INTRODUCTION
CDX2 is a nuclear homeobox transcription factor that belongs to the caudal-related family of CDX homeobox genes[1-3]. The gene encoding CDX2 is a nonclustered hexapeptide located on chromosome 13q12-13[1,2]. Homeobox genes play an essential role in the control of normal embryonic development[1,2]. CDX2 is crucial for axial patterning of the alimentary tract during embryonic development[4,5] and is involved in the processes of intestinal cell proliferation, differentiation, adhesion, and apoptosis[4-6]. CDX2 functions within the cell to induce differentiation and inhibit proliferation at the level of gene transcription[4]. It stimulates intestinal epithelium differentiation through activating the transcription of proteins specific to the intestine, such as MUC2, sucrase, isomaltase, and carbonic anhydrase I[4,5]. CDX2 inhibits epithelial proliferation through upregulating WAF1/p21, a cdk inhibitor that arrests the cell cycle upon DNA damage[6]. CDX2 expression has been reported to be organ specific and is normally expressed throughout embryonic and postnatal life within the nuclei of epithelial cells of the alimentary tract from the proximal duodenum to the distal rectum[4-7].
The majority of homeobox genes are considered as proto-oncogenes, with few exceptions[8]. Expression of CDX2 decreases in human colorectal cancers in proportion to the tumor grade and it is lost in minimally differentiated colon carcinomas[9]. In addition, CDX2 is downregulated by oncogenic pathways in colon cancer cells. These observations have suggested that CDX2 has a tumor suppressor function. In addition, Bonhomme et al[8] have provided experimental evidence that CDX2 is a colon tumor suppressor gene. Unlike other colon tumor suppressor genes such as APC and p53[10], which act also outside the gut, CDX2 is the first intestine-specific tumor suppressor[8].
Since CDX2 is a transcription factor, it shows a nuclear immunostaining pattern. In practice, nuclear expression of transcription factors has several distinct advantages over cytoplasmic “differentiation” markers. Firstly, transcription factors generally yield an “all or none” signal, with the vast majority of positive cases containing positive signal in > 90% of the target cell population. Secondly, the nuclear localization of the signal is much less likely to be confused with biotin or other sources of false-positive cytoplasmic signals. Third, there is no association between the levels of expression of nuclear transcription factors and the state of differentiation of the tumor.
CDX2 EXPRESSION IN GASTROINTESTINAL TUMORS
CDX2 expression in colorectal carcinoma
CDX2 is expressed in normal small and large intestinal epithelial cells, including absorptive, endocrine and Paneth cells[4]. Recent immunohistochemical studies have reported that CDX2 is a specific and sensitive marker for adenocarcinoma of the gastrointestinal tract, particularly colorectal adenocarcinoma[7,11-14]. Moskaluk et al[12], examined CDX2 expression in tissue microarrays containing 745 cancers from many anatomic sites and observed strong positive staining in 90% of colonic adenocarcinomas, 20%-30% of carcinomas of the stomach, esophagus and ovary (limited to endometrioid and mucinous types) and in less than 1% of all other carcinoma types. Another study conducted by De Lott et al[13], investigated CDX2 expression in tissue microarrays from 71 colorectal adenocarcinomas, 47 lung adenocarcinomas, 31 hepatocellular carcinomas, 55 squamous cell carcinomas of the lung, 69 neuroendocrine carcinomas of the lung, 43 neuroendocrine carcinomas of the pancreas, 57 pancreatic adenocarcinomas, and 256 endometrial adenocarcinomas. Positive results were found in about 72% of colorectal cancers and in only 6% of endometrial carcinomas[13]. Tumors from other sites were either negative or rarely positive. Similarly, Werling et al[14] found CDX2 expression in the majority of colorectal carcinomas, with only few exceptions. A heterogeneous focal staining pattern was found in pancreatic, gastric and gastroesophageal adenocarcinomas and cholangiocarcinomas. CDX2 was rarely expressed in carcinomas of the breast, genitourinary tracts, gynecologic tracts, lung, head and neck[14]. Bakaris et al[15] observed that CDX2 expression was seen in all cases of colonic adenoma, and the majority of colorectal adenocarcinomas. These previous studies have illustrated the value of CDX2 expression in determining tumor origin in the diagnostic settings[12].
Previous studies have reported a wide variation in the proportion of colorectal adenocarcinomas that express CDX2. Some studies have reported its expression in 98% to 100% of cases, while others have observed loss of CDX2 expression in 14% to 37% of cases[7,12,16]. Loss of CDX2 expression in colorectal cancer has been found to correlate with high tumor grade, microsatellite instability or advanced tumor stage[7,15,17]. Considering the role of CDX2 in promoting cellular differentiation and inhibiting proliferation[2,3], loss of CDX2 expression could conceivably contribute to aggressive tumor behavior and increase the likelihood of metastatic disease[17]. Choi et al[18] analyzed the expression of CDX2 in 123 cases of sporadic colorectal cancers and found loss of its expression in 29/123 (23.6%) specimens. Again, this loss of expression was found to correlate with higher tumor stage and positive lymph node metastasis[18]. Loss of CDX2 is also more frequently encountered in mismatch repair-deficient colorectal cancer[9]. Utilizing a database of 621 colorectal cancers, Baba et al[9] found that CDX2 loss was correleted directly with female gender, high tumor grade, stage IV disease, and inversely with LINE-1 hypomethylation, p53 expression, and β-catenin activation. CDX2 loss was associated with high overall mortality among patients with a family history of colorectal cancer[9]. This implies the importance of CDX2 in the suppression of tumorgenesis in a subset of colorectal cancers and its potential for use as a prognostic marker in identifying high risk patients.
Rectal adenocarcinomas are commonly lumped together with colonic tumors, making their proper immunoprofiling difficult[19]. We recently investigated the expression of CDX2, along with CK7 and CK20, in rectal adenocarcinoma. In our experience, CDX2 was expressed in the majority of cases of rectal adenocarcinoma, and staining was predominantly nuclear with occasional faint cytoplasmic staining[19]. In our study, CDX2 expression did not appear to correlate with tumor grade (tumor differentiation).
CDX2 expression in esophageal neoplasms
CDX2 is not expressed in normal esophageal and gastric epithelial cells but is expressed in intestinal metaplasia of the esophagus[20-22]. In some patients, Barrett’s esophagus is complicated by the development of esophageal adenocarcinoma[20,21]. Lord et al[22] investigated the expression of CDX2 and PITX1 in Barrett’s esophagus and associated adenocarcinoma. Negative CDX2 staining was observed in normal squamous esophageal lining, while strong (3+) nuclear staining was seen in all cases of Barrett’s intestinal metaplasia, dysplasia, and associated adenocarcinoma[22]. The level of CDX2 mRNA expression was found to coincide with immunohistochemical CDX2 expression as both were upregulated in Barrett’s intestinal metaplasia tissues and remained elevated in dysplastic and adenocarcinoma cells[22]. In contrast, a recent study has reported CDX2 expression which was significantly weaker or absent in esophageal dysplasia and adenocarcinoma in comparison to metaplastic cells[23].
CDX2 expression in gastric adenocarcinoma
Gastric carcinoma is frequently found in association with intestinal metaplasia[24]. Studies have reported CDX2 expression in both intestinal metaplasia of the stomach and intestinal-type gastric carcinoma[25-31]. Furthermore, incomplete intestinal metaplasia, which expresses both gastric and intestinal mucins, shows lower CDX2 expression compared with complete intestinal metaplasia[32]. Although incomplete intestinal metaplasia morphologically resembles colon, its CDX2 expression was apparently lower than that seen in the normal colon. Similar to esophageal dysplasia, previous studies showed decreasing CDX2 expression from metaplasia to dysplasia to adenocarcinoma[32]. Intestinal metaplasia or dysplasia with low expression of CDX2 may potentially serve as predictive markers for gastric cancer[32].
Song et al[33] reported a significantly better outcome for CDX2-positive gastric tumors over CDX2-negative tumors. Other studies have similarly demonstrated that positive CDX2 expression in gastric cancer significantly correlated with better differentiation and prognosis[34,35]. CDX2 expression has been evaluated in 69 cases of gastric epithelial dysplasia, 88 early gastric cancers and 56 advanced gastric cancers. Increased CDX2 expression was more frequently associated with adenomatous-type gastric epithelial dysplasia (87%), compared with the foveolar (47%) or hybrid (44%) types. CDX2 expression levels also gradually decreased from gastric dysplasia, to early and advanced gastric cancers. Moreover, a negative correlation was observed between CDX2 expression and the depth of tumor invasion[26]. A recent study showed that absence of nuclear CDX2 expression may serve as a powerful predictor of lymph node metastasis in gastric cancer[36]. Overexpression of CDX2 has recently been shown to inhibit cell growth and proliferation in vitro and can effectively inhibit gastric cancer progression, making this a potential therapeutic target[37].
CDX2 expression in small intestinal adenocarcinoma
Despite the large surface area, malignancies of the small intestine are quite rare and account for only 2% of primary gastrointestinal tumors[38]. Small intestinal adenocarcinoma shows similarities in morphology and risk factors with its colorectal counterpart[38]. However, it has been found to be immunophenotypically distinct from colorectal adenocarcinoma. Zhang et al[38] examined the expression of CDX2 in small intestinal adenocarcinoma and found that CDX2 was expressed in 60% of cases of small intestinal adenocarcinoma in comparison to 98% of colorectal adenocarcinoma.
CDX2 expression in gallbladder adenocarcinoma
Gallbladder adenocarcinoma is a highly malignant neoplasm with variable incidence depending on gender and geographic distribution[39]. Sakamoto et al[40] investigated the expression of CDX2 in human gallbladders with cholelithiasis and reported CDX2 expression in 92% of gallbladder intestinal metaplasias. CDX2 expression has been found in dysplasia, carcinoma and intestinal metaplasia of the gallbladder and carcinogenesis may proceed through intestinal metaplasia as seen in esophageal metaplasia[39,40].
Wu et al[39] examined the expression of CDX2 in 68 primary gallbladder carcinomas and compared its expression with various clinicopathologic factors. Positive staining was observed in 25/68 (36.8%) cases with no significant correlation with clinicopathologic prognostic parameters. Well-differentiated carcinomas had high CDX2 expression (54.8%) compared to moderately differentiated (7.1%) and poorly differentiated carcinomas (0%)[39]. In contrast, Chang et al[41] reported CDX2 positivity in 29% of their cases and that expression was an independent prognostic factor in patients with biliary tract carcinoma.
CDX2 expression in extrahepatic bile duct and pancreatic adenocarcinoma
Hong et al[42] found CDX2 expression in 37% of their extrahepatic bile duct carcinoma cases. They observed more frequent CDX2 expression in tumors with papillary growth (60%) than in those with a nodular (25%) or infiltrative (34.9%) pattern. CDX2 expression was also more frequent in cases without vascular invasion (41.3%) than in those with vascular invasion (23%). In univariant analysis, CDX2/MUC2 positive patients had a significantly higher survival rate than negative patients[42].
CDX2 expression is focal and patchy in normal pancreatic epithelium[20] and CDX2 is infrequently expressed in pancreatic adenocarcinoma. In our experience, CDX2 is focally expressed in less than 10% of pancreatic duct adenocarcinomas[19]. Another report found CDX2 expression in only 3 of the 57 (5%) pancreatic adenocarcinoma cases studied[13]. In general, the staining pattern is usually focal and less intense than that found in colorectal adenocarcinoma.
CDX2 expression in gastrointestinal neuroendocrine tumors
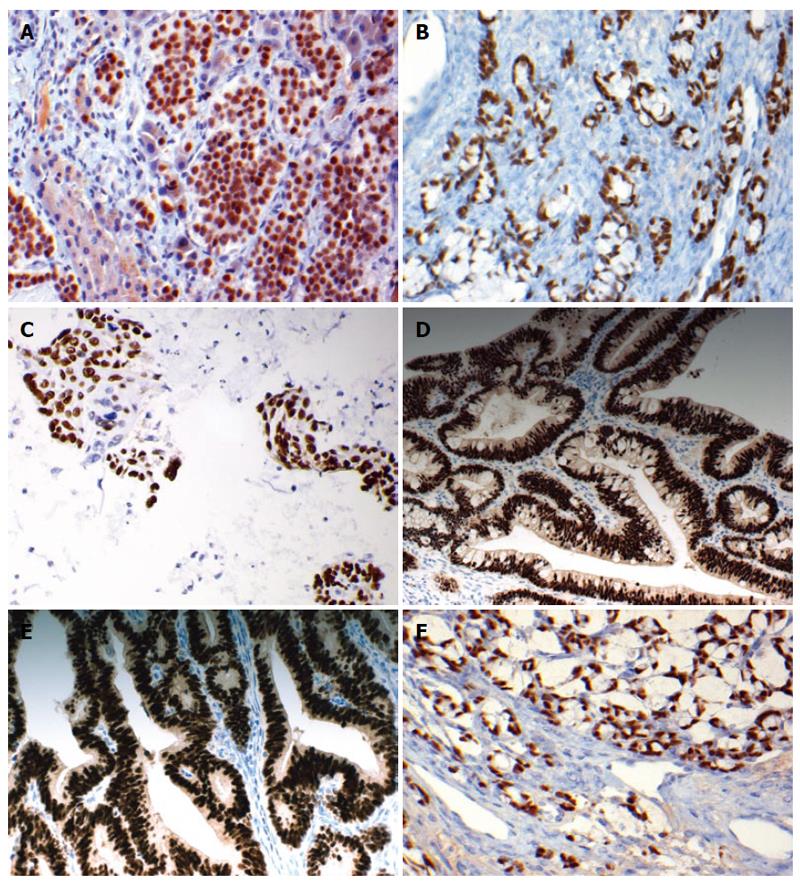
We have also examined the use of CDX2 and TTF1 in differentiating metastatic neuroendocrine neoplasms of unknown origin[43]. Expression of CDX2 was found in 28/60 (47%) gastrointestinal neuroendocrine tumors with high prevalence in ileal, appendiceal and colonic origin[43] (Figure 1). Similarly, previous studies documented exclusive positive staining for CDX2 in ileal and appendiceal neuroendocrine tumors, while all rectal, gastric and duodenal neuroendocrine tumors were negative[44,45]. No CDX2 expression was observed in neuroendocrine tumors of other origins, including skin, ovary or thymus[43]. Pancreatic endocrine tumors show focal and heterogeneous staining for CDX2[43].
Figure 1 CDX2 expression.
A: CDX2 expression in metastatic ileal carcinoid to the liver. Please note the presence of moderate nuclear staining; B: CDX2 expression is also seen in appendiceal goblet carcinoid (Immunohistochemistry × 400); C: CDX2 expression in cytology specimens of metastatic colonic carcinoma to the lung, supporting their colorectal origin. (Immunohistochemistry × 250); D: CDX2 expression is seen in the majority of endocervical adenocarcinomas of intestinal type (Immunohistochemistry × 400); E: CDX2 expression in endometrioid carcinoma (Immunohistochemistry × 400); F: CDX2 expression in sinonasal adenocarcinoma of intestinal type (Immunohistochemistry × 400).
Rabban et al[46] have evaluated CDX2 expression in metastatic and primary ovarian carcinoids. They reported diffuse nuclear CDX-2 expression in majority of primary ovarian and metastatic intestinal carcinoids involving the ovary, particularly insular and mucinous types. They concluded that CDX2 is not specific and cannot be used to determine the site of ovarian carcinoids. All primary ovarian carcinoids were negative for TTF-1, CK7 and CK20[46].
USE OF CDX2 TO CONFIRM METASTATIC ADENOCARCINOMA OF A GASTROINTESTINAL ORIGIN
CDX2 immunohistochemical staining for diagnosis of metastatic adenocarcinoma has recently come into practice[47-49]. Due to its limited expression in the spectrum of human tissues and neoplasia, CDX2 has been investigated for its usefulness in diagnosing a metastatic adenocarcinoma as being of a gastrointestinal origin[47-49].
The diagnostic utility of CDX2 as a marker to identify the gastrointestinal origin of a metastatic tumor was addressed a study where we used CDX2 to distinguish bronchioloalveolar adenocarcinoma of the lung from metastatic mucinous colorectal adenocarcinoma[42]. Using surgical material, Saad et al[47] and Barbareschi et al[49] both found CDX2 expression in metastatic colorectal adenocarcinoma to the lung compared but absent in primary lung adenocarcinoma. Similarly, CDX2 was useful in cytology specimens as a marker of metastatic gastrointestinal adenocarcinoma when compared to other metastatic tumors (Figure 1C). CDX2 expression was found in 19/22 (86%) confirmed metastatic gastrointestinal specimens[50]. All other metastatic adenocarcinomas, from lung, breast, ovaries, pancreas, and prostate sites, were negative for CDX2[50]. Similarly, Lora and Kanitakis investigated CDX2 expression in 68 cutaneous metastatic tumors of various origin and found that CDX2 was a specific immunohistochemical marker for cutaneous metastases from intestinal and urothelial carcinomas[48].
Expression of CDX2 tumors outside the colorectum has been reported[12,14]. Tot[51] reported that the CK20+/CK7- pattern is more specific than CDX2 expression in predicting the colorectal origin of metastatic adenocarcinoma. It is usually recommended to use CDX2 as a part of immunostaining panel including CK7, CK20, mCEA and villin to prove the intestinal origin of a metastatic tumor[50].
DIAGNOSTIC DILEMMAS WITH ABERRANT CDX2 EXPRESSION
Despite the relatively restricted CDX2 expression profile, expression of CDX2 in tumors outside the colorectum has been previously reported. Moskaluk et al[12] and Werling et al[14] reported that a significant fraction of ovarian mucinous carcinomas and primary bladder adenocarcinomas were CDX2-positive and there have been other studies reporting CDX2 expression in adenocarcinomas of various anatomic sites.
CDX2 expression in cervical adenocarcinoma
Intestinal differentiation of cervical adenocarcinoma, in the form of goblet cells and/or Paneth cells, is uncommon but may generate diagnostic dilemmas. Invasive cervical adenocarcinomas with intestinal differentiation could mimic the histology of colorectal adenocarcinoma, raising the possibility of metastasis or direct spread. Also, a distant metastasis from an intestinal type cervical adenocarcinoma could be easily mistaken for a metastatic adenocarcinoma of an intestinal origin, based on morphology alone.
CDX2 immunostaining has been studied in a few large series of cervical adenocarcinomas of various histologic subtypes. McCluggage et al[52] have recently reported CDX2 positivity in the majority of intestinal-type endocervical adenocarcinomas in situ (20/21 cases) and in all the three invasive intestinal-type adenocarcinomas (ITAC) studied. We compared the expression of CDX2 in 119 cases of different types of cervical adenocarcinoma with that in rectal adenocarcinoma[53]. Our study is the largest reported to date and confirms that the majority of invasive and in situ endocervical adenocarcinomas of intestinal-type show CDX2 immunoreactivity[53], in agreement with the results of McCluggage et al[52] (Figure 1D).
CDX2 expression in primary ovarian mucinous tumors
Of all ovarian epithelial tumors, mucinous tumors pose the greatest difficulty with regard to differentiation between primary and metastatic tumors. Previous studies have demonstrated conflicting results regarding the value of CDX2 in distinguishing primary tumors from metastatic carcinomas of the ovary with mucinous morphology. CDX2 expression has been reported in from 0 to 100% of ovarian mucinous tumors and in 0% to 30% of ovarian endometrioid carcinomas[54-64]. In contrast, a recent study showed that almost all primary ovarian carcinomas lacked immunoreactivity for CDX2, while the majority of metastatic colorectal carcinomas of the ovary were CDX2-positive[57]. In an attempt to reconcile the wide gap in data from different studies, the authors claimed that previous studies may have misclassified ovarian metastases as primary tumors. Taken together, the results available to date suggest that the differential diagnosis of primary and metastatic mucinous carcinoma still poses a great problem because these tumors can share their immunophenotype, gross and microscopic features.
CDX2 expression in uterine endometrioid adenocarcinoma
Wani et al[65] investigated CDX2 expression in 225 cases of endometrial biopsies including 101 endometrioid carcinomas. Normal and non-proliferative endometrium showed negative CDX2 staining. Endometrioid carcinoma with squamous differentiation was positive for CDX2 in 73% of cases, whereas only 14% of tumors without squamous differentiation were positive (Figure 1E). In addition, the authors found that the larger the number of squamous foci the greater the number of CDX2 positive cells which correlated strongly with nuclear β-catenin expression. This may suggest an important role of CDX2 in squamous morular formation[65].
CDX2 expression in prostatic adenocarcinoma
Herawi et al[66] have investigated CDX2 expression in prostatic adenocarcinoma, including 708 tissue microarrays containing either benign or malignant prostate tissue as well normal tissues from various anatomic sites. Out of 185 prostatic adenocarcinomas, only four cases (6%) showed focal positive staining while benign prostatic tissue was positive in 12% of cases. No cases of metastatic prostatic carcinoma expressed CDX2[66]. Another study found CDX2 expression in 31% of prostatic adenocarcinoma with mucinous or signet cell differentiation[67]. However, in routine pathology practice, positive PSA immunostaining and clinical findings should prove more helpful when a prostatic origin is suspected for a metastatic adenocarcinoma[66,67].
CDX2 expression in urachal adenocarcinoma
The majority of urachal epithelial neoplasms are adenocarcinomas with enteric or nonenteric histologies. Urachal adenocarcinoma may mimic metastatic adenocarcinoma of different origins. Paner et al[68] studied CDX2 expression in 32 urachal adenocarcinomas and reported CDX2 expression in 85% of their cases. CDX2 expression can be diffuse in urachal adenocarcinomas, even without the classic enteric morphology. In urachal adenocarcinoma subtypes, CDX2 expression was see in 8/8 (100%) of mucinous, 10/11 (91%) of enteric type, 5/7 (71%) of not otherwise specified, and in 4/6 (67%) of signet ring cell type. In addition, CDX2 was expressed by urachal remnants of glandular type, and noninvasive urachal mucinous cystic tumors[68].
CDX2 expression in intestinal-type sinonasal adenocarcinoma
ITAC of the nasal cavity and paranasal sinuses are uncommon[69]. They are clinically aggressive and generally present at an advanced stage. Franchi et al[69] demonstrated nuclear expression of CDX2 in all their cases of ITAC, with strong nuclear staining identified in the majority. CDX2 staining was not present in normal respiratory mucosa or seromucous glands. A similar study detected strong and diffuse nuclear expression of CDX2 in all cases of ITAC[70] (Figure 1F). Choi et al[71] suggested that the development of ITAC is preceded by intestinal metaplasia, with conversion from a normal CK7+/CK20-/CDX2-/villin- phenotype to an abnormal CK7-/CK20+/CDX2+/villin- intestinal phenotype.
CDX2 expression in acute myeloid leukemia
CDX2 is expressed in 90% of acute myeloid leukemia (AML) but not in hematopoietic stem and progenitor cells derived from normal individuals[72,73]. Frequent expression of CDX2 in the adult hematopoietic compartment suggests a role for CDX2 as part of a common effector pathway that promotes the proliferative capacity and self-renewal potential of myeloid progenitor cells[73].