Core Messages
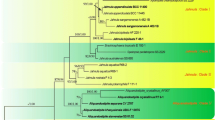
This chapter presents and discusses all techniques and media used to isolate, maintain, preserve, and identify the 13 species that are presently included in the genus. Each species is described morphologically, including features of the colonies and microscopic characteristics of the yeast cells, either with or without filaments; physiologically, including the growth at 37 and 40°C, three enzymatic activities, namely catalase, β-glucosidase and urease, and growth with 5 individual lipid supplements, namely Tween 20, 40, 60 and 80, and Cremophor EL. Their ecological preferences and role in human and veterinary pathology are also discussed. For quite a long time, the genus was known to be related to the Basidiomycota, despite the absence of a sexual state. The phylogeny, based on sequencing of the D1/D2 variable domains of the ribosomal DNA and the ITS regions, as presented in the chapter, confirmed the basidiomycetous nature of these yeasts, which occupy an isolated position among the Ustilaginomycetes. The relationship to the Basidiomycetes is also supported by monopolar and percurrent budding and the multilamellar cell wall ultrastructure. Some characteristics of this cell wall, which is unparalleled in the world of fungi, together with the lipophily demonstrate the uniqueness of this genus in the fungal kingdom.
This chapter presents and discusses all techniques and media used to isolate, maintain, preserve, and identify the 13 species that are presently included in the genus. Each species is described morphologically, including features of the colonies and microscopic characteristics of the yeast cells, either with or without filaments; physiologically, including the growth at 37 and 40°C, three enzymatic activities, namely catalase, β-glucosidase and urease, and growth with 5 individual lipid supplements, namely Tween 20, 40, 60 and 80, and Cremophor EL. Their ecological preferences and role in human and veterinary pathology are also discussed. For quite a long time, the genus was known to be related to the Basidiomycota, despite the absence of a sexual state. The phylogeny, based on sequencing of the D1/D2 variable domains of the ribosomal DNA and the ITS regions, as presented in the chapter, confirmed the basidiomycetous nature of these yeasts, which occupy an isolated position among the Ustilaginomycetes. The relationship to the Basidiomycetes is also supported by monopolar and percurrent budding and the multilamellar cell wall ultrastructure. Some characteristics of this cell wall, which is unparalleled in the world of fungi, together with the lipophily demonstrate the uniqueness of this genus in the fungal kingdom.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Baillon H (1889) Traité de botanique médicale cryptogamique, 4th edn. Octave Doin, Paris
Sabouraud R (1904) Les maladies du cuir chevelu. II. Les maladies desquamatives. Masson et Cie, Paris
Slooff WC (1970) Genus 6 Pityrosporum Sabouraud. In: Lodder J (ed) The yeasts, a taxonomic study, 2nd edn. North-Holland, Amsterdam, pp 1167–1186
Acton HW, Panja G (1927) Seborrhoeic dermatitis or pityriasis capitis: a lesion caused by the Malassezia ovale. Indian Med Gaz 62:603–614
Panja G (1927) The Malassezia of the skin, their cultivation, morphology and species. Trans Th Cong Far Easthern Ass Trop Med 2:442–446
Shifrine M, Marr AG (1963) The requirement of fatty acids by Pityrosporum ovale. J Gen Microbiol 32:263–270
Van Abbe NJ (1964) The investigation of dandruff. J Soc Cosmetic Chem 52:173–188
Midgley G (1989) The diversity of Pityrosporum (Malassezia) yeasts in vivo and in vitro. Mycopathologia 106:143–153
Leeming JP, Notman FH (1987) Improved methods for isolation and enumeration of Malassezia furfur from human skin. J Clin Microbiol 25:2017–2019
Kaneko T, Makimura K, Onozaki M et al (2005) Vital growth factors of Malassezia species on modified CHROMagar Candida. Med Mycol 43:699–704
Lorenzini R, de Bernadis F (1987) Studies on the isolation, growth and maintenance of Malassezia pachydermatis. Mycopathologia 99:129–131
Renker C, Alphei J, Buscot F (2003) Soil nematodes associated with the mammal pathogenic fungal genus Malassezia (Basidiomycota: Ustilaginomycetes) in Central European forests. Biol Fertil Soils 37:70–72
Bjelland T, Ekman S (2005) Fungal diversity in rock beneath a crustose lichen as revealed by molecular markers. Microbiol Ecol 49:598–603
Fell JW, Scorzetti G, Connell L et al (2006) Biodiversity of micro-eukaryotes in Antarctic Dry Valley soils with <5% soil moisture. Soil Biol Biochem 38:3107–3115
Lai X, Cao L, Tan H et al (2007) Fungal communities from methane hydrate-bearing deep-sea marine sediments in South China Sea. ISME J 1:756–762
Bond R, Anthony RM (1995) Characterization of markedly lipid-dependent Malassezia pachydermatis isolates from dogs. J Appl Microbiol 78:537–542
Guého E, Batra R, Boekhout T (2010) The genus Malassezia Baillon. In: Kurtzman CP, Fell JW, Boekhout T (eds) The yeasts, a taxonomic study, 5th edn. Elsevier, Amsterdam (in press)
Cabañes FJ, Theelen B, Castellá G et al (2007) Two new lipid-dependent Malassezia species from domestic animals. FEMS Yeast Res 7:1064–1076
Crespo MJ, Abarca ML, Cabañes FJ (2000) Evaluation of different preservation and storage methods for Malassezia spp. J Clin Microbiol 38:3872–3875
Breierová E, Kocková-Kratochilová A, Šajbidor J et al (1991) Malassezia pachydermatis: properties and storage. Mycoses 34:349–352
Guillot J, Guého E (1995) The diversity of Malassezia yeasts confirmed by rRNA sequence and nuclear DNA comparisons. Antonie van Leeuwenhoek 67:297–314
Crespo Erchiga V, Guého E (2005) Superficial diseases caused by Malassezia species. In: Merz WG, Hay RJ (eds) Topley and Wilson’s microbiology and microbial infections, medical mycology, 10th edn, vol 5. Hodder Arnold, London, pp 202–219
Guého E, Midgley G, Guillot J (1996) The genus Malassezia with description of four new species. Antonie van Leeuwenhoek 69:337–355
Guého E, Boekhout T, Ashbee HR et al (1998) The role of Malassezia species in the ecology of human skin and as pathogen. Med Mycol 36(suppl 1):220–229
Guillot J, Guého E, Lesourd M et al (1996) Identification of Malassezia species. A practical approach. J Mycol Méd 6:103–110
Mayser P, Haze P, Papavassilis C et al (1997) Differentiation of Malassezia species: selectivity of Cremophor EL, castor oil and ricinoleic acid for M. furfur. Br J Dermatol 137:208–213
Hägler AN, Ahearn DG (1981) Rapid diazonium blue B test to detect basidiomycetous yeasts. Int J Syst Bacteriol 31:204–208
Kurtzman CP (2010) Methods for the isolation, maintenance and identification of yeasts. In: Kurtzman CP, Fell JW, Boekhout T (eds) The yeasts, a taxonomic study, 5th edn. Elsevier, Amsterdam, pp (in press)
Kaneko T, Makimura K, Sugita T et al (2006) Tween 40-based precipitate production observed on chromogenic agar and development of biological identification kit for Malassezia species. Med Mycol 44:227–231
Kaneko T, Makimura K, Abe M et al (2007) Revised culture-based system for identification of Malassezia species. J Clin Microbiol 45:3737–3742
Boekhout T, Guého E (2003) Basidiomycetous yeasts. In: Howard DH (ed) Pathogenic fungi in humans and animals, 2nd edn. Marcel Dekker, New York, pp 535–564
Kurtzman CP, Fell JW, Boekhout T (2010) The Yeasts, a taxonomic study, 5th edn. Elsevier, Amsterdam
Hamamoto M, Uchida K, Yamaguchi H (1992) Ubiquinone system and DNA base composition of strains in the genus Malassezia determined by high-performance liquid chromatography. J Gen Appl Microbiol 38:79–82
Dodge CW (1935) Medical mycology. fungous diseases of men and other mammals. Mosby, St. Louis
Weidman FD (1925) Exfoliative dermatitis in the Indian rhinoceros (Rhinoceros unicornis), with description of a new species: Pityrosporum pachydermatis. In: Fox H (ed). Rep Lab Museum Comp Pathol Zoo Soc Philadelphia, Philadelphia, pp 36–43
Gustafson BA (1955) Otitis externa in the dog. A bacteriological and experimental study, Thesis, Stockholm
Guillot J, Guého E, Chermette R (1995) Confirmation of the nomenclatural status of Malassezia pachydermatis. Antonie van Leeuwenhoeck 67:173–176
Guillot J, Guého E, Chévrier G et al (1997) Epidemiological analysis of Malassezia pachydermatis isolates by partial sequencing of the large subunit ribosomal RNA. Res Vet Sci 6:22–25
Midreuil F, Guillot J, Guého E et al (1999) Genetic diversity in the yeast species Malassezia pachydermatis analysed by multilocus enzyme electrophoresis. Int J Syst Bacteriol 49:1287–1294
Guého E, Guillot J (1999) Comments on Malassezia species from dogs and cats. Mycoses 42:673–674
Ahearn DG, Simons RB (1998) Malassezia Baillon. In: Kurtzman CP, Fell JW (eds) The yeasts, a taxonomic study, 4th edn. Elsevier, Amsterdam, pp 782–784
Guillot J, Bond R (1999) Malassezia pachydermatis: a review. Med Mycol 37:295–306
Guillot J, Guého E, Mialot M et al (1998) Importance des levures du genre Malassezia en dermatologie vétérinaire. Point Vét 29:21–31
Guillot J, Petit T, Degorce-Rubiales F et al (1998) Dermatitis caused by Malassezia pachydermatis in a California sea lion (Zalophus californianus). Vet Rec 142:311–312
Bauwens L, De Vroey Ch, De Meurichy W (1996) A case of exfoliative dermatitis in a captive southern white rhinoceros (Ceratotherium simum simum). JK Zoo Wildlife Med 27:271–274
Wesche P, Bond R (2003) Isolation of Malassezia pachydermatis from the skin of captive rhinoceroses. Vet Rec 153:404–405
Chang HJ, Miller HL, Watkins N et al (1998) An epidemic of Malassezia pachydermatis in an intensive care nursery associated with colonization of health care workers’ pet dogs. New Engl J Med 338:706–711
Midgley G (2000) The lipohilic yeasts: state of the art and prospects. Med. Mycol 38(suppl 1): 9–16
Morris DO, O’Shea K, Shofer FS et al (2005) Malassezia pachydermatis carriage in dog owners. Emerg Infect Dis 11:83–88
Castellani A, Chalmers AJ (1913) Manual of tropical medicine, 3rd edn. Baillière, Tindal and Cox, London, p 1747
González A, Sierra R, Cárdenas ME et al (2009) Physiological and molecular characterization of atypical isolates of Malassezia furfur. J Clin Microbiol 47:48–53
Duarte ER, Lachance M-A, Hamdan JS (2002) Identification of atypical strains of Malassezia spp. from cattle and dog. Can J Microbiol 48:749–752
Murai T, Nakamura Y, Kano R et al (2002) Differentiation of Malassezia furfur and Malassezia sympodialis by glycine utilization. Mycoses 45:180–183
Salkin IF, Gordon MA (1977) Polymorphism of Malassezia furfur. Can J Microbiol 23:471–475
Yarrow D, Ahearn D (1984) Genus 7 Malassezia Baillon. In: Kreger-van Rij NJW (ed) The yeasts, a taxonomic study, 3rd edn. Elsevier, Amsterdam, pp 882–885
Tanaka R, Nishimura K, Kamei K et al (2001) Assimilation test of Malassezia furfur isolated from the environment. Nippon Ishinkin Gakkai Zasshi 42:123–126
Colombo S, Nardoni S, Cornegliani L et al (2007) Prevalence of Malassezia spp. yeasts in feline nail folds: a cytological and mycological study. Vet Dermatol 18:278–283
Crespo MJ, Abarca ML, Cabañes FJ (1999) Isolation of Malassezia furfur from a cat. J Clin Microbiol 37:1573–1574
Crespo MJ, Abarca ML, Cabañes FJ (2000) Atypical lipid-dependent Malassezia species isolated from dogs with otitis externa. J Clin Microbiol 38:2383–2385
Crespo MJ, Abarca ML, Cabañes FJ (2002) Occurrence of Malassezia spp. in horses and domestic ruminants. Mycoses 45:33–337
Duarte ER, Melo MM, Hahn RC et al (1999) Prevalence of Malassezia spp. in the ears of asymptomatic cattle and cattle with otitis in Brazil. Med Mycol 37:159–162
Duarte ER, Resende CP, Rosa CA et al (2001) Prevalence of yeasts and mycelial fungi in bovine paratitic otitis in the State of Minas Gerals, Brazil. J Vet Med B Infect Dis Public Health 48:631–635
Gandra RF, Gambale W, de Cássia Garcia Simão R et al (2008) Malassezia spp. in acoustic meatus of bats (Molossus molossus) of the Amazon region, Brazil. Mycopathologia 165:21–26
Sugita T, Tajima M, Takashima M et al (2004) A new yeast, Malassezia yamatoensis, isolated from a patient with seborrheic dermatitis, and its distribution in patients and healthy subjects. Microbiol Immunol 48:579–583
Simmons RB, Guého E (1990) A new species of Malassezia. Mycol Res 94:1146–1149
Cunningham AC, Leeming JP, Ingham E et al (1990) Differentiation of three serovars of Malassezia furfur. J Appl Bacteriol 68:439–446
Cabañes FJ, Hernández JJ, Castellá G (2005) Molecular analysis of Malassezia sympodialis-related strains from domestic animals. J Clin Microbiol 43:277–283
Saadatzadeh MR, Ashbee HR, Holland KT et al (2001) Production of the mycelial phase of Malassezia in vitro. Med Mycol 39:487–493
Crespo Erchiga V, Ojeda Martos A, Vera Casaño A (1999) Mycology of pityriasis versicolor. J Mycol Méd 9:143–148
Gupta AK, Batra R, Bluhm R et al (2004) Skin diseases associated with Malassezia species. J Am Acad Dermatol 51:785–798
Bond R, Anthony RM, Dodd M et al (1996) Isolation of Malassezia sympodialis from feline skin. J Med Vet Mycol 34:145–147
Bond R, Howell SA, Haywood PJ et al (1997) Isolation of Malassezia sympodialis and Malassezia globosa from healthy pet cats. Vet Rec 141:200–201
Crespo MJ, Abarca ML, Cabañes FJ (2000) Otitis externa associated with Malassezia sympodialis in two cats. J Clin Microbiol 38:1263–1266
Sugita T, Takashima M, Shinoda T et al (2002) New yeast species, Malassezia dermatis, isolated from patients with atopic dermatitis. J Clin Microbiol 40:1363–1367
Lee YW, Kim SM, Oh BH et al (2008) Isolation of 19 strains of Malassezia dermatis from healthy human skin in Korea. J Dermatol 35:772–777
Ähman S, Perrins N, Bond R (2007) Carriage of Malassezia spp. yeasts in healthy and seborrhoeic Devon Rex cats. Med Mycol 45:449–455
Bond R, Stevens K, Perrins N et al (2008) Carriage of Malassezia spp. yeasts in Cornish Rex, Devon Rex and domestic short hair cats: a cross selectional survey. Vet Dermatol 198:299–304
Uzal FA, Paulson D, Eigenheer AL et al (2007) Malassezia slooffiae-associated dermatitis in a goat. Vet Dermatol 18:348–352
Sugita T, Takashima M, Kodama M et al (2003) Description of a new species, Malassezia japonica, and its detection in patients with atopic dermatitis and healthy subjects. J Clin Microbiol 41:4695–4699
Hirai A, Kano R, Makimura K et al (2004) Malassezia nana sp. nov., a novel lipid-dependent yeast species isolated from animals. Int J Syst Evol Microbiol 54:623–627
Perrins N, Gaudinio F, Bond R (2007) Carriage of Malassezia spp. yeasts in cats with diabetes mellitus, hyperthyroidism and neoplasia. Med Mycol 45:541–546
Nell A, James SA, Bond CJ et al (2002) Identification and distribution of a novel Malassezia species yeast on normal equine skin. Veter Rec 150:395–398
Gordon MA (1951) The lipophilic mycoflora of the skin. I. In vitro culture of Pityrosporum orbiculare sp. Mycologia 43:524–535
Sugita T, Kodama M, Saito M et al (2003) Sequence diversity of the intergenic spacer region of the rRNA gene of Malassezia globosa colonizing the skin of patients with atopic dermatitis and healthy individuals. J Clin Microbiol 41:3022–3027
Aspiroz C, Moreno LA, Rezusta A et al (1999) Differentiation of three biotypes of Malassezia species on normal human skin. Correspondence with M. globosa, M. sympodialis and M. restricta. Mycopathologia 145:69–74
Sugita T, Tajima M, Amaya M et al (2004) Genotype analysis of Malassezia restricta as the major cutaneous flora in patients with atopic dermatitis and healthy subjects. Microbiol Immunol 48:755–759
Pitkäranta M, Meklin T, Hyvärinen A et al (2008) Analysis of fungal flora in indoor dust by ribosomal DNA sequence analysis, quantitative PCR, and culture. Appl Environ Microbiol 74:233–244
Zhang N, Suh SO, Blackwell M (2003) Microorganisms in the gut of beetles: evidence from molecular cloning. J Invertebr Pathol 84:226–233
Moore RT (1980) Taxonomic proposals for the classification of marine yeasts and other yeasts-like fungi including the smuts. Bot Mar 23:361–373
Guého E, Kurzman CP, Peterson SW (1989) Evolutionary affinities of heterobasidiomycetous yeasts estimated from 18 S and 25 S ribosomal RNA sequence divergence. Syst Appl Microbiol 12:230–236
Begerow D, Bauer R, Boekhout T (2000) Phylogenetic placements of ustilaginomycetous anamorphs as deduced from LSU rDNA sequence. Mycol Res 104:53–60
Fell JW, Boekhout T, Fonseca A et al (2000) Biodiversity and systematics of basidiomycetous yeasts as determined by large-subunit rDNA D1/D2 domain sequence analysis. Int J Syst Evol Biol 50:1351–1371
Begerow D, Stoll M, Bauer R (2006) A phylogenetic hypothesis of Ustilaginomycotina based on multiple gene analyses and morphological data. Mycologia 98:906–916
Matheny PB, Wang Z, Binder M et al (2006) Contributions of rpb2 and tef1 to the phylogeny of mushrooms and allies (Basidiomycota, Fungi). Mol Phyl Evol 43:430–451
Hibbett DS, Binder M, Bischoff JF et al (2007) A higher-level phylogenetic classification of the Fungi. Mycol Res 111:509–547
Mittag H (1995) Fine structural investigation of Malassezia furfur. II. The envelope of the yeast cells. Mycoses 38:13–21
Simmons RB, Ahearn DG (1987) Cell wall ultrastructure and diazonim blue B reaction of Sporopachydermia quercuum, Bullera tsugae, and Malassezia spp. Mycologia 79:38–43
Guillot J, Guého E, Prévost MC (1995) Ultrastructural features of the dimorphic yeast Malassezia furfur. J Mycol Méd 5:86–91
Winiarczyk S (1992) The ultrastructure of Pityrosporum pachydermatis. Arch Vet Polonicum 32:5–13
Abou-Gabal M, Fagerland JA (1979) Electron microscopy of Pityrosporum canis “pachydermatis”. Mykosen 22:85–90
Kreger-van Rij NJ, Veenhuis M (1970) An electron microscope study of the yeast Pityrosporum ovale. Arch Mikrobiol 71:123–131
Marek D, Miroslav G, Kopecká M (2007) Microtubular and actin cytoskeletons and ultrastructural characteristics of the potentially pathogenic basidiomycetous yeast Malassezia pachydermatis. Cell Biol Inter 31:16–23
Mittag H (1994) An approach of the fine structure of Malassezia furfur. XII Congress of the International Society for Human and Animals Mycology, March 1994, Abst D139, Adelaide, Australie
Nishimura K, Asada Y, Tanaka S et al (1991) Ultrastructure of budding process of Malassezia pachydermatis. J Med Vet Mycol 29:387–393
Keddie FM (1966) Electron microscopy of Malassezia furfur in tinea versicolor. Sabouraudia 5:134–137
Takeo K, Nakai E (1980) Tilting method for distinguishing the side of freeze-fracture replicas and for describing mirror-image asymmetry. J Electron Microsc 29:91–97
Takeo K, Nakai E (1986) Mode of cell growth of Malassezia (Pityrosporum) as revealed by using plasma membrane configurations as natural markers. Can J Microbiol 32:389–394
Matakieff E (1899) Le pityriasis versicolor et son parasite. Thèse Fac Méd Univ Nancy
Breathnach AS, Gross M, Martin B (1976) Freeze-fracture replication of cultured Pityrosporum orbiculare. Sabouraudia 14:105–113
Cole GT, Samson RA (1979) Patterns of development in conidial fungi. Pitman, London, p 70
Mittag H (1995) Fine structural investigation of Malassezia furfur: I. Size and shape of the yeast cells and a consideration of their ploidy. Mycoses 37:393–399
David M, Gabriel M, Kopecká M (2007) Microtubular and actin cytoskeletons and ultrastructural characteristics of the potentially pathogenic basidiomycetous yeast Malassezia pachydermatis. Cell Biol Intern 31:16–23
Simmons RB (1989) Comparison of chitin localization in Saccharomyces cerevisiae, Crytococcus neoformans, and Malassezia spp. Mycol Res 94:551–553
Acknowledgements
The authors would like to warmly thank Anne-Françoise Miegeville (Laboratoire de Bactériologie-Antibiologie, Faculté de Médecine, Nantes, France), who made many scanning electron micrographs (SEM), namely all the pictures on filters presented in this chapter, and also Bart Theelen (Centraalbureau for Schimmelcultures, Utrecht, The Netherlands) for his help in preparing one of the phylogenetic trees.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2010 Springer-Verlag Berlin Heidelberg
About this chapter
Cite this chapter
Guého-Kellermann, E., Boekhout, T., Begerow, D. (2010). Biodiversity, Phylogeny and Ultrastructure. In: Boekhout, T., Mayser, P., Guého-Kellermann, E., Velegraki, A. (eds) Malassezia and the Skin. Springer, Berlin, Heidelberg. https://doi.org/10.1007/978-3-642-03616-3_2
Download citation
DOI: https://doi.org/10.1007/978-3-642-03616-3_2
Publisher Name: Springer, Berlin, Heidelberg
Print ISBN: 978-3-642-03615-6
Online ISBN: 978-3-642-03616-3
eBook Packages: MedicineMedicine (R0)