Watala C, Kinlough-Rathbone RL (1992) Membrane fluidity is related to the extent of glycation of proteins, but not to alterations in the cholesterol to phospholipid molar ratio in isolated platelet membranes from diabetic and control subjects. Thromb Haemostas 65: 567–571
Bryszewska M, Szosland K (1988) Association between the glycation of erythrocyte membrane proteins and membrane fluidity. Clin Biochem 21: 49–51
Esposito C, Gerlach H, Brett J, Stern D, Vlassara H (1989) Endothelial receptor-mediated binding of glucose-modified albumin is associated with increased monolayer permeability and modulation of cell surface coagulant properties. J Exp Med 170: 1387–1407
Baynes JW (1991) Role of oxidative stress in development of complications in diabetes. Diabetes 40: 405–412
Ceriello A, Quatraro A, Giugliano D (1992) New insights on non-enzymatic glycosylation may lead to therapeutic approaches for the prevention of diabetic complications. Diabetic Med 9: 297–299
Ceriello A, Giugliano D, Quatraro A, Dello Russo P, Lefebvre PJ (1991) Metabolic control may influence the increased Superoxide anion generation in diabetic serum. Diabetic Med 8: 540–542
Barrowcliffe TW, Gutteridge JM, Gray E (1987) Oxygen radicals, lipid peroxidation and the coagulation system. Agent Actions 22: 347–348
Collier A, Rumley AG, Paterson JR, Leach JP, Lowe GDO, Small M (1992) Free radical activity and hemostatic factor in NIDDM patients with and without microalbuminuria. Diabetes 41: 909–913
Gray E, Barrowcliffe TW (1985) Inhibition of antithrombin III by lipid peroxides. Thromb Res 37: 241–250
Jain SK, Levine SN, Duett J, Hollier B (1991) Reduced vitamin E and increased lipofuscin products in erythrocytes of diabetic rats. Diabetes 40: 1241–1244
Karpen CW, Pritchard KA Jr, Arnold JH, Cornwell DG, Panganamala RV (1982) Restoration of prostacyclin thromboxane A2 balance in the diabetic rat: influence of dietary vitamin E. Diabetes 31: 947–951
Curcio F, Ceriello A (1992) Decreased cultured endothelial cell proliferation in high glucose medium is reversed by antioxidants: new insights on the pathophysiological mechanism of diabetic vascular complications. In Vitro Cell Dev Biol 28: 787–790
Karpen CW, Merola AJ, Trewyn RW, Cornwell DG, Pangamala RV (1981) Modulation of platelet thromboxane A2 and arterial prostacyclin by dietary vitamin E. Prostaglandins 22: 651–661
Rohrbach DH, Wagner CW, Star VL, Martin GR, Brown KS, Yoon JW (1983) Reduced synthesis of basement membrane heparan-sulfate proteoglycan in streptozotocin-induced diabetic mice. J Biol Chem 258: 11672–11677
Witmer MR, Hadcock SJ, Peltier SL, Winocour PD, Richardson M, Hatton MW (1992) Altered levels of antithrombin III and fibrinogen in the aortic wall of the alloxan-induced diabetic rabbit: evidence of a prothrombotic state. J Lab Clin Med 119: 221–230
Raz I, Havivi Y, Yarom R (1988) Reduced negative surface charge on arterial endothelium of diabetic rats. Diabetologia 31: 618–620
Moran A, Brown DM, Kin Y, Klein DJ (1991) Effects of IGF-1 and glucose on protein and proteoglycan synthesis by human fetal mesangial cells in culture. Diabetes 40: 1346–1355
Kashihara N, Wanatabe Y, Makino H, Wellner EI, Kanwar YS (1992) Selective decreased de novo synthesis of glomerular proteoglycans under the influence of reactive oxygen species. Proc Natl Acad Sci USA 89: 6309–6313
Gisinger C, Jeremy J, Speiser P, Mikhailidis D, Daudona P, Schernthaner G (1988) Effect of vitamin E supplementation on platelet thromboxane A2 production in type I diabetic patients. Double-blind crossover trial. Diabetes 37: 1260–1264
Gavish D, Breslow JL (1991) Lipoprotein (a) reduction by Nacetylcysteine. Lancet I: 203–204
Klaff LJ, Kernoff L, Vinik AI, Jackson WPU, Jacobs P (1981) Sulfonylureas and platelet function. Am J Med 70: 627–630
Gram J, Jesperson J, Kold A (1988) Effects of oral antidiabetic drug on the fibrinolytic system of blood in insulin-treated diabetic patients. Metabolism 37: 937–943
Ceriello A, Quatraro A, Ettorre M, Marchi E, Barbanti M, Giugliano D (1993) Glucosaminoglycans administration decreases high fibrinogen plasma levels in diabetic patients. Diab Nutr Metab 6
Durrington PN, Winocour PH, Bhatuagar D (1990) Bezafibrate retard in patients with insulin-dependent diabetes: effects on serum lipoproteins, fibrinogen, and glycemic control. J Cardiovasc Pharmacol 16 [Suppl 9]: 30–34
Mathur S, Barrades MA, Mikhailidis DP, Dandona P (1990) The effect of slow release formulation of bezafibrate on lipids, glucose homeostasis, platelets and fibrinogen in type II diabetics; a pilot study. Diabetes Res 14: 133–138
Leaf A, Weber PC (1988) Cardiovascular effect of n-3 fatty acids. N Engl J Med 318: 549–557
Malasonos TH, Stacpoole PW (1991) Biological effects of omega-3 fatty acids in diabetes mellitus. Diabetes Care 14: 1160–1179
Simonutti M, Ceriello A, Taboga C, Faletti E, Giacomello R (1993) Effect of omega 3 fatty acids on lipid pattern in type II diabetic subjects. In: European Atherosclerosis Society “Thrombosis and the Vessel Wall Highlights'93”. Ariello Bros Press, Naples, p 161 (Abstract)
Cirillo F, Margaglione M, Vecchione G et al. (1991) In vitro inhibition by defibrotide of monocyte Superoxide anion generation: a possible mechanism for the antithrombotic effect of a polydeoxyribonucleotide-derived drug. Haemostasis 21: 98–105
Singer DE, Nathan DM, Anderson KM, Wilson PWF, Evans JC (1992) Association of HbA1c with prevalent cardiovascular disease in the original cohort of the Framingham Heart Study. Diabetes 41: 202–208
Giugliano D, Quatraro A, Minei A et al. (1992) Does a positive family history of diabetes convey a more atherogenetic blood profile? Diabetologia 35 [Suppl 1]: A 93 (Abstract)
Riemersa RA, Wood DA, Macintyre CCA, Elton RA, Gey KF, Oliver MF (1991) Risk of angina pectoris and plasma concentrations of vitamins A, C and E and carotene. Lancet I: 1–5
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Ceriello, A. Coagulation activation in diabetes mellitus: the role of hyperglycaemia and therapeutic prospects. Diabetologia 36, 1119–1125 (1993). https://doi.org/10.1007/BF00401055
Issue Date:
DOI: https://doi.org/10.1007/BF00401055