Summary
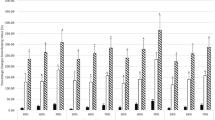
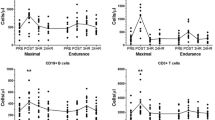
Ten healthy sedentary subjects [age, 27.5 (SD 3.5) years; height, 180 (SD 5) cm; mass, 69.3 (SD 6.3) kg] performed two periods of maximal incremental graded cycle ergometer exercise in a supine position. Randomly ordered and using an open spirometric system, one exercise was carried out during normoxia [maximal oxygen consumption (\(\dot V\)O2max)=38.6 (SD 3.5) ml·min−1·kg−1; maximal blood lactate concentration, 9.86 (SD 1.85) mmol·l−1; test duration, 22.6 (SD 2.7) min], the other during hypoxia [\(\dot V\)O2max=33.2 (SD 3.2) ml·min−1· kg−1; maximal blood lactate concentration, 10.38 (SD 2.02) mmol·l−1; test duration, 19.7 (SD 2.8) min]. At rest, immediately (0 p) and 60 min (60 p) after exercise, counts of leucocyte subpopulations (flow cytometry), cortisol and catecholamine concentrations were determined. At 0 p in contrast to normoxia, during hypoxia there was no significant increase of granulocytes. There were no significant differences between normoxia and hypoxia in the increases from rest to 0 p in counts of monocytes, total lymphocytes and lymphocyte subpopulations [clusters of differentiation (CD), CD3+, CD4+CD45RO−, CD4+CD45RO+, CD8+CD45RO−, CD8+CD45RO+, CD3+HLA-DR+, CD3−CD16/CD56+, CD3+CD16/CD56+, CD 19+] as well as adrenaline, noradrenaline and cortisol concentrations. The counts of CD3 −CD16/CD56+-and CD8 +CD45RO+-cells increased most. At 60 p, CD3−CD16/CD56+ and CD3+CD16/CD56+-cell counts were below pre-exercise levels and under hypoxia slightly but significantly lower than under normoxia. We concluded that the exercise-induced mobilization and redistribution of most leucocyte and lymphocyte subpopulations were unimpaired under acute hypoxia at sea level. Reduced increases of granulocyte counts during the study and reduced cell numbers of natural killer cells and cytotoxic, not major histocompatibility complex-restricted T-cells, only indicated marginal effects on the immune system.
Similar content being viewed by others
References
Akbar AN, Salmon M, Janossy G (1991) The synergy between naive and memory T cells during activation. Immunol Today 12:184–188
Berry IL, Mitchell RB (1983) Influence of simulated altitude on resistence-susceptibility to S. typhimurium infection in mice. Tex Rep Biol Med 11:379–401
Buskirk ER, Kollias J, Picon-Reatequi E (1966) Physiology and performance of track athletes at various altitudes in the Unites States and Peru. In: Goddard RT (ed) The international symposium on the effects of altitude on physical performance. The Athletic Institute, Chicago
Da Prada M, Zürcher G (1976) Simultanous radioenzymatic determination of plasma and tissue adrenaline, noradrenaline, and dopamine within the femtomole range. Life Sci 19:1161–1174
Deuster PA, Curiale AM, Cowan ML, Finkelman FD (1988) Exercise-induced changes in populations of peripheral blood mononuclear cells. Med Sci Sports Exerc 20:276–280
Dill DB, Costill DL (1974) Calculation of percentage changes of blood, plasma, and red cells in dehydration. J Appl Physiol 37:247–248
Fiatorone MA, Morley JE, Bloom ET, Benton D, Solomon GF, Makinodan T (1989) The effect of exercise on natural killer cell activity in young and old subjects. J Gerontol 44:M37-M45
Gabriel H, Urhausen A, Kindermann W (1991) Circulating leucocyte and lymphocyte subpopulations before and after an exhaustive, intensive endurance exercise. Eur J Appl Physiol 63:449–457
Gabriel H, Urhausen A, Kindermann W (1992a) Mobilization of circulating leucocyte and lymphocyte subpopulations during and after a short, anaerobic exercise. Eur J Appl Physiol 65:164–170
Gabriel H, Steffens G, Schwarz L, Kindermann W (1992b) Immunoregulatory hormones, circulating leucocyte and lympho cyte subpopulations before and after endurance exercise of different intensities. Int J Sports Med 13:359–366
Gordon FB, Gillmore JD (1974a) Parabosis and experimental infections 4. Effect of varying O2 tensions on chlamydial infections in mice and cell cultures. Aerosp Med 45:257–262
Gordon FB, Gillmore JD. (1974b) Parabosis and experimental infections. Effect of varying O2 tensions on influenza virus infctions in mice. Aerosp Med 45:241–248
Hohorst HJ (1962) L-(+)-Lactat, Bestimmung mit Lactatdehydrogenase and DPN. In: Bergmeyer HU (ed) Methoden der enzymatischen Analyse. Verlag Chemie, Weinheim, pp 266–277
Jackson CGR, Sharkey BJ (1988) Altitude, training and human performance. Sports Med 6:279–284
Keast D, Cameron K, Morton AR (1988) Exercise and the immune response. Sports Med 5:248–267
Kitayev MI, Tokhtabayev AG (1981) T and B lymphocytes as related to adaption to high altitudes. Kosm Biol Aviat Kosm Med 15:87–89
Krupina TN, Korotaev MM, Pukhovy II (1974) Characteristics of the human immunologic state during hypoxic hypoxia. Kosm Biol Aviat Kosm Med 856–60
Krupina TN, Kotaev MM, Pukhovy II (1975) Comparative evaluation of studies of different levels of hypoxia on the human immunological status. Aviat Kosm Med 11:38–43
Lanier LL, Le AM, Civin CI, Loken MR, Phillips JH (1986) The relationship of CD16 (Leu-11) and Leu-19 (NKH-1) antigen expression on human peripheral blood NK-cells and cytotoxic T lymphocytes. J Immunol 136:4480–4486
Mackay CR (1991) T-cell memory: the connection between function, phenotype and migration pathways. Immunol Today 12:189–192
McCarthy DA, Dale MM (1988) The leucocytosis of exercise. Sports Med. 6:333–363
Meehan RT, Duncan U, Neale L (1986) Effect of hypoxia on human lymphocyte activation (abstract). Fed Proc 45:1031
Meehan RT (1987) Immune suppression at high altitude. Ann Emerg Med 16:974–979
Michie CA, McLean A, Alcock C, Beverley PCL (1992) Lifespan of human lymphocyte subsets defined by CD45 isoforms. Nature 360:264–265
Mirrakhimov MM, Kitayev MI (1978) Basic trends of imunologic studies of high altitude adaption. Zdravookhr Kirg 6:3–7
Nehlsen-Cannarella SL, Nieman DC, Jessen J, Chang L, Gusewitch G, Blix GG, Ashley E (1991) The effects of acute moderate exercise on lymphocyte function and serum immunoglobin levels. Int J Sports Med 12:391–398
Niemann DC, Berk LS, Simpson-Westerberg M, Arabatzis K, Youngberg S, Tan SA, Lee JW, Eby WC (1989) Effects of long-endurance running on immune system parameters and lymphocyte function in experienced marathoners. Int J Sports Med 10:317–323
Pedersen BK, Tvede N, Klarlund K, Christensen LD, Hansen FR, Galbo H, Kharazmi A, Halkjer-Kristensen J (1990) Indomethacin in vitro and in vivo abolishes post exercise suppression of natural killer cell activity in peripheral blood. Int J Sports Med 11:127–131
Prince HE, York J, Jensen ER (1992) Phenotypic comparison of the three populations of human lymphocytes defined by CD45RO and CD45RA expression. Cell Immunol 145:254–262
Shinkai S, Shore S, Shek PN, Shephard RJ (1992) Acute exercise and immune function: relationship between lymphocyte activity and changes in subset counts. Int J Sports Med 13:452–461
Stauber WT, Fritz VK, Vogelbach DW, Dahlmann B (1988) Characterization of muscle injured by forced lengthening. I. Cellular infiltrates. Med Sci Sports Exerc 20:345–353
Westermann J, Pabst R (1990) Lymphocyte subsets in the blood: a diagnostic window on the lamphoid system? Immunol Today 11:406–410
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Gabriel, H., Kullmer, T., Schwarz, L. et al. Circulating leucocyte subpopulations in sedentary subjects following graded maximal exercise with hypoxia. Eur J Appl Physiol 67, 348–353 (1993). https://doi.org/10.1007/BF00357634
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00357634