Summary
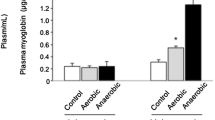
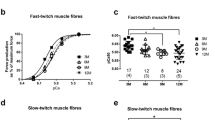
Intramuscular collagen in a slow (m. soleus) and a fast (m. rectus femoris) skeletal muscle was studied by biochemical, morphometric, and immunohistochemical methods. Wistar white rats of 1, 4, 10, and 24 months were used as experimental animals. Our aim was to evaluate the effects of life-long physical training (treadmill running, 5 days a week for 1, 3, 9, and 23 months depending on the age attained). The biochemical concentration of collagen was higher in m. soleus than in m. rectus femoris and it increased in youth and in old age in m. soleus. The trained rats had higher concentrations of collagen than the untrained rats at 10 and 24 months. The morphometrically measured area-fractions of both the endomysium and perimysium were higher in m. soleus than in m. rectus femoris. The age-related increase in intramuscular connective tissue was of endomysial origin. The immunohistochemical staining of type-I, -III, and -IV collagens indicated the more collagenous nature of m. soleus as compared with m. rectus femoris for all major collagen types; this was most marked for type-IV collagen of basement membrane. The results indicate that both age and endurance-type physical training further distinguish the slow and fast muscles with respect to their connective tissue.
Similar content being viewed by others
References
Alnaqeeb MA, Zaid NS, Goldspink G (1984) Connective tissue changes and physical properties of developing and ageing skeletal muscle. J Anat 139:677–689
Bailey AJ, Shellswell GB, Duance VC (1979) Identification and change of collagen types in differentiating myoblasts and developing chick muscle. Nature 278:67–69
Booth FW, Gould EW (1975) Effects of training and disuse on connective tissue. In: Willmore JH, Keogh JF (eds) Exercise and Sport Sciences reviews. Academic Press, New York, pp 83–112
Bornstein P, Sage H (198) Structurally different collagen types. Ann Rev Biochem 49:957–1003
Campbell MJ, McComas AJ, Petito F (1973) Physiological changes in aging muscles. J Neurol, Neurosurg Psych 36:174–182
Duance VC, Rostall DJ, Beard H, Bourne FJ, Bailey AJ (1977) The location of three collagen types in skeletal muscle. FEBS Lett 79:248–252
Duance VC, Black CM, Dubowitz V, Hughes GRV, Bailey AJ (1980a) Polymyositis — an immunofluorescence study on the distribution of collagen types. Muscle Nerve 3:487–490
Duance VC, Stephens HR, Dunn M, Bailey AJ, Dubowitz V (1980b) A role for collagen in the pathogenesis of muscular dystrophy. Nature 284:470–472
Dubowitz V, Brooke MH (eds) (1973) Muscle Biopsy: A Modern Approach. WB Saunders, London
Eyre DR (1980) Collagen molecular diversity in the body's protein scaffold. Science 27:1315–1322
Foidart M, Foidart J-M, Engel KW (1981) Collagen localization in normal and fibrotic human skeletal muscle. Arch Neurol 38:152–157
Gay S, Balleisen L, Remberger K, Fietzek PP, Adelmann BC, Kühn K (1975) Immunohistochemical evidence for the presence of collagen type III in human arterial walls, arterial thrombi, and in leukocytes, incubated with collagen in vitro. Klin Wschr 53:899–902
Holloszy JO, Booth FW (1976) Biochemical adaptations to endurance exercise in muscle. Ann Rev Physiol 38:273–291
Kivirikko KI, Laitinen O, Prockop DJ (1967) Modification of a specific assay for hydroxyproline in urine. Anal Biochem 19:249–255
Kovanen V, Suominen H, Heikkinen E (1980) Connective tissue of “fast” and “slow” skeletal muscle in rats — effects of endurance training. Acta Physiol Scand 108:173–180
Kovanen V, Suominen H, Heikkinen E (1984a) Collagen of slow twitch and fast twitch muscle fibres in different types of rat skeletal muscle. Eur J Appl Physiol 52:235–242
Kovanen V, Suominen H, Heikkinen E (1984b) Mechanical properties of fast and slow skeletal muscle with special reference to collagen and endurance training. J Biomech 17:725–735
Laurent GJ, Sparrow P, Bates PC, Millward DJ (1978) Collagen content and turnover in cardiac and skeletal muscles of the adult fowl and the changes during stretch-induced growth. Biochem J 176:419–427
Lawrie RA (1979) Meat Science. Pergamon Press, Oxford New York Toronto Sidney Paris Frankfurt
Light N, Champion AE (1984) Characterization of muscle epimysium, perimysium and endomysium collagens. Biochem J 219:1017–1026
McLennan WR, Hall MRP, Timothy JI, Robinson M (1980) Is weakness in old age due to muscle wasting? Age Ageing 9:188–192
Minor RR (1980) Collagen metabolism. Am J Pathol 98:226–278
Mohan S, Radha E (1980) Age-related changes in rat muscle collagen. Gerontology 26:61–67
Orkin RW, Gehron P, McGoodwin EB, Martin GR, Valentine T, Swarm R (1977) A murine tumor producing a matrix of basement membrane. J Exp Med 145:204–220
Padykula HA, Herman E (1955) The specificity of the histochemical method for adenosine triphosphatase. J. Histochem Cytochem 3:170–195
Pras M, Glynn LE (1973) Isolation of a non-collagenous reticulin component and its primary characterization. Br J Exp Pathol 54:449–456
Puchtler H, Waldrop FS (1978) Silver impregnation methods for reticulum fibers and reticulin. A re-investigation of their origins and specificity. Histochemistry 57:177–187
Rojkind M, Gianbrone M-A, Biempica L (1979) Collagen types in normal and cirrhotic liver. Gastroenterology 76:710–719
Romeis B (1968) Mikroskopische Technik. Oldenburg Verlag, München
Suominen H, Kovanen V (1984) Effects of age and physical training on fibre composition of fast and slow skeletal muscle in rats. Olympic Scientific Congress “Sport, Health & Well-being”, Eugene, pp 140–141
Timpl R, Wich G, Gay S (1977) Antibodies to distinct types of collagens and procollagens and their application in immunohistology. J Immunol Methods 18:165–182
Weibel ER (1979) Stereological methods. Vol 1. Practical methods for biological morphology. Academic Press, London
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kovanen, V., Suominen, H. & Peltonen, L. Effects of aging and life-long physical training on collagen in slow and fast skeletal muscle in rats. Cell Tissue Res. 248, 247–255 (1987). https://doi.org/10.1007/BF00218191
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00218191