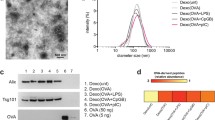
Abstract
Initial studies in our laboratory were focused on the use of dendritic cells (DC) genetically modified to express Th2-derived cytokines (i.e., interleukin [IL]-4 and IL-10) or apoptotic proteins (i.e., Fas Ligand [FasL]) to reduce inflammation in a mouse model of experimentally induced arthritis. Exosomes are nano-sized vesicles (40–100 nm diameter) released by different cell types, including DC, that contain many of the proteins thought to be involved in regulating the immune response. We have demonstrated that exosomes derived from immature DC treated with immunomodulatory cytokines (i.e., IL-10, IL-4) are able to inhibit inflammation in a murine footpad model of delayed-type hypersensitivity (DTH) and reduce the severity of established collagen-induced arthritis (CIA). In fact, the exosomes were as therapeutic as the parental DC. Because purified DC-derived exosomes are very stable vesicles, they may be a better approach for future treatment of arthritis and other autoimmune disorders than the more unstable DC. In this chapter we detail a protocol for preparing the exosomes produced by murine bone marrow-derived DC. We also review methods to assess the purity and concentration of purified exosomes, by using electron microscopy, Western blot analysis, and flow cytometry. Finally, we describe methods to assess the function of exosomes in vitro, using the mixed leukocytes reaction, and in vivo by means of DTH and an experimental model of CIA.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Pan, B. T. and Johnstone, R. M. (1983) Fate of the transferrin receptor during maturation of sheep reticulocytes in vitro: selective externalization of the receptor. Cell 33, 967–978.
Denzer, K., Kleijmeer, M. J., Heijnen, H. R, Stoorvogel, W., and Geuze, H. J. (2000) Exosome: from internal vesicle of the multivesicular body to intercellular signaling device. J. Cell Sci. 113, 3365–3374.
Thery, C., Zitvogel, L., and Amigorena, S. (2002) Exosomes: composition, biogenesis and function. Nat. Rev. Immunol. 2, 569–579.
Johnstone, R. M., Adam, M., Hammond, J. R., Orr, L., and Turbide, C. (1987) Vesicle formation during reticulocyte maturation. Association of plasma membrane activities with released vesicles (exosomes). J. Biol. Chem. 262, 9412–9420.
Zitvogel, L., Regnault, A., Lozier, A., et al. (1998) Eradication of established murine tumors using a novel cell-free vaccine: dendritic cell-derived exosomes. Nat. Med. 4, 594–600.
Chaput, N., Schartz, N. E., Andre, F., et al. (2004) Exosomes as potent cell-free peptide-based vaccine. II. Exosomes in CpG adjuvants efficiently prime naive Tc1 lymphocytes leading to tumor rejection. J. Immunol. 172, 2137–2146.
Morse, M. A., Garst, J., Osada, T., et al. (2005) A phase I study of exosome immunotherapy in patients with advanced non-small cell lung cancer. J. Transl. Med. 3, 9.
Escudier, B., Dorval, T., Chaput, N., et al. (2005) Vaccination of metastatic melanoma patients with autologous dendritic cell (DC) derived-exosomes: results of the first phase I clinical trial. J. Transl. Med. 3, 10.
Altieri, S. L., Khan, A. N., and Tomasi, T. B. (2004) Exosomes from plasmacytoma cells as a tumor vaccine. J. Immunother. 27, 282–288.
Aline, F., Bout, D., Amigorena, S., Roingeard, P., and Dimier-Poisson, I. (2004) Toxoplasma gondii antigen-pulsed-dendritic cell-derived exosomes induce a protective immune response against T. gondii infection. Infect. Immun. 72, 4127–4137.
Karlsson, M., Lundin, S., Dahlgren, U., Kahu, H., Pettersson, I., and Telemo, E. (2001) “Tolerosomes” are produced by intestinal epithelial cells. Eur. J. Immunol. 31, 2892–2900.
Peche, H., Heslan, M., Usal, C., Amigorena, S., and Cuturi, M. C. (2003) Presentation of donor major histocompatibility complex antigens by bone marrow dendritic cell-derived exosomes modulates allograft rejection. Transplantation 76, 1503–1510.
Andreola, G., Rivoltini, L., Castelli, C., et al. (2002) Induction of lymphocyte apoptosis by tumor cell secretion of FasL-bearing microvesicles. J. Exp. Med. 195, 1303–1316.
Martinez-Lorenzo, M. J., Anel, A., Gamen, S., et al. (1999) Activated human T cells release bioactive Fas ligand and APO2 ligand in microvesicles. J. Immunol. 163, 1274–1281.
Abrahams, V. M., Straszewski, S. L., Kamsteeg, M., et al. (2003) Epithelial ovarian cancer cells secrete functional Fas ligand. Cancer Res. 63, 5573–5581.
Abusamra, A. J., Zhong, Z., Zheng, X., et al. (2005) Tumor exosomes expressing Fas ligand mediate CD8+ T-cell apoptosis. Blood Cells Mol. Dis. 35, 169–173.
Frangsmyr, L., Baranov, V., Nagaeva, O., Stendahl, U., Kjellberg, L., and Mincheva-Nilsson, L. (2005) Cytoplasmic microvesicular form of Fas ligand in human early placenta: switching the tissue immune privilege hypothesis from cellular to vesicular level. Mol. Hum. Reprod. 11, 35–41.
Kim, S. H., Lechman, E. R., Bianco, N., et al. (2005) Exosomes derived from IL-10-treated dendritic cells can suppress inflammation and collagen-induced arthritis. J. Immunol. 174, 6440–6448.
Hee Kim, S., Bianco, N., Menon, R., et al. (2005) Exosomes derived from genetically modified DC expressing FasL are anti-inflammatory and immunosuppressive. Mol. Ther. 13, 289–300.
Son, Y. L, Egawa, S., Tatsumi, T., Redlinger, R. E., Jr., Kalinski, P., and Kanto, T. (2002) A novel bulk-culture method for generating mature dendritic cells from mouse bone marrow cells. J. Immunol. Methods 262, 145–157.
Thery, C., Boussac, M., Veron, P., et al. (2001) Proteomic analysis of dendritic cell-derived exosomes: a secreted subcellular compartment distinct from apoptotic vesicles. J. Immunol. 166, 7309–7318.
Wubbolts, R., Leckie, R. S., Veenhuizen, P. T., et al. (2003) Proteomic and biochemical analyses of human B cell-derived exosomes. Potential implications for their function and multivesicular body formation. J. Biol. Chem. 278, 10,963–10,972.
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2007 Humana Press Inc., Totowa, NJ
About this protocol
Cite this protocol
Bianco, N.R., Kim, SH., Morelli, A.E., Robbins, P.D. (2007). Modulation of the Immune Response Using Dendritic Cell-Derived Exosomes. In: Fairchild, P.J. (eds) Immunological Tolerance. Methods in Molecular Biology™, vol 380. Humana Press. https://doi.org/10.1007/978-1-59745-395-0_28
Download citation
DOI: https://doi.org/10.1007/978-1-59745-395-0_28
Publisher Name: Humana Press
Print ISBN: 978-1-58829-652-8
Online ISBN: 978-1-59745-395-0
eBook Packages: Springer Protocols