Abstract
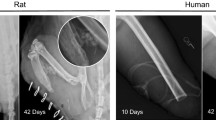
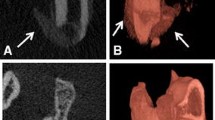
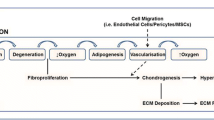
Heterotopic ossification (HO), characterized by the formation of mature bone in the soft tissues, is a complication that can accompany musculoskeletal injury, and it is a frequent occurrence within the military population that has experienced orthopaedic combat trauma. The etiology of this disease is largely unknown. Our laboratory has developed strategies to investigate the cellular and molecular events leading to HO using clinical specimens that were obtained during irrigation and debridement of musculoskeletal injuries. Our approach enables to study (1) the cell types that are responsible for pathological transformation and ossification, (2) the cell- and tissue-level signaling that induces the pathologic transformation, and (3) the effect of extracellular matrix topography and force transduction on HO progression. In this review, we will report on our findings in each of these aspects of HO etiology and describe our efforts to recapitulate our findings in an animal model for traumatic HO.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Potter BK et al (2007) Heterotopic ossification following traumatic and combat-related amputations. Prevalence, risk factors, and preliminary results of excision. J Bone Joint Surg Am 89(3):476–86
Potter BK et al (2006) Heterotopic ossification in the residual limbs of traumatic and combat-related amputees. J Am Acad Orthop Surg 14(10 Spec No.):S191–7
Pape HC et al (2004) Current concepts in the development of heterotopic ossification. J Bone Joint Surg Br 86(6):783–7
Stover SL, Hataway CJ, Zeiger HE (1975) Heterotopic ossification in spinal cord-injured patients. Arch Phys Med Rehabil 56(5):199–204
Peterson SL et al (1989) Postburn heterotopic ossification: insights for management decision making. J Trauma 29(3):365–9
Vanden Bossche L et al (2008) Free radical scavengers have a preventive effect on heterotopic bone formation following manipulation of immobilized rabbit legs. Eur J Phys Rehabil Med 44(4):423–8
Shore EM et al (2006) A recurrent mutation in the BMP type I receptor ACVR1 causes inherited and sporadic fibrodysplasia ossificans progressiva. Nat Genet 38(5):525–7
Shafritz AB et al (1996) Overexpression of an osteogenic morphogen in fibrodysplasia ossificans progressiva. N Engl J Med 335(8):555–61
Ahn J et al (2003) Paresis of a bone morphogenetic protein-antagonist response in a genetic disorder of heterotopic skeletogenesis. J Bone Joint Surg Am 85-A(4):667–74
Shore EM, Kaplan FS (2008) Insights from a rare genetic disorder of extra-skeletal bone formation, fibrodysplasia ossificans progressiva (FOP). Bone 43(3):427–33
Nesti LJ et al (2008) Differentiation potential of multipotent progenitor cells derived from war-traumatized muscle tissue. J Bone Joint Surg Am 90(11):2390–8
Chamberlain G et al (2007) Concise review: mesenchymal stem cells: their phenotype, differentiation capacity, immunological features, and potential for homing. Stem Cells 25(11):2739–2749
Owen ME, Cave J, Joyner CJ (1987) Clonal analysis in vitro of osteogenic differentiation of marrow CFU-F. J Cell Sci 87(5):731–738
Grefte S et al (2007) Skeletal muscle development and regeneration. Stem Cells Dev 16(5):857–68
Zheng B et al (2007) Prospective identification of myogenic endothelial cells in human skeletal muscle. Nat Biotechnol 25(9):1025–34
Lumelsky NL (2007) Commentary: engineering of tissue healing and regeneration. Tissue Eng 13(7):1393
Caplan AI, Dennis JE (2006) Mesenchymal stem cells as trophic mediators. J Cell Biochem 98(5):1076–84
Jackson WM et al (2010) Differentiation and regeneration potential of mesenchymal progenitor cells derived from traumatized muscle tissue. J Cell Mol Med 2010 Dec 3. doi: 10.1111/j.1582-4934.2010.01225.x. [Epub ahead of print]
Sivakumar P, Das A (2008) Fibrosis, chronic inflammation and new pathways for drug discovery. Inflamm Res 57(9):410–418
Jackson WM et al (2009) Mesenchymal progenitor cells derived from traumatized human muscle. J Tissue Eng Regen Med 3(2):129–38
Jackson WM et al (2009) Putative heterotopic ossification progenitor cells derived from traumatized muscle. J Orthop Res 27(12):1645–51
Velnar T, Bailey T, Smrkolj V (2009) The wound healing process: an overview of the cellular and molecular mechanisms. J Int Med Res 37(5):1528–42
Singer AJ, Clark RA (1999) Cutaneous wound healing. N Engl J Med 341(10):738–46
Quintero AJ et al (2009) Stem cells for the treatment of skeletal muscle injury. Clin Sports Med 28(1):1–11
Jackson WM et al (2011) Cytoke expression in muscle following traumatic injury. J Orthop Res 2011 Mar 30. doi: 10.1002/jor.21354. [Epub ahead of print]
Cowin AJ et al (2001) Expression of TGF-beta and its receptors in murine fetal and adult dermal wounds. Eur J Dermatol 11(5):424–31
Lu L et al (2005) The temporal effects of anti-TGF-beta1, 2, and 3 monoclonal antibody on wound healing and hypertrophic scar formation. J Am Coll Surg 201(3):391–7
Schofield R (1978) The relationship between the spleen colony-forming cell and the haemopoietic stem cell. Blood Cells 4(1–2):7–25
Jones DL, Wagers AJ (2008) No place like home: anatomy and function of the stem cell niche. Nat Rev Mol Cell Biol 9(1):11–21
Lutolf MP, Hubbell JA (2005) Synthetic biomaterials as instructive extracellular microenvironments for morphogenesis in tissue engineering. Nat Biotechnol 23(1):47–55
Discher DE, Mooney DJ, Zandstra PW (2009) Growth factors, matrices, and forces combine and control stem cells. Science 324(5935):1673–7
Place ES, Evans ND, Stevens MM (2009) Complexity in biomaterials for tissue engineering. Nat Mater 8(6):457–70
Guilak F et al (2009) Control of stem cell fate by physical interactions with the extracellular matrix. Cell Stem Cell 5(1):17–26
Lutton C, Goss B (2008) Caring about microenvironments. Nat Biotechnol 26(6):613–4
Lim SH, Mao HQ (2009) Electrospun scaffolds for stem cell engineering. Adv Drug Deliv Rev 61(12):1084–96
Li WJ et al (2005) Multilineage differentiation of human mesenchymal stem cells in a three-dimensional nanofibrous scaffold. Biomaterials 26(25):5158–66
Yim EK, Pang SW, Leong KW (2007) Synthetic nanostructures inducing differentiation of human mesenchymal stem cells into neuronal lineage. Exp Cell Res 313(9):1820–9
Baker BM, Mauck RL (2007) The effect of nanofiber alignment on the maturation of engineered meniscus constructs. Biomaterials 28(11):1967–77
Sefcik LS et al (2008) Collagen nanofibres are a biomimetic substrate for the serum-free osteogenic differentiation of human adipose stem cells. J Tissue Eng Regen Med 2(4):210–20
Nie H et al (2008) Three-dimensional fibrous PLGA/HAp composite scaffold for BMP-2 delivery. Biotechnol Bioeng 99(1):223–34
Engler AJ et al (2006) Matrix elasticity directs stem cell lineage specification. Cell 126(4):677–89
Shih YR et al (2006) Growth of mesenchymal stem cells on electrospun type I collagen nanofibers. Stem Cells 24(11):2391–7
Michelsson JE, Granroth G, Andersson LC (1980) Myositis ossificans following forcible manipulation of the leg. A rabbit model for the study of heterotopic bone formation. J Bone Joint Surg Am 62(5):811–5
Lin L et al (2006) Adenovirus-mediated transfer of siRNA against Runx2/Cbfa1 inhibits the formation of heterotopic ossification in animal model. Biochem Biophys Res Commun 349(2):564–72
O’Connor JP (1998) Animal models of heterotopic ossification. Clin Orthop Relat Res 346:71–80
Kaplan FS et al (2004) Heterotopic ossification. J Am Acad Orthop Surg 12(2):116–25
Acknowledgements
This work was supported acknowledged as part of the NIH Intramural Research Program. (Z01 AR41131 and 1ZIAAR041191), grants from the Department of Defense Military Amputee Research Program at WRAMC (PO5-A011), Comprehensive Neurosciences Program (CNP-2008-CR01) and Peer-Reviewed Orthopaedic Research Program (W81XWH-10-2-0084).
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2011 Springer Science+Business Media, LLC
About this chapter
Cite this chapter
Ji, Y., Christopherson, G.T., Kluk, M.W., Amrani, O., Jackson, W.M., Nesti, L.J. (2011). Heterotopic Ossification Following Musculoskeletal Trauma: Modeling Stem and Progenitor Cells in Their Microenvironment. In: Rhim, J., Kremer, R. (eds) Human Cell Transformation. Advances in Experimental Medicine and Biology, vol 720. Springer, New York, NY. https://doi.org/10.1007/978-1-4614-0254-1_4
Download citation
DOI: https://doi.org/10.1007/978-1-4614-0254-1_4
Published:
Publisher Name: Springer, New York, NY
Print ISBN: 978-1-4614-0253-4
Online ISBN: 978-1-4614-0254-1
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)