Summary
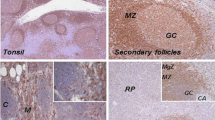
Expression of oncoprotein 18 (Op18), an intracellular phosphoprotein up-regulated in many malignant cell types, was evaluated in a series of normal lymphoid tissue and malignant lymphomas. In normal tonsils and reactive lymph nodes, the majority of Op18-positive cells were present in the germinal centres, whereas cells in the mantle zone were essentially negative and the interfollicular areas showed occasional positive cells. Double staining for PCNA and Op18 revealed that Op18 expression only to some extent was correlated with cell proliferation, as determined by PCNA expression.
Non-Hodgkin's lymphomas exhibited a variable Op18 expression, and in Hodgkin's disease, Reed-Sternberg and Hodgkin cells frequently expressed Op18 with a strong staining intensity. Using Op18-PCNA double staining in malignant lymphomas, Op18 expression could also be partially dissociated from cell proliferation. By using confocal microscopy, the intracellular localization of Op18 was studied, demonstrating diffuse reactivity in the cytoplasm in interphase cells and during mitosis, whereas nuclei and condensed chromosomes were negative. In conclusion, Op18 was expressed at variable levels in most, perhaps all, proliferating lymphocytes in benign lymphoid tissue as well as in malignant lymphomas. However, the Op18 protein was also detected in a significant fraction of apparently non-cycling normal and neoplastic lymphocytes.
Similar content being viewed by others
References
Brattsand, G., Roos, G., Marklund, U., Ueda, H., Landberg, G., Nånberg, E., Sideras, P. & Gullberg, M. (1993) Quantitative studies on the expression and regulation of an activation regulated phosphoprotein (Oncoprotein 18) in normal and neoplastic cells. Leukemia 7, 569–79.
Brattsand, G., Marklund, U., Nylander, K., Roos, G. & Gullberg, M. (1994) Cell cycle regulated phosphorylation of Oncoprotein 18 on serine 16, 25 and 38. Eur. J. Biochem. 220, 359–68.
Cooper, H. L., McDuffie, E. & Braverman, R. (1989) Human peripheral lymphocyte growth regulation and response to phorbol esters is linked to synthesis and phosporylation of the cytosolic protein, prosolin. J. Immunol. 143, 956–63.
Doye, V., Soubrier, F., Bauw, G., Boutterin, M. C., Beretta, L., Koppel, J., Vandekerckhove, J. & Sobel, A. (1989) A single cDNA encodes two isoforms of stathmin, a developmentally regulated neuron-enriched phosphoprotein. J. Biol. Chem. 264, 12134–7.
Doye, V., Kellermann, O., Buc-Caron, M. H. & Sobel, A. (1992) High expression of stathmin in multipotential teratocarcinoma and normal embryonic cells versus their early differentiated derivatives. Differentiation 50, 89–96.
Friedrich, B., Noreus, K., Cantrell, D. A. & Gullberg, M. (1988) Activation dependent phosphorylation of endogenous protein kinase C substrates in human T lymphocytes. Immunobiology 176, 465–78.
Friedrich, B., Cantrell, D. A. & Gullberg, M. (1989) Evidences for protein kinase C activation in T lymphocytes by stimulation of either the CD2 or CD3 antigens. Eur. J. Immunol. 19, 17–23.
Gerdes, J., VanBaarlen, J., Pileri, S., Schwarting, R., VanUnnik, J. A. M. & Stein, H. (1987) Tumor growth fraction in Hodgkin's disease. Am. J. Pathol. 128, 390.
Gullberg, M., Noreus, K., Brattsand, G., Friedrich, B. & Shingler, V. (1990) Purification and characterization of a 19 kilodalton intracellular protein: an activation regulated putative protein kinase C substrate of T lymphocytes. J. Biol. Chem. 265, 17 499–505.
Hailat, N., Strahler, J. R., Melham, R. F., Zhu, X. X., Brodeur, G., Seeger, R. C., Reynolds, C. P. & Hanash, S. M. (1990) N-myc gene amplification in neuroblastoma is associated with altered phosphorylation of a proliferation related polypeptide (Op18). Oncogene 5, 1615–8.
Hanash, S. M., Strahler, J. R., Kuick, R., Chu, E. H. Y. & Nichols, D. (1988) Identification of a polypeptide associated with the malignant phenotype in acute leukemia. J. Biol. Chem. 263, 12 813–5.
McCormick, D. & Hall, P. A. (1992) The complexities of proliferating cell nuclear antigen. Histopathology 21, 591–4.
Marklund, U., Brattsand, G., Shingler, V. & Gullberg, M. (1993a) Serine 25 of Oncoprotein 18 is a major cytosolic target for the mitogen-activated protein kinase. J. Biol. Chem. 268, 15039–49.
Marklund, U., Brattsand, G., Osterman, Ö, Ohlsson, P. I. & Gullberg, M. (1993b) Multiple signal transduction pathways induce phosphorylation of serine 16, 25 and 38 of Oncoprotein 18 in T-lymphocytes. J. Biol. Chem. 268, 25 671–80.
Melham, R. F., Zhu, X. X., Hailat, R., Strahler, J. R. & Hanash, S. M. (1991a) Characterization of the gene for a proliferationrelated phosphoprotein (Oncoprotein 18) expressed in high amounts in acute leukemia. J. Biol. Chem. 266, 17 747–53.
Melham, R. F., Strahler, J. R., Hailat, R., Zhu, X. X. & Hanash, S. M. (1991b) Involvement of Op18 in cell proliferation. Biochem. Biophys. Res. Commun. 179, 1649–55.
Roos, G., Brattsand, G., Landberg, G., Marklund, U. & Gullberg, M. (1993) Expression of Oncoprotein 18 in human leukemias and lymphomas. Leukemia 7, 1538–46.
Shubart, U. K., Das Banerjee, M. & Eng, J. (1989) Homology between the cDNAs encoding phosphoprotein p19 and SCG10 reveals a novel mammalian gene family preferentially expressed in developing brain. DNA 8, 389–98.
Zhu, X. X., Kosarsky, K., Strahler, J. R., Eckerskorn, C., Lottspeich, F., Melham, R., Lowe, J., Fox, D. A., Hanash, S. M. & Atweh, G. F. (1989) Molecular cloning of a novel leukemia associated gene: evidence of conservation in animal species. J. Biol. Chem. 264, 14 556–60.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Nylander, K., Marklund, U., Brattsand, G. et al. Immunohistochemical detection of oncoprotein 18 (Op18) in malignant lymphomas. Histochem J 27, 155–160 (1995). https://doi.org/10.1007/BF00243911
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00243911