Summary
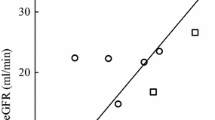
The disposition of cyclophosphamide and its alkylating metabolites was investigated in a group of myeloma patients with varying degrees of renal function impairment. No correlation between renal function and clearance of cyclophosphamide or its alkylating metabolites was found. No evidence of accumulation of cyclophosphamide or alkylating activity was found in four patients receiving radiolabelled cyclophosphamide. Renal function was found to be related to the reciprocal of the area under curve of alkylating activity, indicating that this area increased as renal function decreased. In view of the large nonrenal component of alkylating activity elimination and the large inter-subject variability, it is recommended that dose of cyclophosphamide is not altered in moderate impairment of renal function.
Similar content being viewed by others
References
Alberts, D. S., Van Daalen Wetter, T.: The effect of phenobarbital on cyclophosphamide antitumour activity. Cancer Res. 36, 2785–2789 (1976)
Bagley, C. M., Bostick, F. W., DeVita, V. T.: Clinical pharmacology of cyclophosphamide, Cancer Res. 33, 226–233 (1973)
Bischoff, K. B.: Pharmacokinetics and cancer chemotherapy. In: Pharmacology and pharmacokinetics. Teorell, T., Dedrick, R. L., Condliffe, P. G. (eds.). New York: Plenum 1974
Brock, N., Gross, R., Hohorst, H. J., Klein, O. H., Schneider, B.: Activation of cyclophosphamide in man and animals. Cancer 27, 1512–1529 (1971)
Cohen, J. L., Jos, J. Y., Jusko, W. J.: Pharmacokinetics of cyclophosphamide in man. Br. J. Pharmacol. 43, 677–680 (1971)
Colvin, M.: A review of the pharmacology and clinical use of cyclophsophamide. In: Clinical pharmacology of anti-neoplastic drugs, Pinedo, H. M. (ed.). Amsterdam: Elsevier 1978
Fenselau, C., Kan, M. N., Subbaras, S., Myles, A., Friedman, O. M., Colvin, M.: Identification of aldophosphamide as a metabolite of cyclophosphamide in vitro and in vivo in humans. Cancer Res. 2538–2543 (1977)
Foley, G. E., Friedman, O. M., Drolet, B. P.: Studies of the mechanism of action of cytoxan. Evidence of activation in vivo and vitro. Cancer Res. 21, 57–63 (1961)
Friedman, O. M., Boger, E.: Colorimetric estimation of nitrogen mustard in aqueous media. Anal. Chem. 33, 906–910 (1961)
Gibaldi, M., Perrier, D.: In: Pharmacokinetics. Gibaldi, M., Perrier, D. (eds.). New York: Marcel Dekker 1975
Jardine, I., Brundnett, R., Colvin, M., Fenselau, C.: Approach to the pharmacokinetics of cyclophosphamide. Quantitation of metabolites. Cancer Treat. Rep. 60, 403–408 (1976)
Jenne, S. W., Wyze, E., Rood, F. S., Macdonald, F. M.: Pharmacokinetics of theophylline. Application to adjustment of the clinical use of aminophylline. Clin. Pharmacol. Ther. 12, 349–360 (1972)
Metzler, C. M.: NONLIN: A computer programme for parameter estimation in nonlinear situations. Kalamazoo, Mich.: Upjohn Co. 1969
Mouridsen, H. T., Faber, O., Skovested, L.: The biotransformation of cyclophosphamide in man: Analysis of the variation in normal subjects. Acta Pharmacol. Toxicol. (Kbh.) 35, 98–106 (1974)
Mouridsen, H. T., Jacobsen, E.: Pharmacokinetics of cyclophosphamide in renal failure. Acta Pharamcol. Toxicol. (Kbh.) 36, 409–414 (1975)
Pantarotto, C., Bossi, A., Belvedere, G.: Quantitative GLC determination of cyclophosphamide and isophosphamide in biological specimens. J. Pharm. Sci. 63, 1554–1558 (1974)
Struck, R. F., Kirk, M. C., Witt, M. H., Laster, W. R.: Isolation and mass spectral identification of blood metabolites of cyclophosphamide: Evidence for phosphoramide mustard as the biologically active metabolite. Biomed. Mass Spectrom. 2, 46–52 (1975)
Wagner, T., Peter, G., Voelcker, G., Hohorst, H. J.: Characterization and quantitative estimation of activated cyclophosphamide in blood and urine. Cancer Res. 37, 2592–2596 (1977)
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Bramwell, V., Calvert, R.T., Edwards, G. et al. The disposition of cyclophosphamide in a group of myeloma patients. Cancer Chemother. Pharmacol. 3, 253–259 (1979). https://doi.org/10.1007/BF00254741
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00254741